Abstract
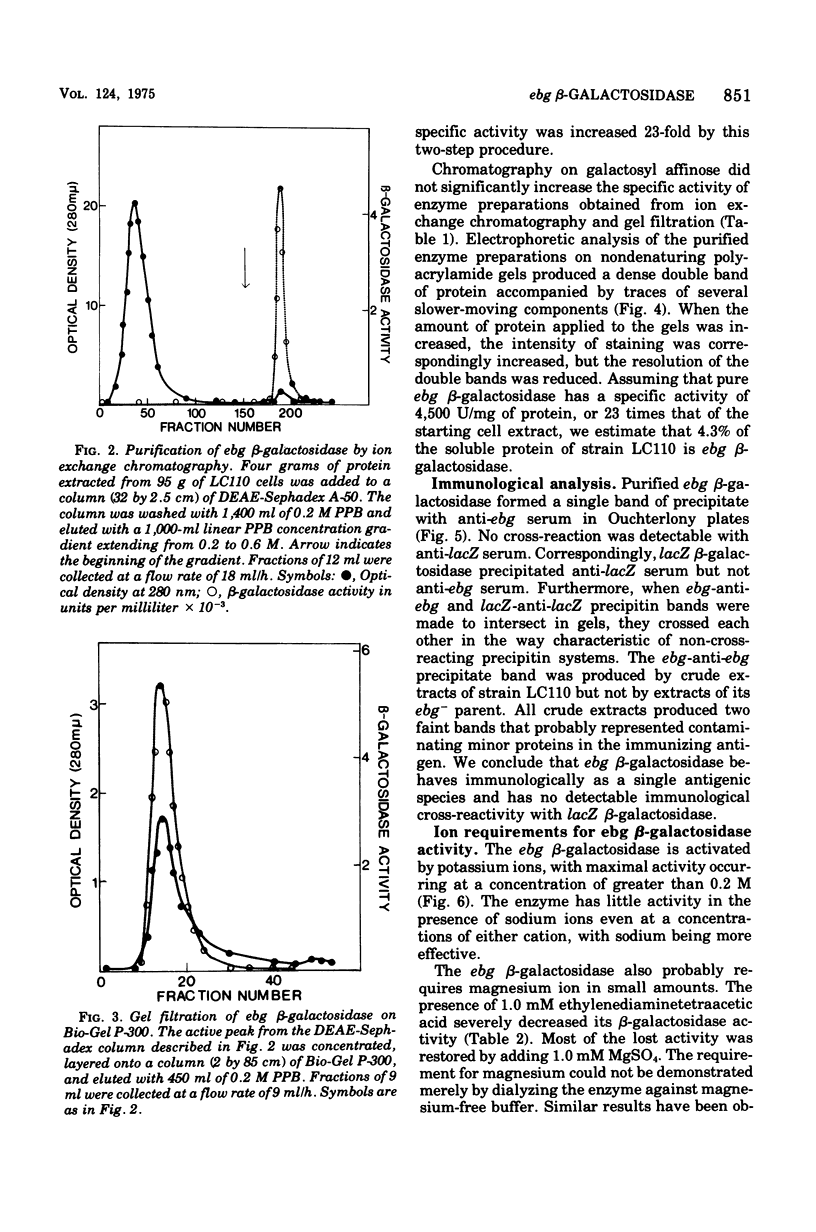
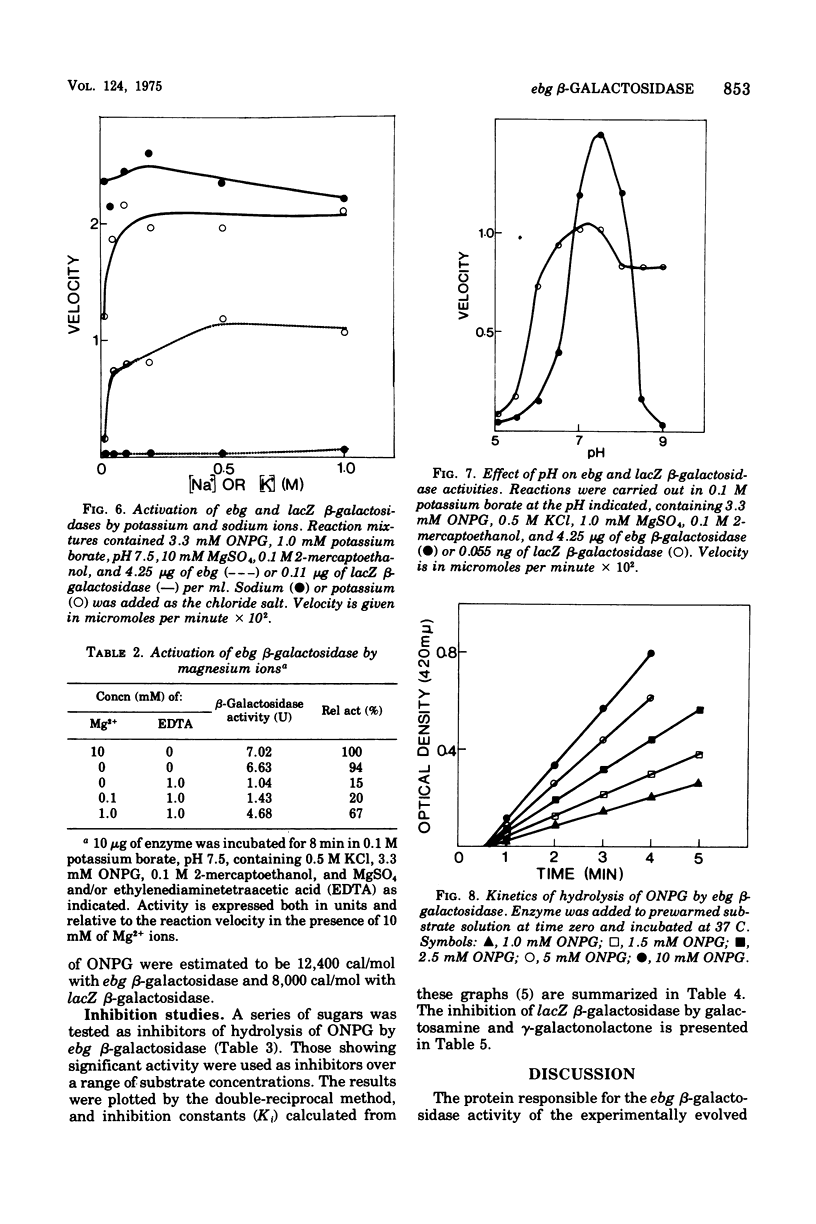
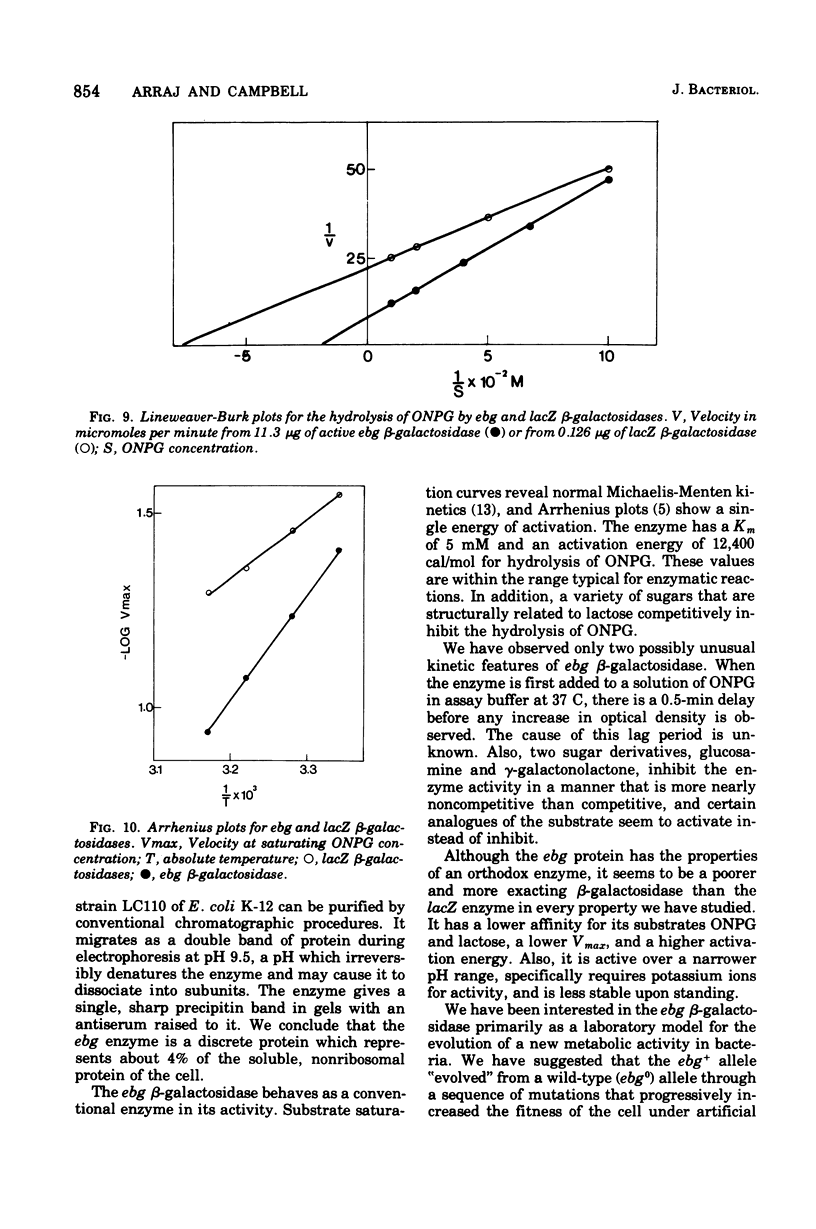
The ebg beta-galactosidase of Escherichia coli K-12 strain LC110 has been purified and characterized. Strain LC110 is a Lac+ revertant of a mutant with a deletion of the lacZ beta-galactosidase gene. Its new ebg beta-galactosidase activity was shown to be due to a discrete protein, immunologically unrelated to lacZ beta-galactosidase. Its kinetics of action conformed to those of a simple conventional enzyme. With o-nitrophenyl-beta-D-galactoside as substrate, the Vmax was 11,200 nmol/min per mg of enzyme, the Km was 5 mM, and the activation energy was 12,400 cal/mol. Corresponding values for lacZ beta-galactosidase of wild-type E. coli K-12 were 350,000 nmol/min per mg of enzyme, 1.3 mM, and 8,000 cal/mol. A series of sugars has been examined as competitive inhibitors of ebg beta-galactosidase. Kinetic analyses suggest that ebg beta-galactosidase has a particularly high affinity for galactosamine and gamma-galactonolactone, binds galatose more tightly than lactose, and shows a general preference for monosaccharides rather than beta-galactosides. We conclude that the ebg beta-galactosidase may have arisen by modification of a gene involved with the metabolism of a monosaccharide, possibly a 2-amino sugar.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- COHN M. Contributions of studies on the beta-galactosidase of Escherichia coli to our understanding of enzyme synthesis. Bacteriol Rev. 1957 Sep;21(3):140–168. doi: 10.1128/br.21.3.140-168.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CONCHIE J., LEVVY G. A. Inhibition of glycosidases by aldonolactones of corresponding configuration. Biochem J. 1957 Feb;65(2):389–395. doi: 10.1042/bj0650389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell J. H., Lengyel J. A., Langridge J. Evolution of a second gene for beta-galactosidase in Escherichia coli. Proc Natl Acad Sci U S A. 1973 Jun;70(6):1841–1845. doi: 10.1073/pnas.70.6.1841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cuatrecasas P. Protein purification by affinity chromatography. Derivatizations of agarose and polyacrylamide beads. J Biol Chem. 1970 Jun;245(12):3059–3065. [PubMed] [Google Scholar]
- Hall B. G., Hartl D. L. Regulation of newly evolved enzymes. I. Selection of a novel lactase regulated by lactose in Escherichia coli. Genetics. 1974 Mar;76(3):391–400. doi: 10.1093/genetics/76.3.391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartl D. L., Hall B. G. Second naturally occurring beta-galactosidase in E. coli. Nature. 1974 Mar 8;248(5444):152–153. doi: 10.1038/248152a0. [DOI] [PubMed] [Google Scholar]
- Hill J. A., Huber R. E. Effects of various concentrations of Na + and Mg 2+ on the activity of -galactosidase. Biochim Biophys Acta. 1971 Dec 15;250(3):530–537. doi: 10.1016/0005-2744(71)90253-1. [DOI] [PubMed] [Google Scholar]
- JACOB F., MONOD J. Genetic regulatory mechanisms in the synthesis of proteins. J Mol Biol. 1961 Jun;3:318–356. doi: 10.1016/s0022-2836(61)80072-7. [DOI] [PubMed] [Google Scholar]
- LEVVY G. A., McALLAN A., HAY A. J. Inhibition of glycosidases by aldonolactones of corresponding configuration. 3. Inhibitors of beta-D-galactosidase. Biochem J. 1962 Feb;82:225–232. doi: 10.1042/bj0820225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- REITHEL F. J., KIM J. C. Studies on the beta-galactosidase isolated from Escherichia coli ML 308. 1. The effect of some ions on enzymic activity. Arch Biochem Biophys. 1960 Oct;90:271–277. doi: 10.1016/0003-9861(60)90579-8. [DOI] [PubMed] [Google Scholar]
- Steers E., Jr, Cuatrecasas P., Pollard H. B. The purification of beta-galactosidase from Escherichia coli by affinity chromatography. J Biol Chem. 1971 Jan 10;246(1):196–200. [PubMed] [Google Scholar]
- Warren R. A. Lactose-utilizing mutants of lac deletion strains of Escherichia coli. Can J Microbiol. 1972 Sep;18(9):1439–1444. doi: 10.1139/m72-221. [DOI] [PubMed] [Google Scholar]





