Abstract
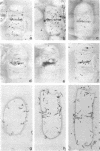
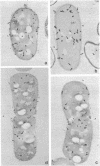
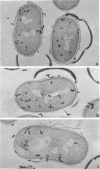
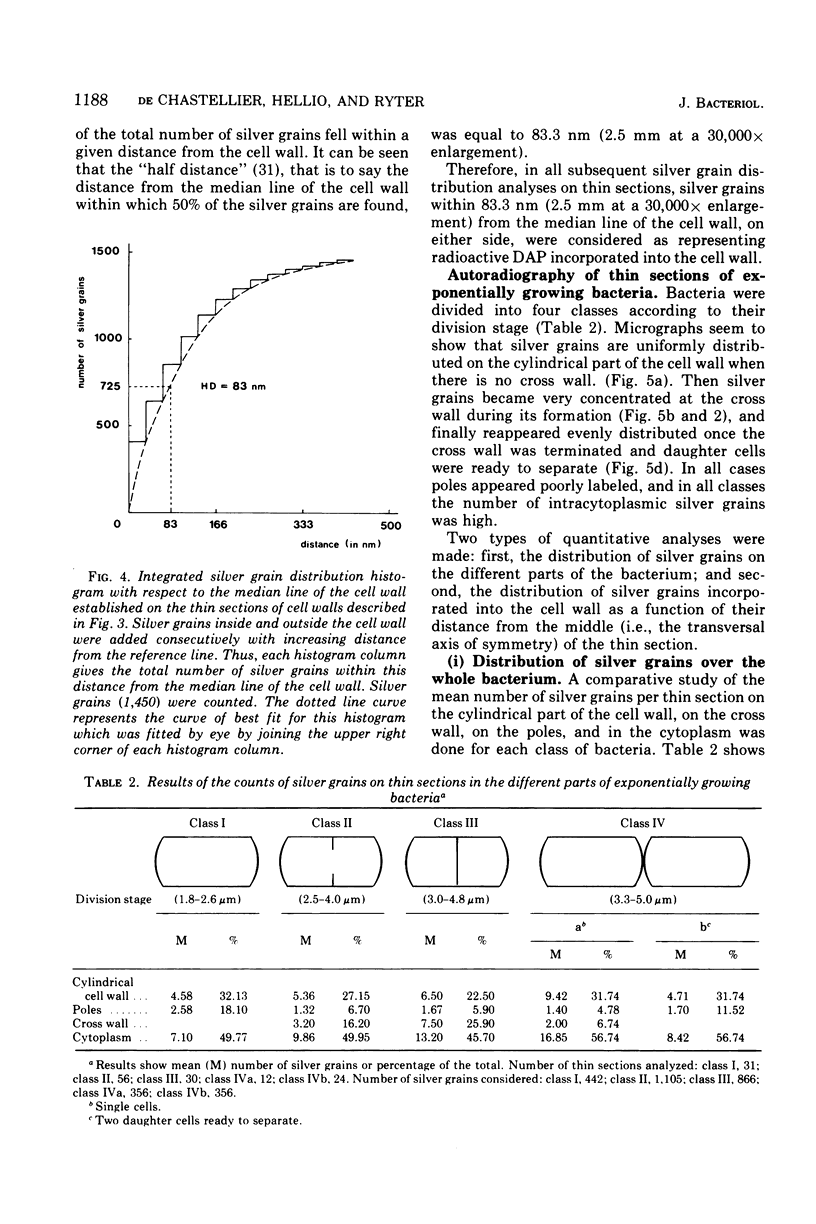
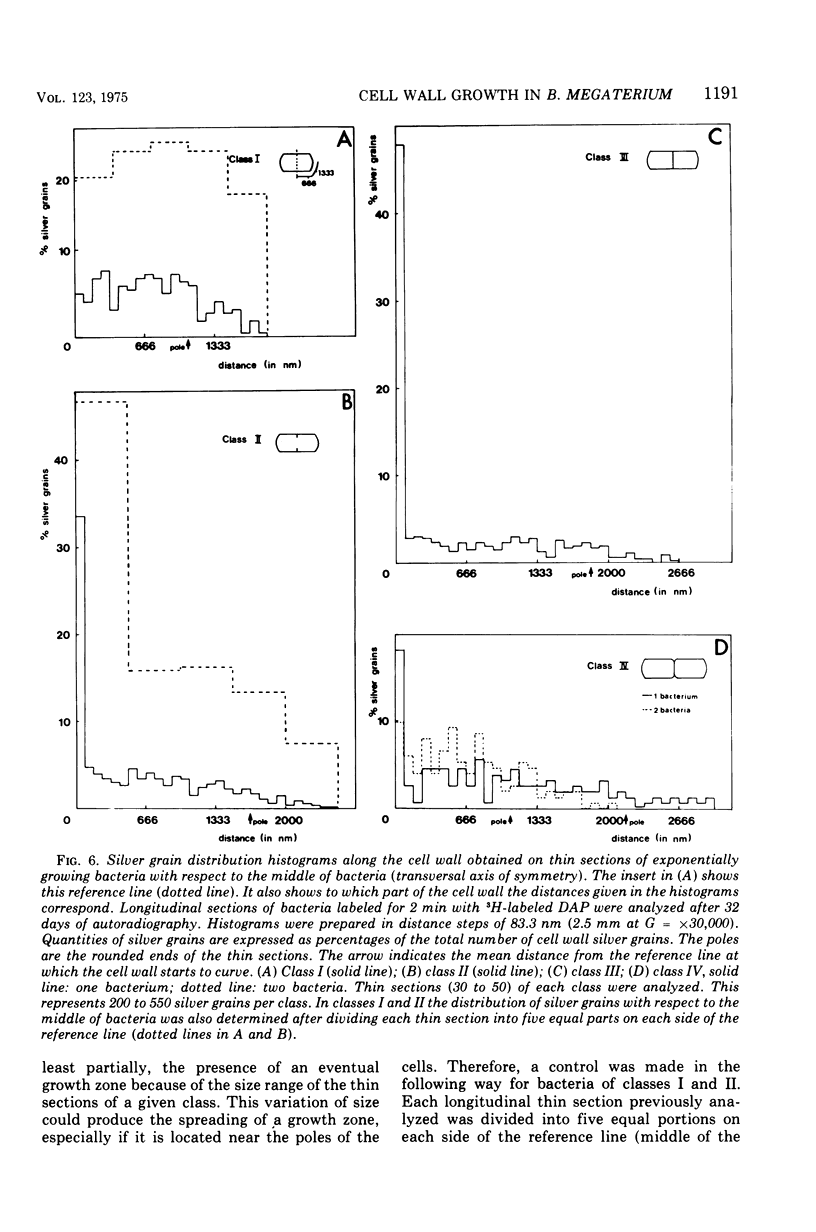
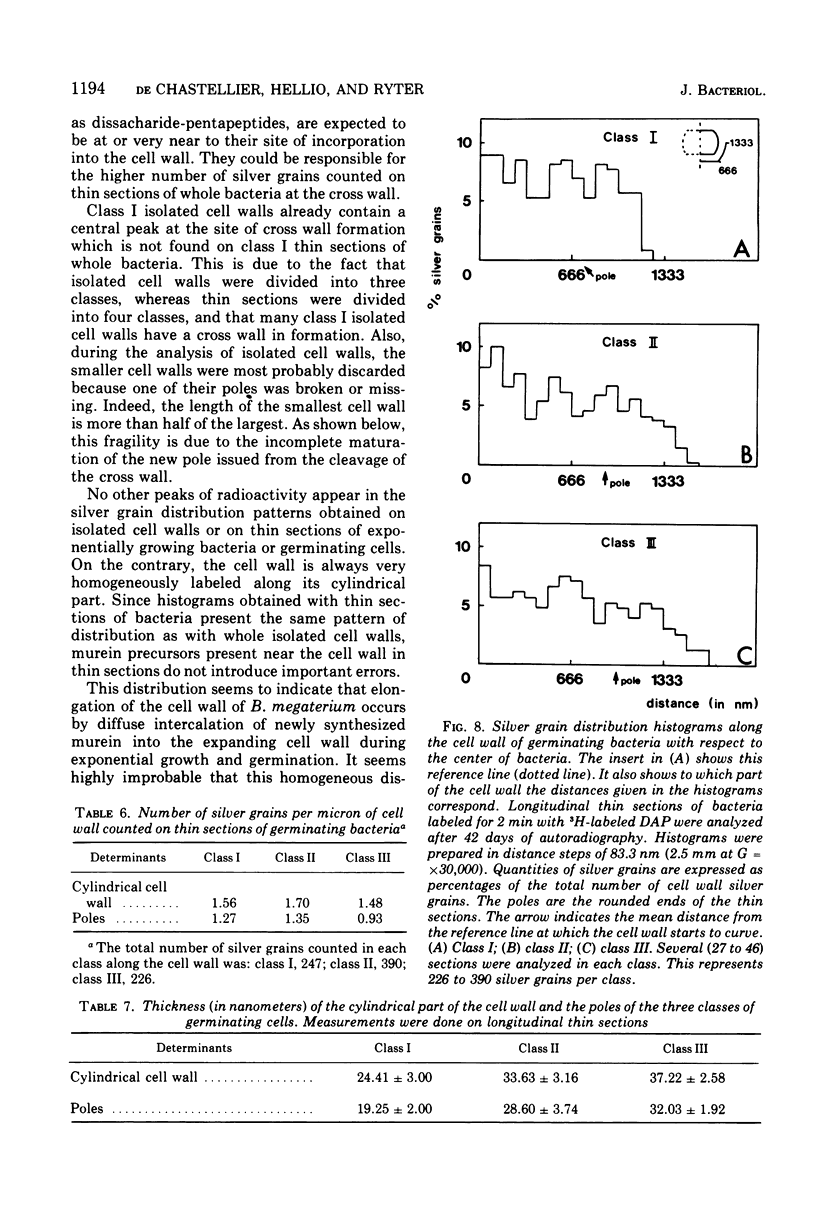
Growth of the cell wall of Bacillus megaterium was studied by pulse-labeling the cell wall of a DAP- Lys- mutant for a very short time with tritium-labeled diaminopimelic acid. The distribution of radioactivity along the cell wall was examined by high-resolution autoradiography on isolated cell walls and thin sections of bacteria. The results indicate that cell wall elongation occurs by diffuse intercalation of newly synthesized murein into the expanding cell wall during exponential growth, as well as during germination, and that the only zone of highly localized diaminopimelic acid incorporation is found at the cross wall during its synthesis. This zone contains about 30% of the radioactivity incorporated into the cell wall. Analysis of autoradiographs of thin sections of bacteria shows that the total radioactivity incorporated per bacterium doubles during the life cycle. This doubling occurs in the cylindrical part of the cell wall but not in the polar caps. This seems to indicate that elongation of the bacterium is not constant during the life cycle but increases with the length of the cell.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- AUBERT J. P., MILLET J., PINEAU E., MILHAUD G. [N-Succinyl-L-glutamic acid in Bacillus megaterium during sporulation]. Biochim Biophys Acta. 1961 Aug 19;51:529–537. doi: 10.1016/0006-3002(61)90610-2. [DOI] [PubMed] [Google Scholar]
- Boothby D., Daneo-Moore L., Higgins M. L., Coyette J., Shockman G. D. Turnover of bacterial cell wall peptidoglycans. J Biol Chem. 1973 Mar 25;248(6):2161–2169. [PubMed] [Google Scholar]
- Briles E. B., Tomasz A. Radioautographic evidence for equatorial wall growth in a gram-positive bacterium. Segregation of choline-3H-labeled teichoic acid. J Cell Biol. 1970 Dec;47(3):786–790. doi: 10.1083/jcb.47.3.786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CHALOUPKA J., KRECKOVA P., RIHOVA L. The mucopeptide turnover in the cell walls of growing cultures of Bacillus megaterium KM. Experientia. 1962 Aug 15;18:362–363. doi: 10.1007/BF02172250. [DOI] [PubMed] [Google Scholar]
- CHALOUPKA J., RIHOVA L., KRECKOVA P. DEGRADATION AND TURNOVER OF BACTERIAL CELL WALL MUCOPEPTIDES IN GROWING BACTERIA. Folia Microbiol (Praha) 1964 Jan;24:9–15. doi: 10.1007/BF02875894. [DOI] [PubMed] [Google Scholar]
- CHUNG K. L., HAWIRKO R. Z., ISAAC P. K. CELL WALL REPLICATION. I. CELL WALL GROWTH OF BACILLUS CEREUS AND BACILLUS MEGATERIUM. Can J Microbiol. 1964 Feb;10:43–48. doi: 10.1139/m64-007. [DOI] [PubMed] [Google Scholar]
- CHUNG K. L., HAWIRKO R. Z., ISAAC P. K. CELL WALL REPLICATION. II. CELL WALL GROWTH AND CROSS WALL FORMATION OF ESCHERICHIA COLI AND STREPTOCOCCUS FAECALIS. Can J Microbiol. 1964 Jun;10:473–482. doi: 10.1139/m64-057. [DOI] [PubMed] [Google Scholar]
- COLE R. M., HAHN J. J. Cell wall replication in Streptococcus pyogenes. Science. 1962 Mar 2;135(3505):722–724. doi: 10.1126/science.135.3505.722. [DOI] [PubMed] [Google Scholar]
- COLLINS J. F., RICHMOND M. H. Rate of growth of Bacillus cereus between divisions. J Gen Microbiol. 1962 Apr;28:15–33. doi: 10.1099/00221287-28-1-15. [DOI] [PubMed] [Google Scholar]
- Caro L. Progress in high-resolution autoradiography. Prog Biophys Mol Biol. 1966;16:171–190. doi: 10.1016/0079-6107(66)90006-x. [DOI] [PubMed] [Google Scholar]
- Chaloupka J. Synthesis and degradation of surface structures by growing and non-growing Bacillus megaterium. Folia Microbiol (Praha) 1967;12(3):264–273. doi: 10.1007/BF02868742. [DOI] [PubMed] [Google Scholar]
- Chung K. L. Thickened cell walls of Bacillus cereus grown in the presence of chloramphenicol: their fate during cell growth. Can J Microbiol. 1971 Dec;17(12):1561–1565. doi: 10.1139/m71-249. [DOI] [PubMed] [Google Scholar]
- Fan D. P., Beckman B. E., Beckman M. M. Cell wall turnover at the hemispherical caps of Bacillus subtilis. J Bacteriol. 1974 Mar;117(3):1330–1334. doi: 10.1128/jb.117.3.1330-1334.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frehel C., Beaufils A. M., Ryter A. Etude au microscope électronique de la croissance de la paroi chez B. subtilis et B. megaterium. Ann Inst Pasteur (Paris) 1971 Aug;121(2):139–148. [PubMed] [Google Scholar]
- Hakenbeck R., Messer W. Activity of murein hydrolases and membrane synthesis in synchronized Escherichia coli B/r. Ann Microbiol (Paris) 1974 Sep;125 B(2):163–166. [PubMed] [Google Scholar]
- Higgins M. L., Shockman G. D. Model for cell wall growth of Streptococcus faecalis. J Bacteriol. 1970 Feb;101(2):643–648. doi: 10.1128/jb.101.2.643-648.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higgins M. L., Shockman G. D. Procaryotic cell division with respect to wall and membranes. CRC Crit Rev Microbiol. 1971 May;1(1):29–72. doi: 10.3109/10408417109104477. [DOI] [PubMed] [Google Scholar]
- Highton P. J., Hobbs D. G. Penicillin and cell wall synthesis: a study of Bacillus cereus by electron microscopy. J Bacteriol. 1972 Mar;109(3):1181–1190. doi: 10.1128/jb.109.3.1181-1190.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Highton P. J., Hobbs D. G. Penicillin and cell wall synthesis: a study of Bacillus licheniformis by electron microscopy. J Bacteriol. 1971 May;106(2):646–658. doi: 10.1128/jb.106.2.646-658.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hughes R. C., Stokes E. Cell wall growth in Bacillus licheniformis followed by immunofluorescence with mucopeptide-specific antiserum. J Bacteriol. 1971 May;106(2):694–696. doi: 10.1128/jb.106.2.694-696.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LUFT J. H. Improvements in epoxy resin embedding methods. J Biophys Biochem Cytol. 1961 Feb;9:409–414. doi: 10.1083/jcb.9.2.409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mauck J., Chan L., Glaser L. Turnover of the cell wall of Gram-positive bacteria. J Biol Chem. 1971 Mar 25;246(6):1820–1827. [PubMed] [Google Scholar]
- Mauck J., Chan L., Glaser L., Williamson J. Mode of cell wall growth of Bacillus megaterium. J Bacteriol. 1972 Jan;109(1):373–378. doi: 10.1128/jb.109.1.373-378.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mauck J., Glaser L. On the mode of in vivo assembly of the cell wall of Bacillus subtilis. J Biol Chem. 1972 Feb 25;247(4):1180–1187. [PubMed] [Google Scholar]
- Mendelson N. H., Reeve J. N. Growth of the Bacillus subtilis cell surface. Nat New Biol. 1973 May 9;243(123):62–64. [PubMed] [Google Scholar]
- REYNOLDS E. S. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol. 1963 Apr;17:208–212. doi: 10.1083/jcb.17.1.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- RYTER A., KELLENBERGER E., BIRCHANDERSEN A., MAALOE O. Etude au microscope électronique de plasmas contenant de l'acide désoxyribonucliéique. I. Les nucléoides des bactéries en croissance active. Z Naturforsch B. 1958 Sep;13B(9):597–605. [PubMed] [Google Scholar]
- Ryter A., Hirota Y., Schwarz U. Process of cellular division in Escherichia coli growth pattern of E. coli murein. J Mol Biol. 1973 Jun 25;78(1):185–195. doi: 10.1016/0022-2836(73)90437-3. [DOI] [PubMed] [Google Scholar]
- Schwarz U., Asmus A., Frank H. Autolytic enzymes and cell division of Escherichia coli. J Mol Biol. 1969 May 14;41(3):419–429. doi: 10.1016/0022-2836(69)90285-x. [DOI] [PubMed] [Google Scholar]
- Swanson J., Hsu K. C., Gotschlich E. C. Electron microscopic studies on streptococci. I. M antigen. J Exp Med. 1969 Nov 1;130(5):1063–1091. doi: 10.1084/jem.130.5.1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Chastellier C., Frehel C., Ryter A. Cell wall growth of Bacillus megaterium: cytoplasmic radioactivity after pulse-labeling with tritiated diaminopimelic acid. J Bacteriol. 1975 Sep;123(3):1197–1207. doi: 10.1128/jb.123.3.1197-1207.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]