Abstract
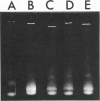
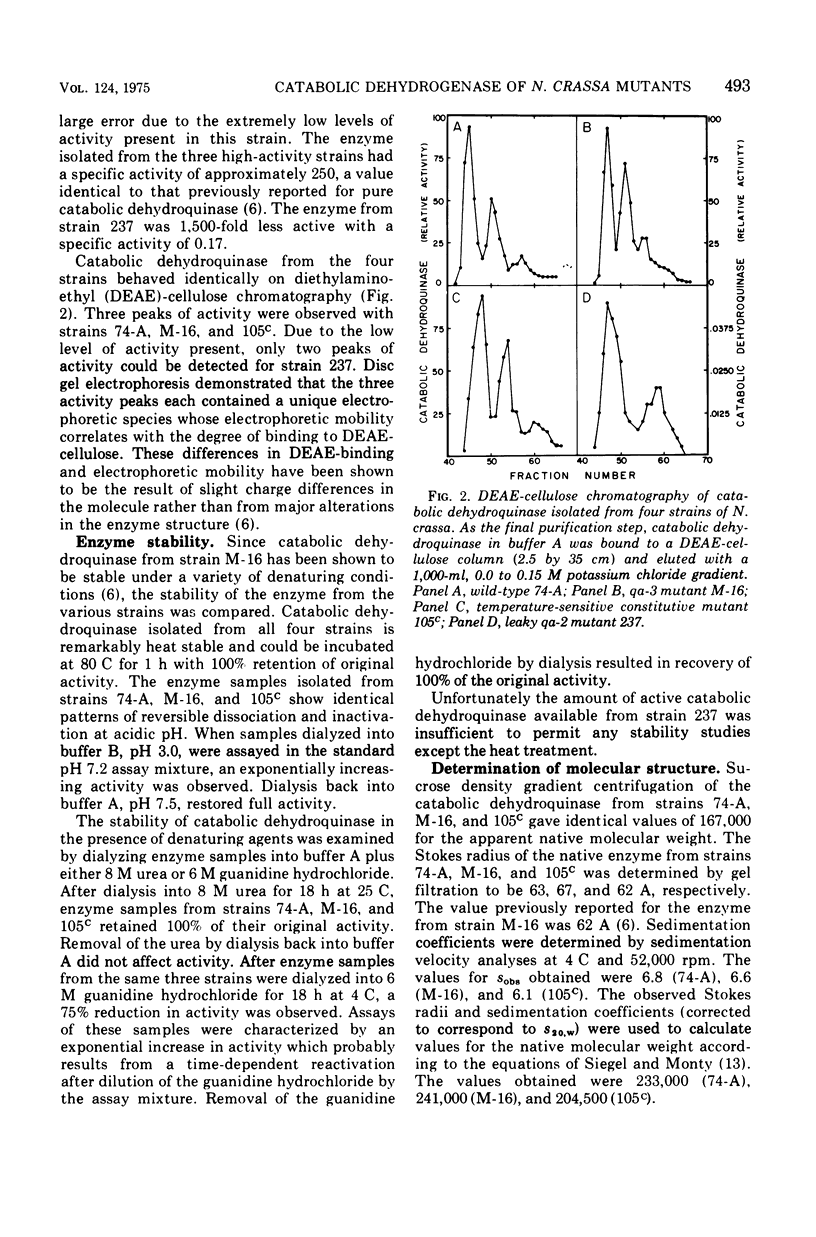
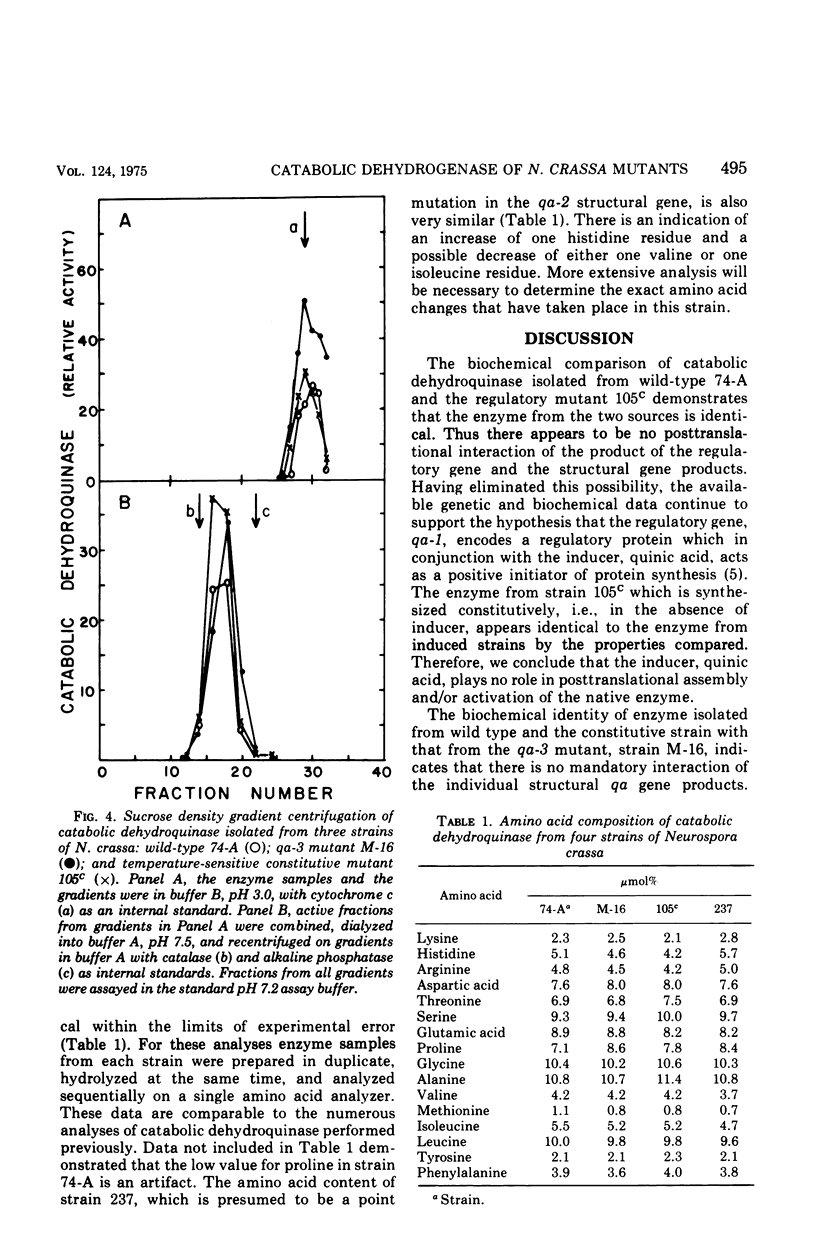
Catabolic dehydroquinase, which functions in the inducible quinic acid catabolic pathway of Neurospora crassa, has been purified from wild type (74-A) and three mutants in the qa gene cluster. The mutant strains were: 105c, a temperature-sensitive constitutive mutant in the qa-1 regulatory locus; M-16, a qa-3 mutant deficient in quinate dehydrogenase activity; and 237, a leaky qa-2 mutant which possess very low levels of catabolic dehydroquinase activity. The enzymes purified from strains 74-A, 105c, and M-16 are identical with respect to behavior during purification, specific activity, electrophoretic behavior, stability, molecular weight, subunit structure, immunological cross-reactivity, and amino acid content. The mutant enzyme from strain 237 is 1,500-fold less active and appears to have a slightly different amino acid content. It is identical by a number of the other criteria listed above and is presumed to be a mutant at or near the enzyme active site. These data demonstrate that the qa-1 gene product is not involved in the posttranslational expression of enzyme activity. The biochemical identity of catabolic dehydroquinase isolated from strains 105c and M-16 with that from wild type also demonstrates that neither the inducer, quinic acid, nor other enzymes encoded in the qa gene cluster are necessary for the expression of activity. Therefore the combined genetic and biochemical data on the qa system continue to support the hypothesis that the qa-1 regulatory protein acts as a positive initiator of qa enzyme synthesis.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Case M. E., Giles N. H., Doy C. H. Genetical and biochemical evidence for further interrelationships between the polyaromatic synthetic and the quinate-shikimate catabolic pathways in Neurospora crassa. Genetics. 1972 Jul;71(3):337–348. doi: 10.1093/genetics/71.3.337. [DOI] [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- Fairbanks G., Steck T. L., Wallach D. F. Electrophoretic analysis of the major polypeptides of the human erythrocyte membrane. Biochemistry. 1971 Jun 22;10(13):2606–2617. doi: 10.1021/bi00789a030. [DOI] [PubMed] [Google Scholar]
- Hautala J. A., Jacobson J. W., Case M. E., Giles N. H. Purification and characterization of catabolic dehydroquinase, an enzyme in the inducible quinic acid catabolic pathway of Neurospora crassa. J Biol Chem. 1975 Aug 10;250(15):6008–6014. [PubMed] [Google Scholar]
- ITZHAKI R. F., GILL D. M. A MICRO-BIURET METHOD FOR ESTIMATING PROTEINS. Anal Biochem. 1964 Dec;9:401–410. doi: 10.1016/0003-2697(64)90200-3. [DOI] [PubMed] [Google Scholar]
- MARTIN R. G., AMES B. N. A method for determining the sedimentation behavior of enzymes: application to protein mixtures. J Biol Chem. 1961 May;236:1372–1379. [PubMed] [Google Scholar]
- Mancini G., Carbonara A. O., Heremans J. F. Immunochemical quantitation of antigens by single radial immunodiffusion. Immunochemistry. 1965 Sep;2(3):235–254. doi: 10.1016/0019-2791(65)90004-2. [DOI] [PubMed] [Google Scholar]
- Siegel L. M., Monty K. J. Determination of molecular weights and frictional ratios of proteins in impure systems by use of gel filtration and density gradient centrifugation. Application to crude preparations of sulfite and hydroxylamine reductases. Biochim Biophys Acta. 1966 Feb 7;112(2):346–362. doi: 10.1016/0926-6585(66)90333-5. [DOI] [PubMed] [Google Scholar]