Abstract
Current nutritional approaches to metabolic syndrome and type 2 diabetes generally rely on reductions in dietary fat. The success of such approaches has been limited and therapy more generally relies on pharmacology. The argument is made that a re-evaluation of the role of carbohydrate restriction, the historical and intuitive approach to the problem, may provide an alternative and possibly superior dietary strategy. The rationale is that carbohydrate restriction improves glycemic control and reduces insulin fluctuations which are primary targets. Experiments are summarized showing that carbohydrate-restricted diets are at least as effective for weight loss as low-fat diets and that substitution of fat for carbohydrate is generally beneficial for risk of cardiovascular disease. These beneficial effects of carbohydrate restriction do not require weight loss. Finally, the point is reiterated that carbohydrate restriction improves all of the features of metabolic syndrome.
Background
The epidemic of diabetes continues unabated, and impassioned calls for better treatment and prevention strategies are common features of scientific conferences. While it is generally acknowledged that total dietary carbohydrate is the major factor in glycemic control, strategies based on reduction of dietary carbohydrate have received little support. The American Diabetes Association, for example, has traditionally recommend against low carbohydrate diets (less than 130 g/day[1]; while the most recent guidelines [2] admit such diets as an alternative approach to weight loss, they continue to emphasize concerns and downplay benefits. Similarly, the Diabetes and Nutrition Study Group of the European Association for the Study of Diabetes [3] reported "no justification for the recommendation of very low carbohydrate diets in persons with diabetes." We feel, however, that there is ample evidence to warrant an alternative perspective and that diets based on carbohydrate restriction should be re-evaluated in light of current understanding of the underlying biochemistry and available clinical data.
Whatever success low fat dietary approaches have had in improving diabetes is to be applauded but it is reasonable for patients to be aware of the potential benefits of an alternative approach which we present here. The key feature is that low carbohydrate diets are based on mechanism. That is, glucose directly or indirectly through insulin, is a major control element in gluconeogenesis, glycogen metabolism, lipolysis and lipogenesis. The downstream stimulus-response processes are a current research interest (see e.g. [4,5]) but, according to the view considered here, dietary fat has a generally passive role and deleterious effects of fat are almost always seen in the presence of high carbohydrate.
While low carbohydrate diets may not be appropriate for everyone, choices should be left to individual physicians and patients. Key points that bear on the assessment of benefit vs. risk of carbohydrate restriction are presented below. The discussion focuses on type 2 diabetes but many of the principles will apply to metabolic syndrome and possibly to type 1 as well[6,7].
1. Carbohydrate restriction improves glycemic control, the primary target of nutritional therapy and reduces insulin fluctuations.
2. Carbohydrate-restricted diets are at least as effective for weight loss as low-fat diets.
3. Substitution of fat for carbohydrate is generally beneficial for markers for and incidence of CVD.
4. Carbohydrate restriction improves the features of metabolic syndrome.
5. Beneficial effects of carbohydrate restriction do not require weight loss.
Carbohydrate restriction is an intuitive and rational approach to improvement of glycemic and metabolic control. Data demonstrating that weight loss and cardiovascular risk are also improved remove these barriers to the acceptance of carbohydrate restriction as a reasonable if not the preferred treatment for type 2 diabetes. Finally, carbohydrate restriction is a potentially favorable diet for improving components of the metabolic syndrome and thereby for the prevention of diabetes.
1. Carbohydrate restriction improves glycemic control, the primary target of nutritional therapy and reduces insulin fluctuations
Figure 1 shows glycemic and insulin responses in a carefully controlled inpatient comparison of 10 obese patients with type 2 diabetes[8]. Fourteen days of a low-carbohydrate diet led to a mean decrease in energy intake of approximately 1000 kcal/d, a reduction in plasma glucose levels and average hemoglobin A1c (HbA1c) from 7.3% to 6.8%. Insulin sensitivity improved by approximately 75%. No adverse effects were reported, and the carbohydrate that was removed was not replaced by substantial protein or fat.
Figure 1.
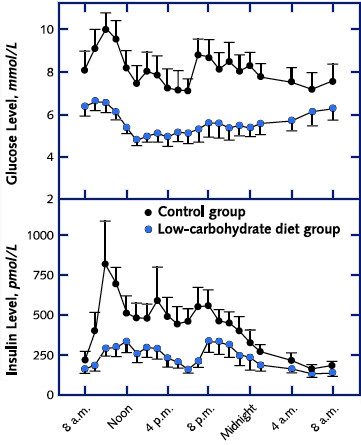
Glucose and Insulin response for patients with type 2 diabetes on low carbohydrate diet vs. control. Data (means ± SE) are for 9 patients with type 2 diabetes after seven days on their usual high-carbohydrate diet (control) and after 2 weeks) on a low-carbohydrate diet. Medication was reduced in 4 patients and discontinued in one during the low-carbohydrate diet. Figure redrawn from Boden, et al. [8].
Dashti, et al. showed dramatic and sustained reduction in blood glucose in 31 obese diabetic patients on a ketogenic diet over 56 weeks. Normal levels were reached by week 48 [9]. Similarly, Nielsen, et al.[10] reported that a 20% carbohydrate diet was superior to a 55–60% carbohydrate diet with regard to bodyweight, glycemic control and reduction in HbA1c. At follow-up, after 22 months, HbA1c remained improved. In a 16-week pilot study of Yancy, et al., 21 overweight participants with type 2 diabetes showed a mean decrease in HbA1c from 7.4% to 6.3%.
These results are not isolated. Many studies have demonstrated the benefits of carbohydrate reduction [11-16] on glycemic control. Reaven, Garg, Grundy and coworkers have shown benefits of even moderate carbohydrate reduction, from 55% to 40%[17,18].
Reduction or elimination of medication
A striking effect of carbohydrate restriction is reduction or elimination of medication. Table 1 shows results from Yancy, et al. [19] in which 17 of 21 patients with type 2 diabetes reduced or discontinued diabetes medication upon carbohydrate restriction. Similar results were found by Boden [8] and Nielsen [10,20]. Practitioners have pointed out the need to reduce medication in advance of undertaking a low carbohydrate diet [6,10,20,21] highlighting the power of carbohydrate restriction to bring about the same therapeutic effect as drugs.
Table 1.
Changes in diabetes medication of 19 overweight participants with type 2 diabetes who underwent a 16-week diet intervention trial. Patients were provided with VLCKD counseling with an initial goal of <20 g carbohydrate/day. Medication was reduced at diet initiation. Data from Yancy, et al. [62].
| Patient number | Daily Dose – Week 0 | Daily Dose – Week 16 |
| Medications discontinued (n = 7 of 19 originally on medication) | ||
| 5 | glipizide 10 mg | none |
| metformin 1000 mg | ||
| 6 | metformin 1500 mg | none |
| 7 | none | |
| 9 | metformin 1000 mg | none |
| 15 | metformin 1000 mg | none |
| 22 | metformin 1000 mg | none |
| 24 | metformin 1000 mg | none |
| Medications reduced (n = 10 of 19) | ||
| 3 | 70/30 insulin 50 units | metformin 1000 mg |
| metformin 1000 mg | ||
| 11 | metformin 2000 mg | metformin 2000 mg |
| glyburide 20 mg | ||
| 16 | metformin 2000 mg | metformin 2000 mg |
| pioglitazone 45 mg | ||
| glypizide 20 mg | ||
| 21 | metformin 1500 mg | metformin 1000 mg |
| pioglitazone 30 mg | ||
| 8 | NPH 145 units | NPH 25 units |
| metformin 1000 mg | metformin 1000 mg | |
| 13 | 70/30 insulin 70 units | 70/30 insulin 35 units |
| metformin 2550 mg | metformin 2550 mg | |
| 23 | 70/30 insulin 110 units | 70/30 insulin 80 units |
| pioglitazone 45 mg | pioglitazone 45 mg | |
| metformin 1000 mg | ||
| 25 | NPH 70 units, r 30 units | NPH 8 units |
| metformin 2000 mg | metformin 2000 mg | |
| pioglitazone 45 mg | pioglitazone 45 mg | |
| 27 | 70/30 insulin 86 units | 70/30 insulin 18 units |
| metformin 2000 mg | metformin 2000 mg | |
| 28 | NPH insulin 90 units | NPH insulin 30 units |
| lispro insulin 90 units | glypizide 20 mg | |
| glypizide 20 mg | ||
2. Carbohydrate-restricted diets are at least as effective for weight loss as low-fat diets
Low-carbohydrate diets generally perform better than low-fat diets for weight loss in normal subjects, and patients with metabolic syndrome or diabetes [22-25]. Studies by Foster, et al. [26] and Samaha, et al. [27] are often cited as examples where low carbohydrate diets are more effective at 6 months but no better than low-fat diets at 1 year. The experimental design, however, allowed re-introduction of carbohydrate in the low carbohydrate group as the study proceeded. Even if there were equal weight loss at one year, other physiologic markers, particularly TG and HDL, were greatly improved on the low-carbohydrate diet compared with the high carbohydrate diet as shown in Figure 2.
Figure 2.
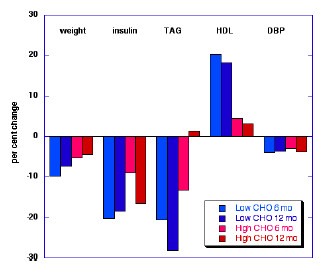
Comparison of low and high carbohydrate diets at 6 and 12 months. Results from a multi-center trial in which 63 obese men and women were randomly assigned to either diet. Data from Foster, et al. [26]. Figure from Volek & Feinman [24], used with permission. DBP, diastolic blood pressure; TAG, triglycerides.
3. Substitution of fat for carbohydrate is generally beneficial for markers for and incidence of CVD
The diet-heart hypothesis states that dietary fat, or at least saturated fat, promotes CVD. There are, however, numerous counter-examples and the popular and scientific literature has seriously challenged many of the underlying assumptions of the hypothesis [28-33]. In fact, total fat in the diet is not associated with an increase in CVD, as shown by experiments going back to Ancel Keys's Seven Country Study [34]and, most recently and dramatically, the Women's Health Initiative [35].
Lipid markers for CVD
Clinically significant elevation of LDL-cholesterol is generally considered a primary indicator of CVD risk but interpretation must be tempered by the effect of particle size: small dense LDL particles are significantly more atherogenic than large, buoyant LDL particles [36,37]. Krauss, et al. identified a genetically influenced pattern (B) in people with higher levels of the smaller particles and found a strong linear relation between carbohydrate intake and prevalence of the atherogenic pattern B phenotype. Thus, replacing dietary fat with carbohydrate tends to worsen LDL size distribution for most of the population[36,37].
Other factors, such as high triglyceride (TG) and low HDL, are independent markers of insulin resistance and CVD risk. Indeed, the triglyceride:HDL ratio has been posited to be a surrogate measure of insulin resistance [38]. This ratio is frequently exacerbated under conditions that lower LDL [24]. An increase in apolipoprotein B (apoB) may be a preferred marker since each atherogenic lipoprotein particle contains one molecule of apoB; total LDL would bias results towards lower risk [39]. There is also strong evidence that the apoB/apoA-I ratio is superior to conventional cholesterol ratios [39] as a predictor of CVD risk. Of particular importance is circulating TG because of its mechanistic link to the formation of atherogenic particles [40,41], and its responsiveness to dietary manipulation. There is probably no dietary outcome as reliable as the reduction in TG due to carbohydrate restriction[41].
The role of saturated fat
A primary goal of current recommendations is to put limits on dietary saturated fat but published results are inconsistent (see e.g. [42]). Several critical reviews have pointed up the general failure to meet the kind of unambiguous outcomes that would justify blanket condemnation of saturated fat, per se [29,30,41,43,44]. Notably, during the obesity and diabetes epidemic, the proportion of dietary saturated fat decreased. In men, the absolute amount decreased by 14%. Similarly, the WHI revealed no difference in CVD incidence for people who consumed < 10% saturated fat or those whose consumption was > 14%[35]. Dreon, et al. [44] showed that increased saturated fat lead to a decrease in small, dense LDL. Perhaps most remarkable was a study by Mozaffarian [45] which showed that greater intake of saturated fat was associated with reduced progression of coronary atherosclerosis; greater carbohydrate intake was linked to increased progression.
In our view, inconsistencies in the experimental results with dietary saturated fat arise from a failure to distinguish between replacement by unsaturated fat or by carbohydrate [3]. In the former case, there is usually improvement in CVD risk or outcome (although it is not excluded that this is due to the effect of the unsaturated fat rather than reduction in the risk from the saturated fat). Replacement of saturated fat with carbohydrate, however, is almost always deleterious [46,47]. Again, the idea that carbohydrate is a control element determining the fate of ingested lipid is overriding.
The assumption that the dietary fatty acid profile is reflected in plasma distribution is not always true, especially for saturated fatty acids which seems to be subject to much metabolic processing [42]. It was also expected that an increase in total fat might show changes in lipid pattern but Raatz, et al. showed that such differences were extremely small between a low fat and high fat diet [48]. A recent report comparing two low-CHO groups that differed in dietary SFA showed little difference in plasma levels of stearic or palmitic acid [49]. Most telling, Volek's group compared a VLCK diet (% CHO:fat 12:59) with a low-fat (LF) diet (56:24) and found that after 12 weeks, SFA in TG and cholesteryl ester were lower in the VLCK group than the LF group even thought the low carbohydrate group had a 3-fold higher intake of dietary SFA [50].
4. Carbohydrate restriction improves the features of metabolic syndrome
An important idea guiding current medical thinking is that clustering of seemingly disparate physiologic states, obesity, atherogenic dyslipidemia, hyperglycemia and hypertension, termed metabolic syndrome (MetS) suggests a common underlying cause. Inherent in this concept lies the possibility that treating one risk factor or disease state might confer benefit for risk of other diseases. A recent review showed that carbohydrate restriction improves all of these markers[24]. Indeed, metabolic syndrome might be consistently defined as those physiologic markers that respond to reduction in dietary carbohydrate. Metabolic syndrome might be seen as a generalization of the carbohydrate intolerance that characterizes frank diabetes[24].
In a prospective study testing the hypothesis linking carbohydrate restriction to MetS, the carbohydrate-restricted group showed greater improvements in weight loss and multiple markers of atherogenic dyslipidemia (increased HDL and LDL diameter and reductions in TG and apo B/apo A1 ratio) compared to a low fat arm. Unexpectedly, the carbohydrate-restricted arm, with three times greater dietary saturated fat, showed a reduction in plasma saturated fat, while plasma saturated fat in the low-fat arm remained unchanged [50].
Similarly, Petersen, et al. [51] showed that ingestion of a high carbohydrate meal led to a greater increase in de novo fatty acid synthesis and hepatic triglyceride formation in insulin-resistant men compared to a similar group of insulin-sensitive controls. Carbohydrate-induced atherogenic dyslipidemia is thus enhanced by insulin resistance.
Carbohydrate restriction will generally reduce the consumption of fructose, which makes up half the mass of common sweeteners (high-fructose corn syrup or sucrose). Fructose consumption has been implicated in the epidemics of obesity, MetS, and type 2 diabetes and is known to induce hypertension, de novo lipogenesis, hepatic insulin resistance and adiposity [52-54].
In summary, carbohydrate restriction is one of the few common interventions that targets all of the features of MetS. If such a straight-forward approach can alleviate a condition for which there is no known effective drug, its potential should be vigorously explored.
5. Beneficial effects of carbohydrate restriction do not require weight loss
Obesity is commonly considered a cause of insulin resistance. Obesity, however, does not occur spontaneously. Obesity is a response. The effects of obesity that lead to insulin resistance in peripheral tissues, largely increased fatty acids, are downstream from the primary impact of diet. This argues for an emphasis of treatment on glycemic control and improved hepatic metabolism rather than weight loss. A simpler alternative hypothesis considers that insulin resistance represents a down-regulation of hormonal response as a result of persistent high levels of insulin, a feature common to other hormonal systems [55]. In this view, diabetes, obesity and the components of MetS are parallel effects of hyperinsulinemia and/or hyperglycemia.
The finding that lipid improvements seen in carbohydrate-restricted diets persist even after no further weight loss (Figure 2) suggests that the benefit of carbohydrate restriction is independent of weight loss. Two additional lines of evidence support this idea:
1) In experiments in which body mass is kept constant in normal-weight men[56]or patients with type 2 diabetes[11,13], a very low carbohydrate diet resulted in dramatic improvements in triglycerides and HDL cholesterol with minimal change in body mass.
2) Experiments in which change in macronutrients and weight loss are separated in time show that eucaloric carbohydrate reduction leads to greater improvement in atherogenic lipid markers (TG, HDL, apoB/apoA1 and mean LDL particle size) even in the presence of higher saturated fat[57,58]. A low fat diet, however, required weight loss to achieve effective improvement in the lipid profile (Figure 3). Notably, the sum of the two effects showed that eucaloric carbohydrate restriction plus weight loss was more effective than eucaloric low fat plus weight loss.
Figure 3.
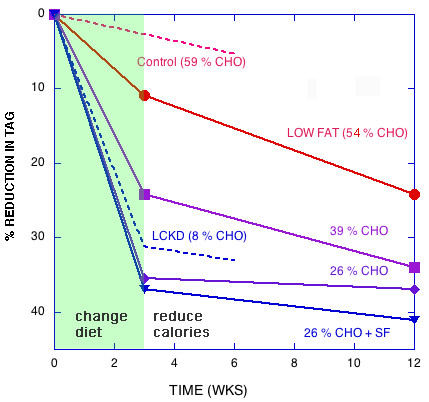
Effect of dietary interventions on reduction in triglycerides. Eucaloric diets of indicated carbohydrate content were begun at time 0. At week 3, a 1000 kcal reduction in energy was implemented and at week 9, dieters were put on maintenance diet. Combined effect of calorie reduction and maintenance are reported at week 12. Solid Lines: data from Krauss, et al. [58] were converted from reported log values in their Table 2 and per cent of baseline was calculated. Dashed line: data from Sharman, et al [56]: A eucaloric ketogenic diet was instituted for six weeks (no weight loss phase). Points were recorded at week 3 and 6. Figure modified from Feinman & Volek [57]. Similar results were found for HDL, apoB/apoA1 and other markers of CVD [57, 58]
Practical considerations and recommendations
Definitions and recommendations
Response to carbohydrate restriction shows both continuous, graded outcomes [17,18] as well as a threshold effects. LDL particle size, e.g. appears to depend linearly on the level of dietary carbohydrate[36,37]. On the other hand, many studies show maximum benefit for very low carbohydrate intake; the early phases of popular low carbohydrate diets target such very low levels [6,15,21,59,60]. The principle rests on the concept of a catalytic or threshold effect for insulin in shifting the body from an anabolic state to fat oxidation. The tipping point is empirically taken as the onset of ketonuria, also used as an indicator of compliance with a very low carbohydrate ketogenic diet (VLCKD). The threshold carbohydrate reduction for ketonuria varies among individuals, but a rough estimate is 50 g of carbohydrate per day or, approximately 10% of energy on a nominal 2000 kcal diet, (a target of 30 g/d is common in the early phases of popular VLCKD diets)[15,21,60].
We suggest the following definitions:
The ADA designates low carbohydrate diets as less than 130 g/d or 26% of a nominal 2000 kcal diet and we consider this a reasonable cutoff for the definition of a low-carbohydrate diet. Carbohydrate consumption before the epidemic of obesity averaged 43%, and we suggest 26% to 45% as the range for moderate-carbohydrate diets. The intake of less than 30 g/d, as noted above should be referred to as a very low carbohydrate ketogenic diet (VLCKD). The term Ketogenic Diet should be reserved for the therapeutic approach to epilepsy. These diets do not independently specify the level of carbohydrate, but rather the sum of carbohydrate and protein.
In practice, many low carbohydrate dieters do not add additional fat. First shown by LaRosa, [61] it has now been observed by many other investigators. [8,62,63] A reduced carbohydrate diet may show significant per cent increase in fat, but there may be no change in the absolute amount consumed. Not everybody on a low carbohydrate diet follows this pattern, but a recommendation based on this behavior would seem more appropriate than unqualified rejection of low-carbohydrate diets.
While some proponents of carbohydrate restriction for the management of diabetes favor sustained adherence to very low levels of carbohydrate intake [6], all options may be considered and therapeutic choices can be determined by individual physicians and their patients
The term low-carbohydrate diet is frequently taken as synonymous with the popular Atkins diet[60] which remains highly controversial. Carbohydrate control, however, has many implementations and the severity of the epidemic of diabetes makes it appropriate to go beyond historical controversy and analyze dietary interventions as they are actually implemented.
There is reluctance to make recommendations for low carbohydrate diets on the grounds that people will not follow them but compliance and efficacy of dietary recommendations are separate phenomena. In fact, all recommendations are specifically intended to be different from average consumption[1] and it is sensibly the purpose of health agencies to encourage conformance to the best therapies.
It is time to re-appraise the role of carbohydrate restriction. Although pessimism exists in the medical community on the efficacy of any diet in the treatment of diabetes 2 and MetS, the success of carbohydrate restriction for many practitioners and individual patients[64] mandates that we should determine how this approach can be consistently and effectively employed.
Finally, while no systematic study of clinical practice has been done, anecdotal evidence suggests that carbohydrate restriction is a common clinical recommendation for diabetes. We believe that there is a need to codify these recommendations in light of current evidence.
Basic biochemistry, clinical experience and an evolving understanding of metabolic syndrome support the need for evaluation of the efficacy and safety of carbohydrate-restricted diets for the treatment of type 2 diabetes. The fact that carbohydrate restriction improves markers of cardiovascular health, even in the absence of weight loss, sensibly removes historical objections to the dangers of this approach. A critical re-appraisal could form the basis for an alternative for those patients for whom current recommendations are not successful.
Competing interests
MCV has been a consultant for Atkins Nutritionals, Inc. All other authors declare that they have nothing to declare.
Authors' contributions
RDF wrote the first version of this article based on published material and correspondence with the other authors who modified and approved the final version.
Abbreviations
apo B, apolipoprotein B; apo A1, apolipoprotein A1: CVD, cardiovascular disease; HbA1c, hemoglobin A1c; HDL, high density lipoprotein; LDL, low density lipoprotein; MetS, metabolic syndrome; SFA, saturated fatty acids; TG, triglycerides (triacylglycerol); VLCKD (VLCK diet), very low carbohydrate ketogenic diet.
Contributor Information
Anthony Accurso, Email: anthony.accurso@downstate.edu.
Richard K Bernstein, Email: md@diabetesscientist.com.
Annika Dahlqvist, Email: annika.dahlqvist@telia.com.
Boris Draznin, Email: Boris.Draznin@va.gov.
Richard D Feinman, Email: rfeinman@downstate.edu.
Eugene J Fine, Email: efine@downstate.edu.
Amy Gleed, Email: amy.gleed@downstate.edu.
David B Jacobs, Email: david.jacobs@downstate.edu.
Gabriel Larson, Email: gabriel.larson@downstate.edu.
Robert H Lustig, Email: rlustig@peds.ucsf.edu.
Anssi H Manninen, Email: anssi.manninen@gmail.com.
Samy I McFarlane, Email: sammy.mcfarlane@downstate.ed.
Katharine Morrison, Email: morrisons@ballochmyle.wanadoo.co.uk.
Jørgen Vesti Nielsen, Email: jvesti_nielsen@hotmail.com.
Uffe Ravnskov, Email: ravnskov@tele2.se.
Karl S Roth, Email: KarlRoth@creighton.edu.
Ricardo Silvestre, Email: blue_ricardo@yahoo.com.
James R Sowers, Email: sowersj@health.missouri.edu.
Ralf Sundberg, Email: ralfsundberg@telia.com.
Jeff S Volek, Email: Jeff.volek@uconn.edu.
Eric C Westman, Email: westm001@mc.duke.edu.
Richard J Wood, Email: rwood@spfldcol.edu.
Jay Wortman, Email: jwortman@interchange.ubc.ca.
Mary C Vernon, Email: mvernonmd@yahoo.com.
References
- American Diabetes Association Nutrition Recommendations and Interventions for Diabetes-2007. Diabetes Care. 2007;30:S48–S65. doi: 10.2337/dc07-S048. [DOI] [PubMed] [Google Scholar]
- American Diabetes Association Nutrition Recommendations and Interventions for Diabetes-2008. Diabetes Care. 2008;31:S61–S78. doi: 10.2337/dc08-S061. [DOI] [PubMed] [Google Scholar]
- Mann JI, De Leeuw I, Hermansen K, Karamanos B, Karlstrom B, Katsilambros N, Riccardi G, Rivellese AA, Rizkalla S, Slama G, et al. Evidence-based nutritional approaches to the treatment and prevention of diabetes mellitus. Nutr Metab Cardiovasc Dis. 2004;14:373–394. doi: 10.1016/S0939-4753(04)80028-0. [DOI] [PubMed] [Google Scholar]
- Draznin B. Molecular mechanisms of insulin resistance: serine phosphorylation of insulin receptor substrate-1 and increased expression of p85alpha: the two sides of a coin. Diabetes. 2006;55:2392–2397. doi: 10.2337/db06-0391. [DOI] [PubMed] [Google Scholar]
- Reusch JE, Draznin BB. Atherosclerosis in diabetes and insulin resistance. Diabetes Obes Metab. 2007;9:455–463. doi: 10.1111/j.1463-1326.2006.00620.x. [DOI] [PubMed] [Google Scholar]
- Bernstein R. Dr Bernstein's Diabetes Solution: The Complete Guide to Achieving Normal Blood Sugars. Little, Brown and Company; 2007. [Google Scholar]
- Nielsen JV, Jönsson E, Ivarsson A. A low carbohydrate diet in type 1 diabetes: clinical experience – a brief report. Ups J Med Sci. 2005;110:267–273. doi: 10.3109/2000-1967-074. [DOI] [PubMed] [Google Scholar]
- Boden G, Sargrad K, Homko C, Mozzoli M, Stein TP. Effect of a low-carbohydrate diet on appetite, blood glucose levels, and insulin resistance in obese patients with type 2 diabetes. Ann Intern Med. 2005;142:403–411. doi: 10.7326/0003-4819-142-6-200503150-00006. [DOI] [PubMed] [Google Scholar]
- Dashti HM, Mathew TC, Khadada M, Al-Mousawi M, Talib H, Asfar SK, Behbahani AI, Al-Zaid NS. Beneficial effects of ketogenic diet in obese diabetic subjects. Mol Cell Biochem. 2007 doi: 10.1007/s11010-007-9448-z. [DOI] [PubMed] [Google Scholar]
- Nielsen JV, Jönsson EA. Low-carbohydrate diet in type 2 diabetes. Stable improvement of bodyweight and glycaemic control during 22 months follow-up. Nutr Metab (Lond) 2006;3:22. doi: 10.1186/1743-7075-3-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allick G, Bisschop PH, Ackermans MT, Endert E, Meijer AJ, Kuipers F, Sauerwein HP, Romijn JA. A low-carbohydrate/high-fat diet improves glucoregulation in type 2 diabetes mellitus by reducing postabsorptive glycogenolysis. J Clin Endocrinol Metab. 2004;89:6193–6197. doi: 10.1210/jc.2004-1041. [DOI] [PubMed] [Google Scholar]
- Daly ME, Paisey R, Paisey R, Millward BA, Eccles C, Williams K, Hammersley S, MacLeod KM, Gale TJ. Short-term effects of severe dietary carbohydrate-restriction advice in Type 2 diabetes – a randomized controlled trial. Diabet Med. 2006;23:15–20. doi: 10.1111/j.1464-5491.2005.01760.x. [DOI] [PubMed] [Google Scholar]
- Gannon MC, Nuttall FQ. Control of blood glucose in type 2 diabetes without weight loss by modification of diet composition. Nutr Metab (Lond) 2006;3:16. doi: 10.1186/1743-7075-3-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vernon M, Mavropoulos J, Transue M, Yancy W, Jr, Westman E. Clinical Experience of a Carbohydrate-Restricted Diet: Effect on Diabetes Mellitus. Metabolic Syndrome and Related Disorders. 2003;1:233–237. doi: 10.1089/154041903322716714. [DOI] [PubMed] [Google Scholar]
- Vernon MC, Eberstein JA. Atkins Diabetes Revolution The Groundbreaking Approach to Preventing and Controlling Type 2 Diabetes. New York: William Morrow; 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yancy WS, Jr, Vernon MC, Westman EC. A Pilot trial of a Low-Carbohydrate, Ketogenic Diet in Patients with Type 2 Diabetes. Metabolic Syndrome and Related Disorders. 2003;1:239–243. doi: 10.1089/154041903322716723. [DOI] [PubMed] [Google Scholar]
- Reaven GM. Effect of dietary carbohydrate on the metabolism of patients with non-insulin dependent diabetes mellitus. Nutr Rev. 1986;44:65–73. doi: 10.1111/j.1753-4887.1986.tb07589.x. [DOI] [PubMed] [Google Scholar]
- Garg A, Bantle JP, Henry RR, Coulston AM, Griver KA, Raatz SK, Brinkley L, Chen YD, Grundy SM, Huet BA, et al. Effects of varying carbohydrate content of diet in patients with non-insulin-dependent diabetes mellitus. JAMA. 1994;271:1421–1428. doi: 10.1001/jama.271.18.1421. [DOI] [PubMed] [Google Scholar]
- Westman EC, Yancy WS, Jr, Haub MD, Volek JS. Insulin Resistance from a Low-Carbohydrate, High Fat Diet Perspective. Metabolic Syndrome and Related Disorders. 2005;3:3–7. doi: 10.1089/met.2005.3.14. [DOI] [PubMed] [Google Scholar]
- Nielsen JV, Jonsson E, Nilsson AK. Lasting improvement of hyperglycaemia and bodyweight: low-carbohydrate diet in type 2 diabetes – a brief report. Ups J Med Sci. 2005;110:69–73. doi: 10.3109/2000-1967-182. [DOI] [PubMed] [Google Scholar]
- Eades MR, Eades MD. Protein Power. New York: Bantam Books; 1996. [Google Scholar]
- Feinman RD, Fine EJ. Thermodynamics and Metabolic Advantage of Weight Loss Diets. Metabolic Syndrome and Related Disorders. 2003;1:209–219. doi: 10.1089/154041903322716688. [DOI] [PubMed] [Google Scholar]
- Krieger JW, Sitren HS, Daniels MJ, Langkamp-Henken B. Effects of variation in protein and carbohydrate intake on body mass and composition during energy restriction: a meta-regression 1. Am J Clin Nutr. 2006;83:260–274. doi: 10.1093/ajcn/83.2.260. [DOI] [PubMed] [Google Scholar]
- Volek JS, Feinman RD. Carbohydrate restriction improves the features of Metabolic Syndrome. Metabolic Syndrome may be defined by the response to carbohydrate restriction. Nutr Metab (Lond) 2005;2:31. doi: 10.1186/1743-7075-2-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westman EC, Feinman RD, Mavropoulos JC, Vernon MC, Volek JS, Wortman JA, Yancy WS, Phinney SD. Low-carbohydrate nutrition and metabolism. Am J Clin Nutr. 2007;86:276–284. doi: 10.1093/ajcn/86.2.276. [DOI] [PubMed] [Google Scholar]
- Foster GD, Wyatt HR, Hill JO, McGuckin BG, Brill C, Mohammed BS, Szapary PO, Rader DJ, Edman JS, Klein S. A randomized trial of a low-carbohydrate diet for obesity. N Engl J Med. 2003;348:2082–2090. doi: 10.1056/NEJMoa022207. [DOI] [PubMed] [Google Scholar]
- Samaha FF, Iqbal N, Seshadri P, Chicano KL, Daily DA, McGrory J, Williams T, Williams M, Gracely EJ, Stern L. A low-carbohydrate as compared with a low-fat diet in severe obesity. N Engl J Med. 2003;348:2074–2081. doi: 10.1056/NEJMoa022637. [DOI] [PubMed] [Google Scholar]
- Colpo A. The Great Cholesterol Con. Lulu Press; 2006. [Google Scholar]
- German JB, Dillard CJ. Saturated fats: what dietary intake? Am J Clin Nutr. 2004;80:550–559. doi: 10.1093/ajcn/80.3.550. [DOI] [PubMed] [Google Scholar]
- Karam J, Nessim F, McFarlane S, Feinman R. Carbohydrate Restriction and Cardiovascular Risk. Current Cardiovascular Risk Reports. 2008;2:88–94. doi: 10.1007/s12170-008-0018-z. [DOI] [Google Scholar]
- Ravnskov U. The Cholesterol Myths: Exposing the Fallacy that Cholesterol and Saturated Fat Cause Heart Disease. Washington, DC: New Trends Publishing, Inc; 2000. [Google Scholar]
- Taubes G. Good Calories, Bad Calories. New York: Alfred A. Knopf; 2007. [Google Scholar]
- Volek JS, Forsythe CE. The case for not restricting saturated fat on a low carbohydrate diet. Nutr Metab (Lond) 2005;2:21. doi: 10.1186/1743-7075-2-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keys A. Coronary heart disease in seven countries. 1970. pp. 1–211. [DOI] [PubMed]
- Howard BV, Van Horn L, Hsia J, Manson JE, Stefanick ML, Wassertheil-Smoller S, Kuller LH, LaCroix AZ, Langer RD, Lasser NL, et al. Low-fat dietary pattern and risk of cardiovascular disease: the Women's Health Initiative Randomized Controlled Dietary Modification Trial. JAMA. 2006;295:655–666. doi: 10.1001/jama.295.6.655. [DOI] [PubMed] [Google Scholar]
- Dreon DM, Fernstrom HA, Miller B, Krauss RM. Low-density lipoprotein subclass patterns and lipoprotein response to a reduced-fat diet in men. FASEB J. 1994;8:121–126. [PubMed] [Google Scholar]
- Krauss RM. Atherogenic lipoprotein phenotype and diet-gene interactions. J Nutr. 2001;131:340S–343S. doi: 10.1093/jn/131.2.340S. [DOI] [PubMed] [Google Scholar]
- McLaughlin T, Reaven G, Abbasi F, Lamendola C, Saad M, Waters D, Simon J, Krauss RM. Is there a simple way to identify insulin-resistant individuals at increased risk of cardiovascular disease? Am J Cardiol. 2005;96:399–404. doi: 10.1016/j.amjcard.2005.03.085. [DOI] [PubMed] [Google Scholar]
- Barter PJ, Ballantyne CM, Carmena R, Castro Cabezas M, Chapman MJ, Couture P, de Graaf J, Durrington PN, Faergeman O, Frohlich J, et al. Apo B versus cholesterol in estimating cardiovascular risk and in guiding therapy: report of the thirty-person/ten-country panel. J Intern Med. 2006;259:247–258. doi: 10.1111/j.1365-2796.2006.01616.x. [DOI] [PubMed] [Google Scholar]
- Ginsberg HN, Zhang YL, Hernandez-Ono A. Regulation of plasma triglycerides in insulin resistance and diabetes. Arch Med Res. 2005;36:232–240. doi: 10.1016/j.arcmed.2005.01.005. [DOI] [PubMed] [Google Scholar]
- Volek JS, Sharman MJ, Forsythe CE. Modification of lipoproteins by very low-carbohydrate diets. J Nutr. 2005;135:1339–1342. doi: 10.1093/jn/135.6.1339. [DOI] [PubMed] [Google Scholar]
- Vessby B. Dietary fat, fatty acid composition in plasma and the metabolic syndrome. Curr Opin Lipidol. 2003;14:15–19. doi: 10.1097/00041433-200302000-00004. [DOI] [PubMed] [Google Scholar]
- Ravnskov U. The questionable role of saturated and polyunsaturated fatty acids in cardiovascular disease. J Clin Epidemiol. 1998;51:443–460. doi: 10.1016/S0895-4356(98)00018-3. [DOI] [PubMed] [Google Scholar]
- Dreon DM, Fernstrom HA, Campos H, Blanche P, Williams PT, Krauss RM. Change in dietary saturated fat intake is correlated with change in mass of large low-density-lipoprotein particles in men. Am J Clin Nutr. 1998;67:828–836. doi: 10.1093/ajcn/67.5.828. [DOI] [PubMed] [Google Scholar]
- Mozaffarian D, Rimm EB, Herrington DM. Dietary fats, carbohydrate, and progression of coronary atherosclerosis in postmenopausal women. Am J Clin Nutr. 2004;80:1175–1184. doi: 10.1093/ajcn/80.5.1175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu FB, Stampfer MJ, Manson JE, Rimm E, Colditz GA, Rosner BA, Hennekens CH, Willett WC. Dietary fat intake and the risk of coronary heart disease in women. N Engl J Med. 1997;337:1491–1499. doi: 10.1056/NEJM199711203372102. [DOI] [PubMed] [Google Scholar]
- Mensink RP, Zock PL, Kester AD, Katan MB. Effects of dietary fatty acids and carbohydrates on the ratio of serum total to HDL cholesterol and on serum lipids and apolipoproteins: a meta-analysis of 60 controlled trials. Am J Clin Nutr. 2003;77:1146–1155. doi: 10.1093/ajcn/77.5.1146. [DOI] [PubMed] [Google Scholar]
- Raatz SK, Bibus D, Thomas W, Kris-Etherton P. Total fat intake modifies plasma fatty acid composition in humans. J Nutr. 2001;131:231–234. doi: 10.1093/jn/131.2.231. [DOI] [PubMed] [Google Scholar]
- Cassady B, Charboneau N, Brys E, Crouse K, Beitz D, Wilson T. Effects of low carbohydrate diets high in red meats or poultry, fish and shellfish on plasma lipids and weight loss. Nutr Metab (Lond) 2007;4 doi: 10.1186/1743-7075-4-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forsythe CE, Phinney SD, Fernandez ML, Quann EE, Wood RJ, Bibus DM, Kraemer WJ, Feinman RD, Volek JS. Comparison of low fat and low carbohydrate diets on circulating Fatty Acid composition and markers of inflammation. Lipids. 2008;43:65–77. doi: 10.1007/s11745-007-3132-7. [DOI] [PubMed] [Google Scholar]
- Petersen KF, Dufour S, Savage DB, Bilz S, Solomon G, Yonemitsu S, Cline GW, Befroy D, Zemany L, Kahn BB, et al. Inaugural Article: The role of skeletal muscle insulin resistance in the pathogenesis of the metabolic syndrome. Proc Natl Acad Sci USA. 2007;104:12587–12594. doi: 10.1073/pnas.0705408104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le KA, Tappy L. Metabolic effects of fructose. Curr Opin Nutr Metab Care. 2006;9:469–475. doi: 10.1097/01.mco.0000232910.61612.4d. [DOI] [PubMed] [Google Scholar]
- Rutledge AC, Adeli K. Fructose and the metabolic syndrome: pathophysiology and molecular mechanisms. Nutr Rev. 2007;65:S13–S23. doi: 10.1301/nr.2007.jun.S13-S23. [DOI] [PubMed] [Google Scholar]
- Zammit VA. Insulin stimulation of hepatic triacylglycerol secretion in the insulin-replete state: implications for the etiology of peripheral insulin resistance. Ann N Y Acad Sci. 2002;967:52–65. doi: 10.1111/j.1749-6632.2002.tb04263.x. [DOI] [PubMed] [Google Scholar]
- Lustig RH. Childhood obesity: behavioral aberration or biochemical drive? Reinterpreting the First Law of Thermodynamics. Nature Clin Pract Endo Metab. 2006;2:447–458. doi: 10.1038/ncpendmet0220. [DOI] [PubMed] [Google Scholar]
- Sharman MJ, Kraemer WJ, Love DM, Avery NG, Gomez AL, Scheett TP, Volek JS. A ketogenic diet favorably affects serum biomarkers for cardiovascular disease in normal-weight men. J Nutr. 2002;132:1879–1885. doi: 10.1093/jn/132.7.1879. [DOI] [PubMed] [Google Scholar]
- Feinman RD, Volek JS. Low carbohydrate diets improve atherogenic dyslipidemia even in the absence of weight loss. Nutr Metab (Lond) 2006;3:24. doi: 10.1186/1743-7075-3-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krauss RM, Blanche PJ, Rawlings RS, Fernstrom HS, Williams PT. Separate effects of reduced carbohydrate intake and weight loss on atherogenic dyslipidemia. Am J Clin Nutr. 2006;83:1025–1031. doi: 10.1093/ajcn/83.5.1025. [DOI] [PubMed] [Google Scholar]
- Agatston A. The South Beach Diet. New York: Random House; 2003. [Google Scholar]
- Atkins RC. Dr Atkins' New Diet Revolution. New York: Avon Books; 2002. [Google Scholar]
- Larosa JC, Fry AG, Muesing R, Rosing DR. Effects of high-protein, low-carbohydrate dieting on plasma lipoproteins and body weight. J Am Diet Assoc. 1980;77:264–270. [PubMed] [Google Scholar]
- Yancy WS, Jr, Foy M, Chalecki AM, Vernon MC, Westman EC. A low-carbohydrate, ketogenic diet to treat type 2 diabetes. Nutr Metab (Lond) 2005;2:34. doi: 10.1186/1743-7075-2-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Volek JS, Sharman MJ, Gomez AL, Judelson DA, Rubin MR, Watson G, Sokmen B, Silvestre R, French DN, Kraemer WJ. Comparison of energy-restricted very low-carbohydrate and low-fat diets on weight loss and body composition in overweight men and women. Nutr Metab (Lond) 2004;1:13. doi: 10.1186/1743-7075-1-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinman RD, Vernon MC, Westman EC. Low carbohydrate diets in family practice: what can we learn from an internet-based support group. Nutr J. 2006;5:26. doi: 10.1186/1475-2891-5-26. [DOI] [PMC free article] [PubMed] [Google Scholar]


