Abstract
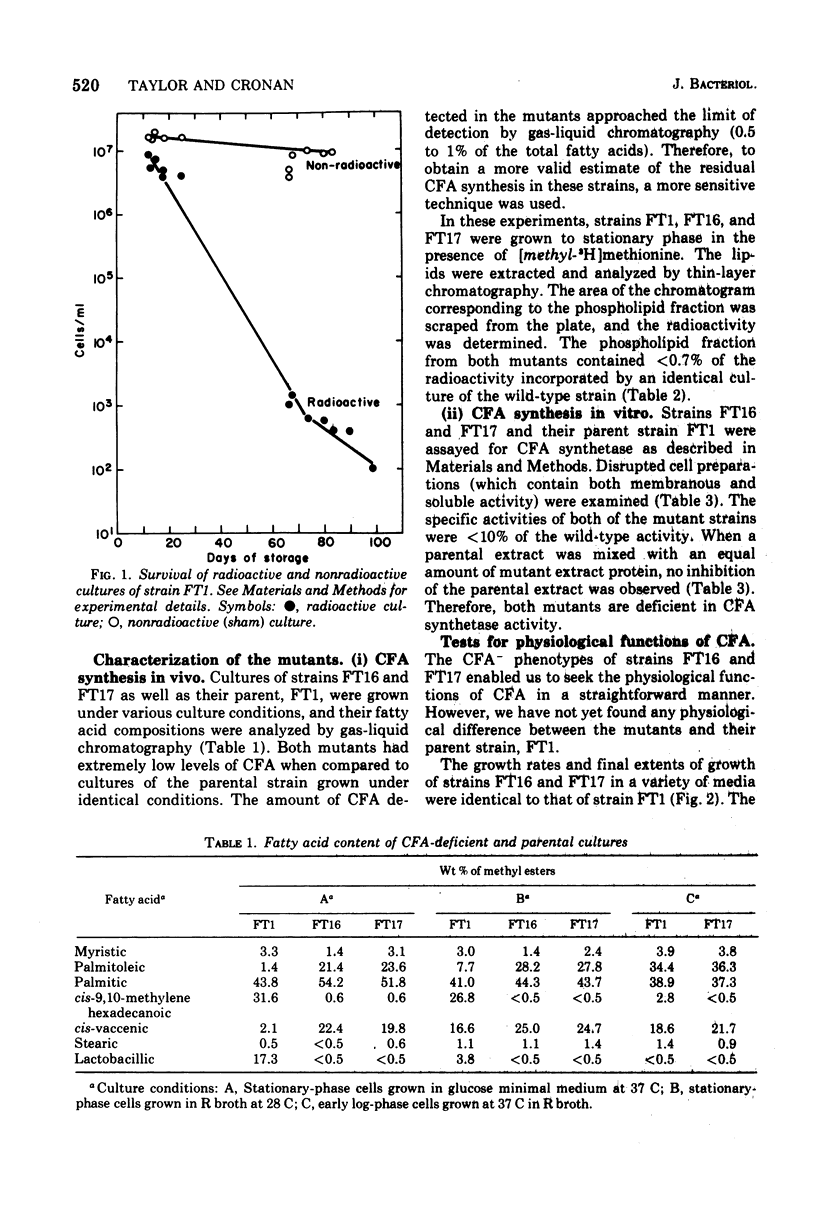
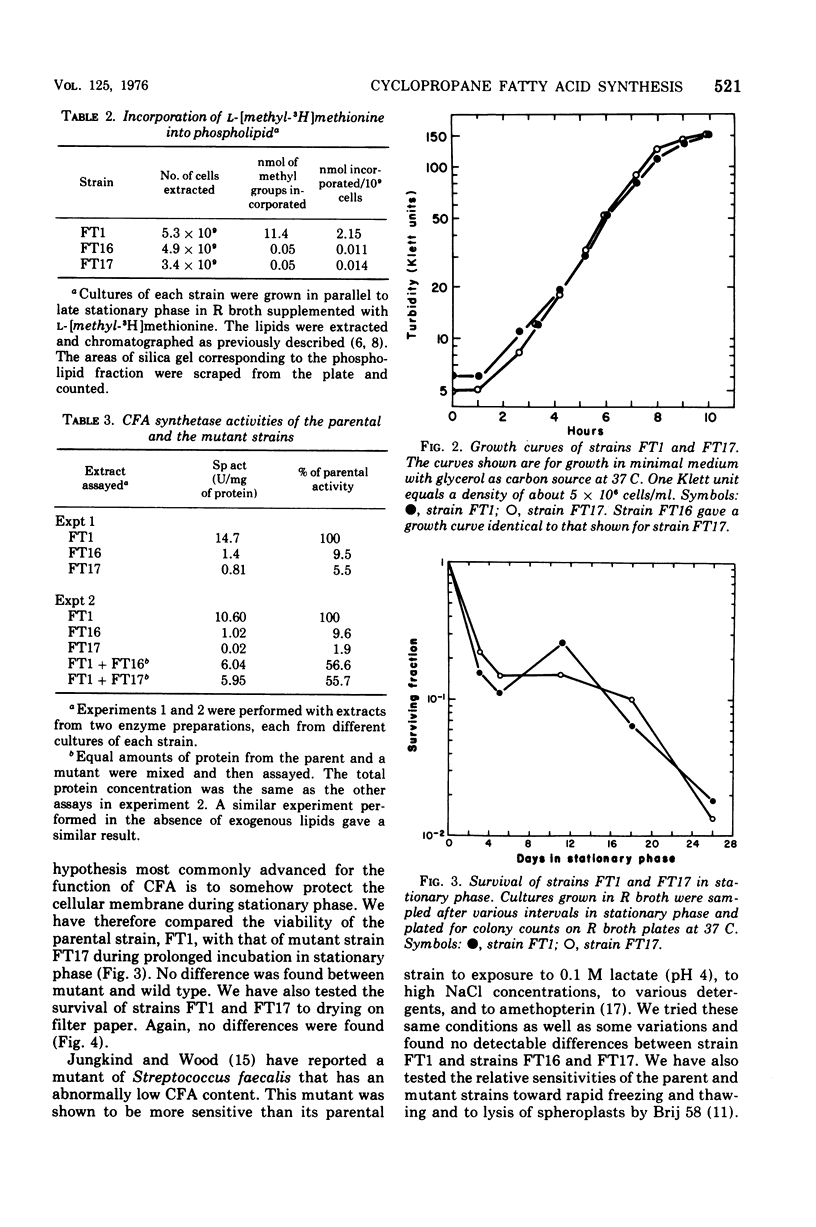
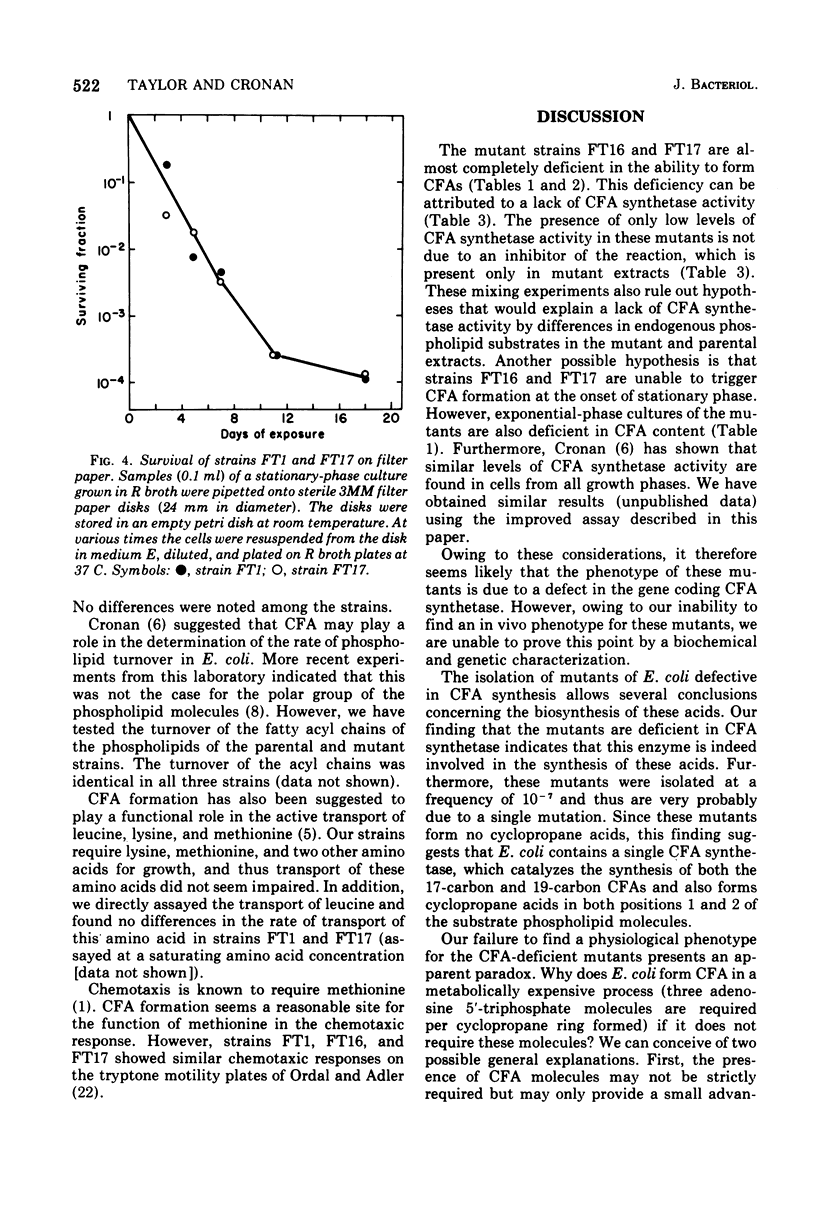
Mutants of Escherichia coli K-12 defective in the synthesis of cyclopropane fatty acids (CFA) have been selected and isolated by a L-[methyl-3H]methionine suicide procedure. Two mutants were isolated. Stationary-phase cultures of both mutants contain less than 0.7% of the CFA content found in the parental strain. The CFA deficiency is attributed to a deficiency of CFA synthetase activity. Extracts of both mutants contain less than 10% of the CFA synthetase activity found in extracts of the parental strain. Experiments in which parental and mutant extracts were mixed indicate that the lack of activity in the mutant strains is not due to an inhibitor of CFA synthetase present in the mutant extracts. We have not yet detected a physiological phenotype for these mutants. These strains grow normally at various temperatures in a variety of media. We have tested survival (colony-forming ability) in response to (i) prolonged incubation in stationary phase, (ii) exposure to drying, and (iii) exposure to detergents, heavy metals, low pH, high salt concentration, and a variety of other environmental conditions. The survival of both mutants is identical to that of the parental strain under all conditions tested. The compositions (excepting the CFA deficiency) and metabolic turnover rates of the phospholipids of both mutant strains are indistinguishable from those of the wild-type strain. The transport of several amino acids also seems normal in these mutants.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adler J. Chemotaxis in bacteria. Annu Rev Biochem. 1975;44:341–356. doi: 10.1146/annurev.bi.44.070175.002013. [DOI] [PubMed] [Google Scholar]
- Berg C. M., Curtiss R., 3rd Transposition derivatives of an Hfr strain of Escherichia coli K-12. Genetics. 1967 Jul;56(3):503–525. doi: 10.1093/genetics/56.3.503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Body D. R. The occurrence of cyclopropane fatty acids in the phospholipids of sheep rumen tissues. FEBS Lett. 1972 Oct 15;27(1):5–8. doi: 10.1016/0014-5793(72)80395-8. [DOI] [PubMed] [Google Scholar]
- Cox G. S., Kaback H. R., Weissbach H. Defective transport in S-adenosylmethionine synthetase mutants of Escherichia coli. Arch Biochem Biophys. 1974 Apr 2;161(2):610–620. doi: 10.1016/0003-9861(74)90344-0. [DOI] [PubMed] [Google Scholar]
- Cronan J. E., Jr A new method for selection of Escherichia coli mutants defective in membrane lipid synthesis. Nat New Biol. 1972 Nov 1;240(96):21–22. doi: 10.1038/newbio240021a0. [DOI] [PubMed] [Google Scholar]
- Cronan J. E., Jr, Nunn W. D., Batchelor J. G. Studies on the biosynthesis of cyclopropane fatty acids in Escherichia coli. Biochim Biophys Acta. 1974 Apr 26;348(1):63–75. doi: 10.1016/0005-2760(74)90093-9. [DOI] [PubMed] [Google Scholar]
- Cronan J. E., Jr Phospholipid alterations during growth of Escherichia coli. J Bacteriol. 1968 Jun;95(6):2054–2061. doi: 10.1128/jb.95.6.2054-2061.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cronan J. E., Jr, Ray T. K., Vagelos P. R. Selection and characterization of an E. coli mutant defective in membrane lipid biosynthesis. Proc Natl Acad Sci U S A. 1970 Mar;65(3):737–744. doi: 10.1073/pnas.65.3.737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Godson G. N., Sinsheimer R. L. Lysis of Escherichia coli with a neutral detergent. Biochim Biophys Acta. 1967 Dec 19;149(2):476–488. doi: 10.1016/0005-2787(67)90175-x. [DOI] [PubMed] [Google Scholar]
- Goldfine H. Comparative aspects of bacterial lipids. Adv Microb Physiol. 1972;8:1–58. doi: 10.1016/s0065-2911(08)60187-3. [DOI] [PubMed] [Google Scholar]
- Goldfine H. Use of a filter-paper disk assay in the measurement of lipid biosynthesis. J Lipid Res. 1966 Jan;7(1):146–149. [PubMed] [Google Scholar]
- Harder M. E., Beacham I. R., Cronan J. E., Jr, Beacham K., Honegger J. L., Silbert D. F. Temperature-sensitive mutants of Escherichia coli requiring saturated and unsaturated fatty acids for growth: isolation and properties. Proc Natl Acad Sci U S A. 1972 Nov;69(11):3105–3109. doi: 10.1073/pnas.69.11.3105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jungkind D. L., Wood R. C. Physiological differences between cyclopropane fatty acid-deficient mutants and the parent strain of Streptococcus faecalis. Biochim Biophys Acta. 1974 Feb 25;337(2):298–310. doi: 10.1016/0005-2760(74)90211-2. [DOI] [PubMed] [Google Scholar]
- Kuiper P. J., Stuiver B. Cyclopropane Fatty acids in relation to earliness in spring and drought tolerance in plants. Plant Physiol. 1972 Mar;49(3):307–309. doi: 10.1104/pp.49.3.307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marinari L. A., Goldfine H., Panos C. Specificity of cyclopropane fatty acid synthesis in Escherichia coli. Utilization of isomers of monounsaturated fatty acids. Biochemistry. 1974 Apr 23;13(9):1978–1983. doi: 10.1021/bi00706a030. [DOI] [PubMed] [Google Scholar]
- Oeschger M. P., Berlyn M. K. A simple procedure for localized mutagenesis using nitrosoguanidine. Mol Gen Genet. 1974;134(1):77–83. doi: 10.1007/BF00332814. [DOI] [PubMed] [Google Scholar]
- Ordal G. W., Adler J. Isolation and complementation of mutants in galactose taxis and transport. J Bacteriol. 1974 Feb;117(2):509–516. doi: 10.1128/jb.117.2.509-516.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Overath P., Schairer H. U., Stoffel W. Correlation of in vivo and in vitro phase transitions of membrane lipids in Escherichia coli. Proc Natl Acad Sci U S A. 1970 Oct;67(2):606–612. doi: 10.1073/pnas.67.2.606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tosa T., Pizer L. I. Biochemical bases for the antimetabolite action of L-serine hydroxamate. J Bacteriol. 1971 Jun;106(3):972–982. doi: 10.1128/jb.106.3.972-982.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tosa T., Pizer L. I. Effect of serine hydroxamate on the growth of Escherichia coli. J Bacteriol. 1971 Jun;106(3):966–971. doi: 10.1128/jb.106.3.966-971.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VOGEL H. J., BONNER D. M. Acetylornithinase of Escherichia coli: partial purification and some properties. J Biol Chem. 1956 Jan;218(1):97–106. [PubMed] [Google Scholar]


