Abstract
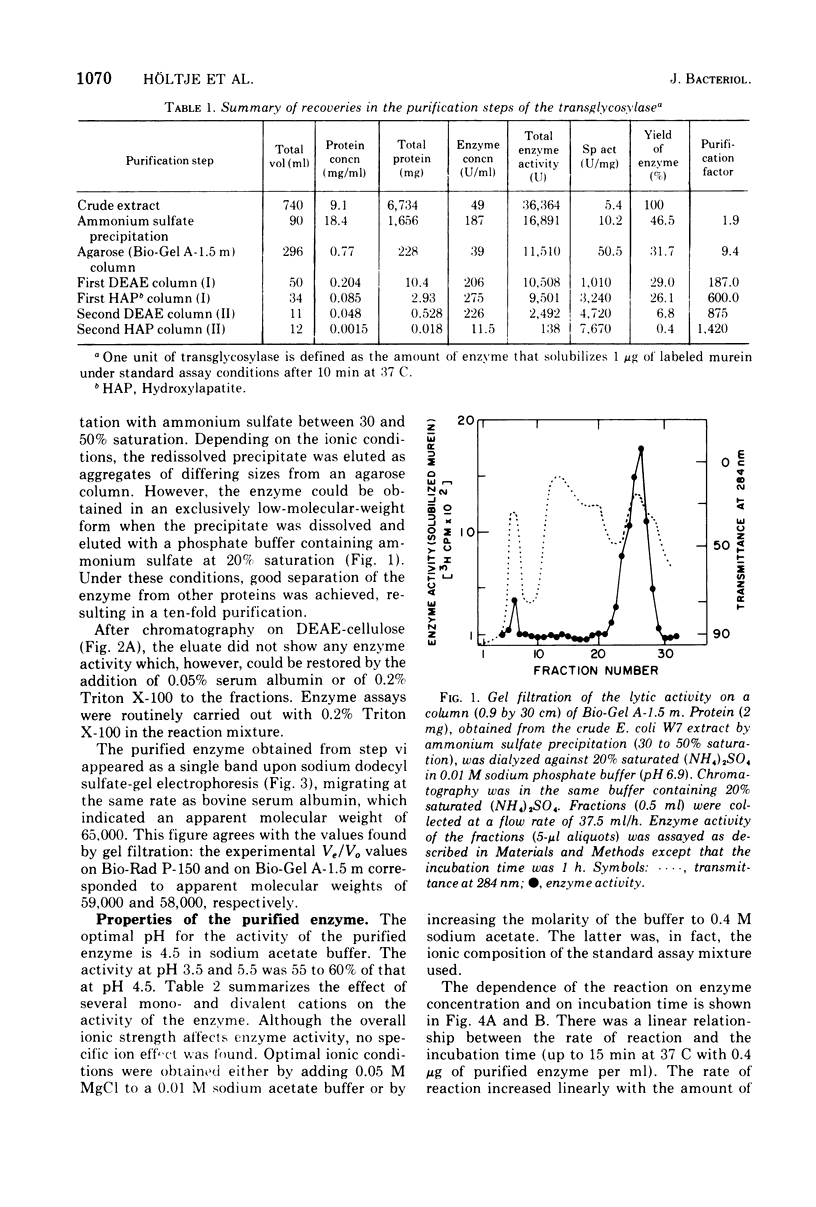
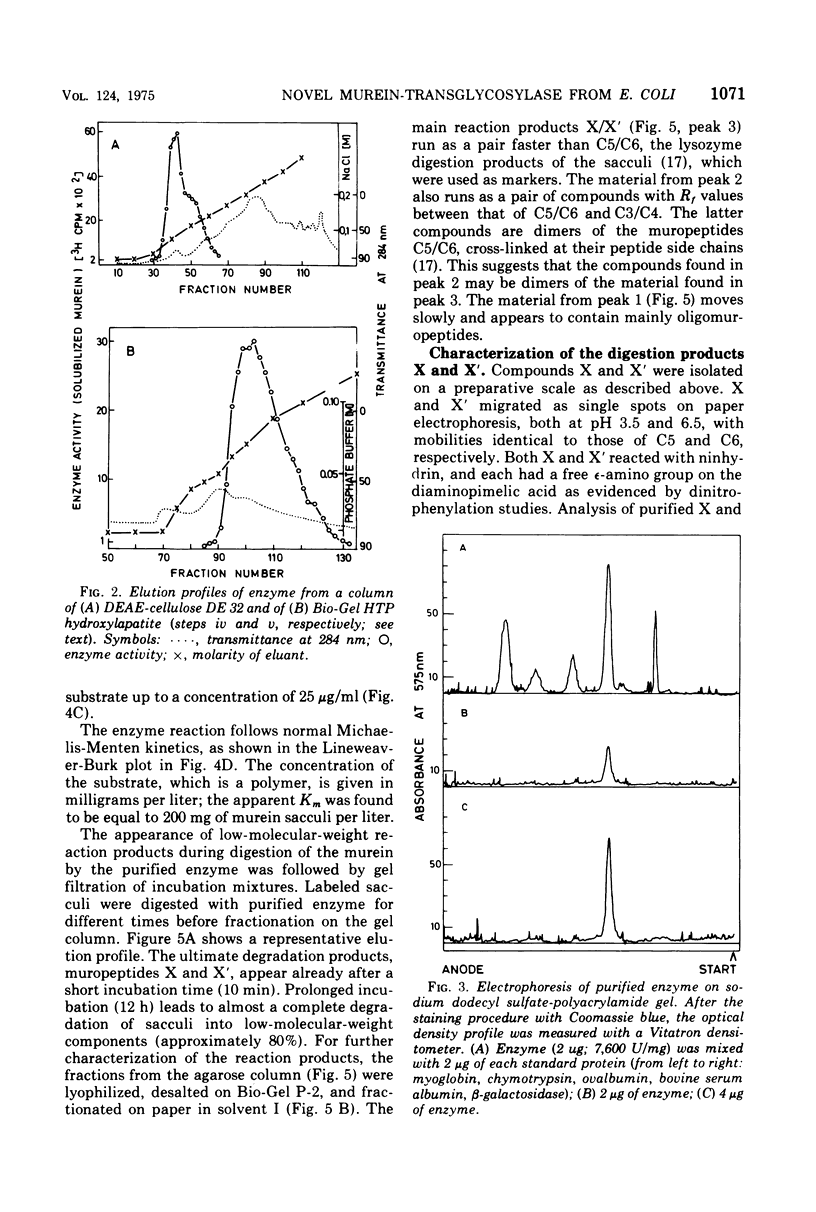
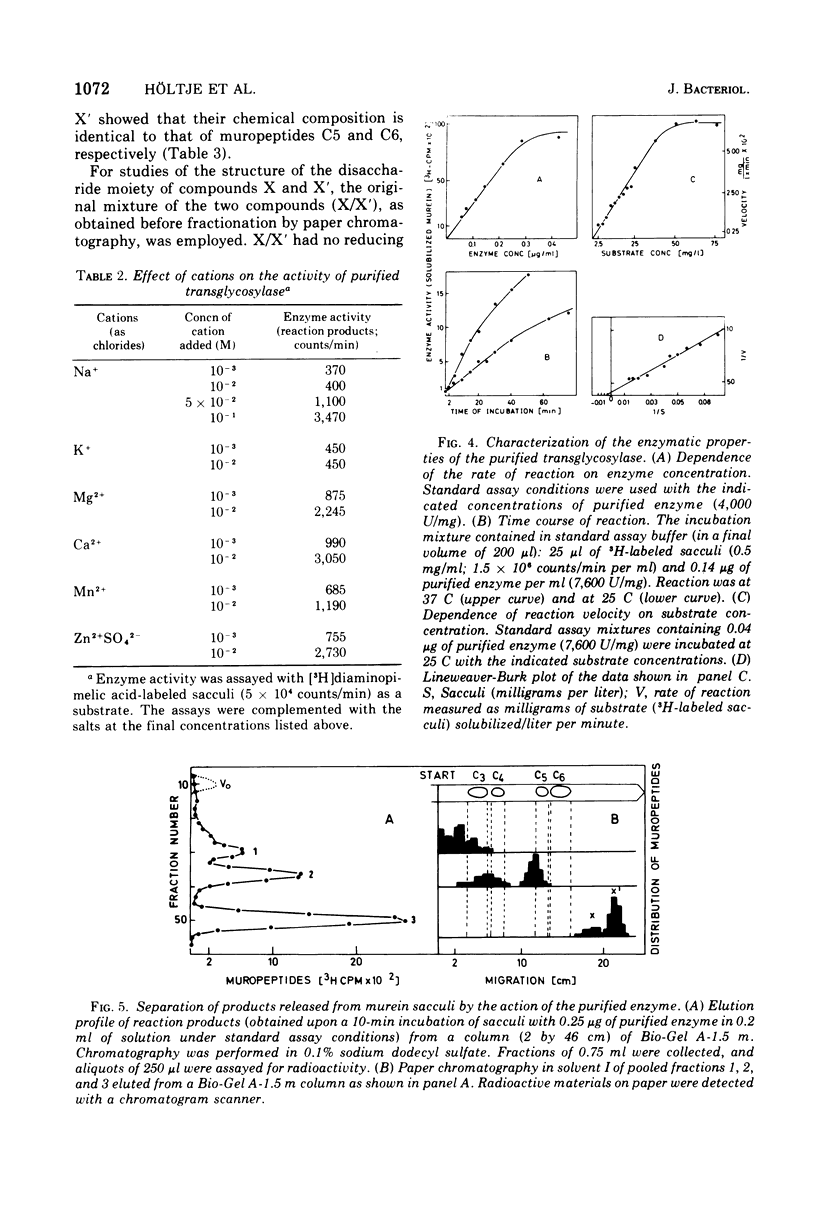
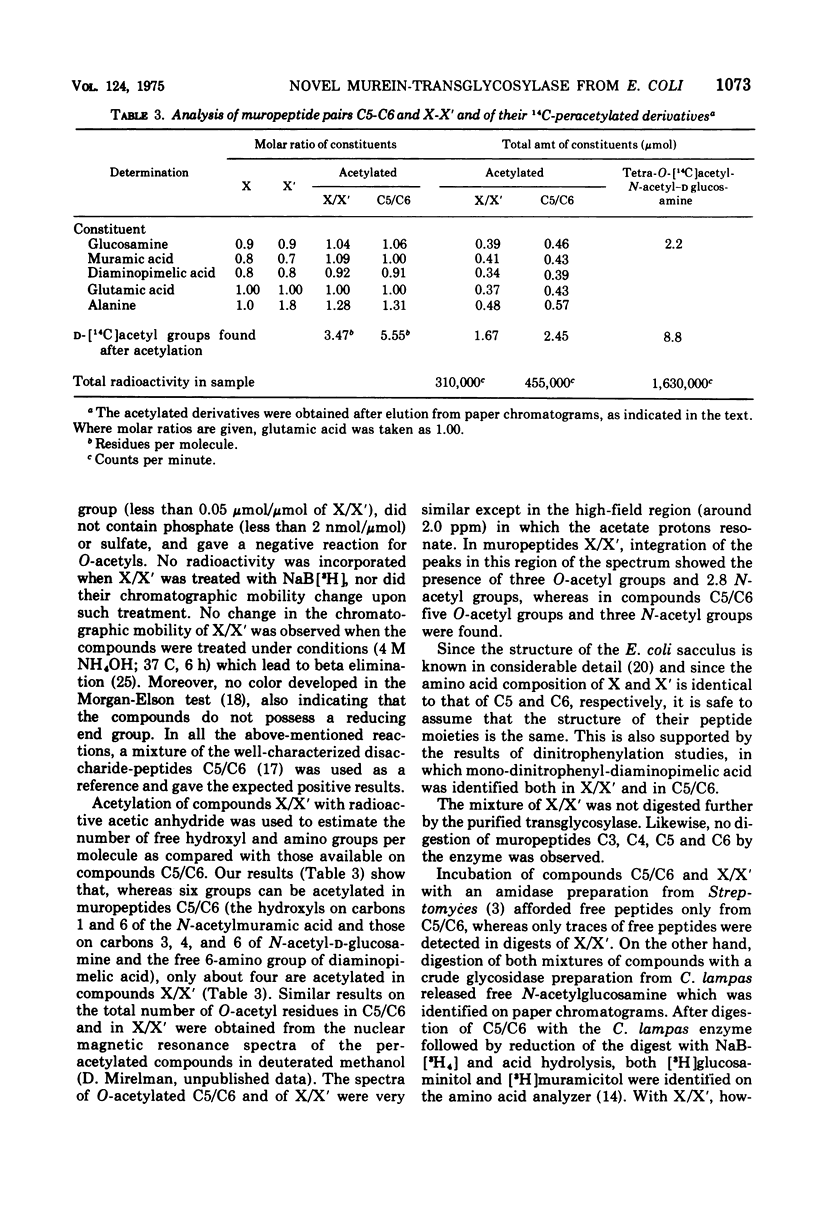
The purification and properties of a novel type of murein transglycosylase from Escherichia coli are described. The purified enzyme appears as a single band on sodium dodecyl sulfate-polyacrylamide gels and has an apparent molecular weight of approximately 65,000 as estimated by gel filtration and gel electrophoresis. It degrades pure murein sacculi from E. coli almost completely into low-molecular-weight products. The two prominent muropeptide fragments in the digest are the disaccharide-tripeptide N-acetylglucosamine-N-acetylmuramic acid-L-alanine-D-iso-glutamic acid-meso-diaminopimelic acid and the corresponding disaccharide-tetrapeptide N-acetylglucosamine-N-acetylmuramic acid-L-alanine-D-iso-glutamic acid-meso-diaminopimelic acid-D-alanine. The unique feature of these compounds is that the disaccharide has no reducing end group and that the muramic acid residue possesses an internal 1 leads to 6 anhydro linkage. The new lytic enzyme is designated as a murein: murein transglycosylase. Its possible role in the rearrangement of murein during cell growth and division is discussed.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andrews P. Estimation of the molecular weights of proteins by Sephadex gel-filtration. Biochem J. 1964 May;91(2):222–233. doi: 10.1042/bj0910222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braun V., Sieglin U. The covalent murein-lipoprotein structure of the Escherichia coli cell wall. The attachment site of the lipoprotein on the murein. Eur J Biochem. 1970 Apr;13(2):336–346. doi: 10.1111/j.1432-1033.1970.tb00936.x. [DOI] [PubMed] [Google Scholar]
- Ghuysen J. M., Dierickx L., Coyette J., Leyh-Bouille M., Guinand M., Campbell J. N. An improved technique for the preparation of Streptomyces peptidases and N-acetylmuramyl-l-alanine amidase active on bacterial wall peptidoglycans. Biochemistry. 1969 Jan;8(1):213–222. doi: 10.1021/bi00829a031. [DOI] [PubMed] [Google Scholar]
- Hakenbeck R., Goodell E. W., Schwarz U. Compartmentalization of murein hydrolases in the envelope of Escherichia coli. FEBS Lett. 1974 Apr 1;40(2):261–264. doi: 10.1016/0014-5793(74)80240-1. [DOI] [PubMed] [Google Scholar]
- Hartmann R., Bock-Hennig S. B., Schwarz U. Murein hydrolases in the envelope of Escherichia coli. Properties in situ and solubilization from the envelope. Eur J Biochem. 1974 Jan 3;41(1):203–208. doi: 10.1111/j.1432-1033.1974.tb03261.x. [DOI] [PubMed] [Google Scholar]
- Hartmann R., Höltje J. V., Schwarz U. Targets of penicillin action in Escherichia coli. Nature. 1972 Feb 25;235(5339):426–429. doi: 10.1038/235426a0. [DOI] [PubMed] [Google Scholar]
- Höltje J. V., Schwarz U. Penicillin and the murein-transglycosaminidase of Escherichia coli. Ann N Y Acad Sci. 1974 May 10;235(0):294–299. doi: 10.1111/j.1749-6632.1974.tb43272.x. [DOI] [PubMed] [Google Scholar]
- LEUTGEB W., WEIDEL W. UBER EIN IN COLI-ZELLWANDPRAEPARATEN ZURUECKGEHALTENES GLYKOGEN. Z Naturforsch B. 1963 Dec;18:1060–1062. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- McLean C., Werner D. A., Aminoff D. Quantitative determination of reducing sugars, oligosaccharides, and glycoproteins with (3H)borohydride. Anal Biochem. 1973 Sep;55(1):72–84. doi: 10.1016/0003-2697(73)90291-1. [DOI] [PubMed] [Google Scholar]
- PARK J. T., JOHNSON M. J. A submicrodetermination of glucose. J Biol Chem. 1949 Nov;181(1):149–151. [PubMed] [Google Scholar]
- PELZER H. MUCOPEPTIDHYDROLASEN IN ESCHERICHIA COLI B. I. NACHWEIS UND WIRKUNGSSPEZIFITAET. Z Naturforsch B. 1963 Nov;18:950–956. [PubMed] [Google Scholar]
- PRIMOSIGH J., PELZER H., MAASS D., WEIDEL W. Chemical characterization of mucopeptides released from the E. coli B cell wall by enzymic action. Biochim Biophys Acta. 1961 Jan 1;46:68–80. doi: 10.1016/0006-3002(61)90647-3. [DOI] [PubMed] [Google Scholar]
- Ryter A., Hirota Y., Schwarz U. Process of cellular division in Escherichia coli growth pattern of E. coli murein. J Mol Biol. 1973 Jun 25;78(1):185–195. doi: 10.1016/0022-2836(73)90437-3. [DOI] [PubMed] [Google Scholar]
- SHARON N., JEANLOZ R. W. The diaminohexose component of a polysaccharide isolated from Bacillus subtilis. J Biol Chem. 1960 Jan;235:1–5. [PubMed] [Google Scholar]
- SHARON N., SEIFTER S. A TRANSGLYCOSYLATION REACTION CATALYZED BY LYSOZYME. J Biol Chem. 1964 Jul;239:PC2398–PC2399. [PubMed] [Google Scholar]
- Schleifer K. H., Kandler O. Peptidoglycan types of bacterial cell walls and their taxonomic implications. Bacteriol Rev. 1972 Dec;36(4):407–477. doi: 10.1128/br.36.4.407-477.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwarz U., Asmus A., Frank H. Autolytic enzymes and cell division of Escherichia coli. J Mol Biol. 1969 May 14;41(3):419–429. doi: 10.1016/0022-2836(69)90285-x. [DOI] [PubMed] [Google Scholar]
- VOGEL H. J., BONNER D. M. Acetylornithinase of Escherichia coli: partial purification and some properties. J Biol Chem. 1956 Jan;218(1):97–106. [PubMed] [Google Scholar]
- WEIDEL W., PELZER H. BAGSHAPED MACROMOLECULES--A NEW OUTLOOK ON BACTERIAL CELL WALLS. Adv Enzymol Relat Areas Mol Biol. 1964;26:193–232. doi: 10.1002/9780470122716.ch5. [DOI] [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]



