Abstract
Transforming growth factor-beta (TGFβ)1 is thought to be implicated in breast cancer progression. However, data about the influence of TGFβ1 on breast cancer development are conflicting. To clarify the clinical relevance of TGFβ1, TGFβ1 protein level has been measured by enzyme-immoassay in 193 breast tumour samples. We found that 94.3% of patients expressed TGFβ1 with a range of 0–684 pg mg−1 protein. In the overall population, an increase of tumoral TGFβ1 was observed in premenopausal patients when compared to postmenopausal subgroup (P=0.0006). When patients were subdivided according to nodal status, TGFβ1 was correlated to type-1 plasminogen activator inhibitor in the node-negative subgroup (P=0.040). Multivariate analysis revealed that, after lymph node status (P=0.0002) and urokinase-type plasminogen activator (P=0.004), TGFβ1 was an independent prognostic marker for DFS (P=0.005) in the overall population. In the node-negative population, TGFβ1 was the prominent prognostic factor (P=0.010). In the same population, Kaplan–Meier curves demonstrated that high TGFβ1 level was correlated with a shorter disease-free survival (P=0.020). These data suggest that the measurement of tumoral TGFβ1 protein level, especially for node-negative patients, might help to identify a high-risk population early in tumour progression.
Keywords: transforming growth factor-beta1, breast cancer, survival
Transforming growth factor-beta (TGFβ) belongs to a superfamily of secreted polypeptides, which regulate cell proliferation, differentiation, motility and apoptosis in a variety of different cell types (Roberts and Sporn, 1990). Three TGFβ isoforms 1–3 are ubiquitously expressed and have been detected in humans and other mammals. TGFβ1 have been associated with both normal mammary gland development and breast carcinogenesis (Wakefield et al, 2000). Thus, in vivo, TGFβ1 appears to regulate normal ductal and alveolar development in the mammary gland (Jhappan et al, 1993). Moreover, TGFβ1 probably mediates the massive cell death and restructuring that takes place during postlactational involution of the mammary gland (Strange et al, 1992). Besides these physiological functions, there is considerable evidence that TGFβ1 is implicated in several aspects of breast cancer onset and progression (Wakefield et al, 2000). A commonly held view is that TGFβ1 prohibits tumour cell proliferation because TGFβ1 is a potent growth inhibitor for nearly all epithelia in vitro (Roberts and Sporn, 1990). Moreover, TGFβ1 can induce apoptosis, a process associated with tumour suppression, promote replicative senescence and exhibit negative regulation of angiogenesis (Alexander and Moses, 1995; Schwarte-Waldhoff et al, 2000; Perlman et al, 2001). Consistent with a tumour-suppressor role, transgenic mouse models have bring evidence that TGFβ1 is able to protect against mammary tumour development in vivo, because either the suppression of TGFβ1 or the inactivation of the TGFβ signalling pathway results in loss of tumour prohibition and promotes carcinogenesis (Pierce et al, 1995; Bottinger et al, 1997). On the other hand, virally transformed tumorigenic mammary epithelial cell lines as well as most of the cell lines derived from invasive human breast carcinomas are resistant to the antiproliferative effects of TGFβ1 in vitro and do not respond to treatment with TGFβ1 in vivo. In a number of cases, this is attributable to inhibiting mutations in either TGFβ type I or II receptors (Chen et al, 1998; Gobbi et al, 2000) or deregulation of the downstream signalling cascade (Xie et al, 2002).
In addition, there is increasing evidence that after cells lose their sensitivity to TGFβ1-mediated growth inhibition, autocrine TGFβ signalling may promote tumorigenesis. Consistent with a pro-oncogenic role for TGFβ1 in late-stage cancer, elevated levels of TGFβ1 are often observed in advanced carcinomas, and have been correlated with increased tumour invasiveness and disease progression in a variety of tumours such as malignant melanoma (Moretti et al, 1999) and colonic (Tsushima et al, 1996), ovarian (Bristow et al, 1999), and prostatic (Shariat et al, 2004) cancers. This suggests that secreting higher levels of TGFβ1 may provide an advantage to tumour cells. Pro-oncogenic effects include direct effects of TGFβ1 on tumoral cells, such as the stimulation of invasion and motility (Oft et al, 1996). Tumour TGFβ1 may also indirectly promote cancer progression by promoting tumour vascularisation (Oh et al, 2000) and inhibiting mechanisms of immune surveillance (Letterio and Roberts, 1998).
In breast cancer, a number of studies have been engaged to evaluate the potential prognostic value of TGFβ1. In most of these studies, tumoral TGFβ1 expression has been examined either by immuno-histochemical assay or by Northern blot analysis. Although several groups demonstrated that increased TGFβ1 was associated with more aggressive tumour behavior and poorer survival (Gorsch et al, 1992; Dalal et al, 1993; Walker et al, 1994), other authors reported the absence of correlation between disease progression and TGFβ1 immunostaining or mRNA level (Dublin et al, 1993; Ghellal et al, 2000). Furthermore, a few number of studies demonstrated that TGFβ1 was related to favorable outcome for patients with breast cancers (Murray et al, 1993; Kesari et al, 1999).
To date, quantitative determination of TGFβ1 protein level has been performed exclusively in plasma of breast cancer patients. While some studies failed to reveal any change in plasma TGFβ value in patients with breast cancer (Wakefield et al, 1995; Lebrecht et al, 2004), other reports demonstrate that patients with more advanced tumours have higher serum levels of TGFβ1 (Kong et al, 1995; Ivanovic et al, 2003), suggesting that serum TGFβ1 may reflect the severity of invasive breast cancer. However, these late studies have been performed on a small cohort of patients and potential prognostic value of TGFβ1 has not been clearly determined. To clarify the relevance of TGFβ1 as a prognostic marker in human breast cancer, we quantified TGFβ1 protein level in 193 breast tissue specimens. The association between TGFβ1 and the usual histological and biological parameters previously validated was examined. The prognostic relevance of TGFβ1 for disease-free (DFS) and overall survival (OS) was studied for all patients by Cox multivariable analysis including the traditional prognostic markers.
MATERIALS AND METHODS
Patient population
This study involved 193 patients diagnosed and treated in Assistance Publique of Marseille (France) between early 1987 and late 1992. These patients were previously included in a retrospective multicenter study (Romain et al, 2000). Patients were selected according to the following criteria: (1) primary unilateral breast tumour; (2) previously untreated, no evidence of metastatic disease or any other malignancy at the time of diagnosis; (3) T1T2, N0N1 status according to UICC criteria; (4) <75 years old; and (5) surgery as the first treatment.
The patients were 29–74 years old at diagnosis, with a median age of 57 years. In total, 37.3% of patients were premenopausal. A total of 83 patients presented a tumour size ⩽2 cm; 48.7% of patients were node-negative (N−), 29.6% presented one to three axillary invaded nodes (N+) and 21.7% had more than three invaded nodes (N++). Among the 193 tumours graded according to Scarff, Bloom and Richardson classification (SBR), 25.5% were classified grade I, 52.8% were grade II, and 21.7% were grade III. Ductal carcinomas were diagnosed in 75% of patients, and invasive lobular carcinomas in 25% of patients.
The primary treatment was tumorectomy or quadrantectomy (92%) or modified radical mastectomy (8%) with axillary dissection, followed by radiotherapy in 98% of cases. Among the 121 postmenopausal patients, 21 received no adjuvant treatment whereas 33 received hormone therapy, 33 were treated with chemotherapy, and 34 received both treatments. Among the 72 premenopausal patients, 36 received no adjuvant therapy and 36 were treated as follow: 10 with chemotherapy, 22 with hormone therapy and four patients with both treatments. The median follow-up was 94 months (range, 1–140). At the cutoff date of this study, 16 local recurrences, 42 metastasis and 28 deaths had been recorded. Tumour samples and clinical informations were obtained under Institutional Clinical Board approval.
Preparation of tumour tissue extracts and ER and PR assays
Tumour tissues were stored in liquid nitrogen and routinely assayed for estrogen (ER) and progesterone receptors (PR) levels, according the recommendations of the European Organization for Research and Treatment of Cancer (EORTC), as previously described (Foekens et al, 1989). Tumour tissues were pulverised in the frozen state with a microdismembrator (Braun, Melsungen, Germany) as recommended by the EORTC. The resulting powder was suspended in buffer containing 10 mM Tris-HCl pH 7.4, 1.5 mM EDTA, 10 mM Na2MoO4, 0.5 mM DTT and 10% glycerol. The suspension was centrifuged for 60 min at 105 000 g at 4°C. The high-speed supernatants (cytosols) were collected and stored in liquid nitrogen. For all samples, cytosolic protein concentration was determined using BCA assay (Pierce Chemical, Rockford, IL, USA). ER and PR levels were determined by enzyme immunoassay as described previously (Foekens et al, 1989). To assess the between-assay variations, in each series of tests an aliquot of a pooled breast cancer cytosol sample was analyzed.
The remaining cytosols were frozen and stored in liquid nitrogen until used for the determination of thymidine kinase (TK) enzyme activity, urokinase-type plasminogen activator (uPA), type-1 plasminogen activator inhibitor (PAi-1), and TGFβ1.
TK, uPA and PAi-1 assays
TK enzyme activity was measured using the Prolifigen TK Radioenzymatic Assay (Sangtec Medical, Bromma, Sweden), with the modifications recommended by the EORTC Receptors and Biomarkers Study Group (Foekens et al, 2001).
uPA levels were measured with the Immunobind® uPA ELISA kit and PAi-1 levels by the Immunobind® PAi1 ELISA kit (American Diagnostic, Greenwich, CT, USA), according to the instructions of the manufacturer. Inactive and active forms of uPA are all recognised by the uPA ELISA kit, as is receptor-bound uPA and uPA complexed with PAi-1 and PAi-2. PAi-1 ELISA detects latent and active forms of human PAi-1 and PAi-1 complexes. The assay is insensitive to PAi-2.
TGFβ1 measurement
TGFβ1 levels in breast tumour cytosols were measured by ELISA. This assay used monoclonal antibody (R&D Systems, UK) as capture antibody and biotinylated polyclonal antibody (R&D Systems, UK) as detection antibody. The assay specifically measures active TGFβ1 forms. To measure total TGFβ1 present in tumour samples, biologically latent TGFβ1 was activated by acid-treatment. For this purpose, cytosols were diluted with four volumes of DPBS buffer (2.7 mM KCl, 137 mM NaCl, 1.5 mM KH2PO4, 3.2 mM Na2HPO4, 1 mM CaCl2, 0.5 mM MgCl2, pH 7.4). Samples were then incubated for 15 min at room temperature in the presence of 0.02 vol of 1 N HCl, then neutralised with equal volume of 1 N NaOH. ELISA analysis was performed in 96-well plates following the instructions of the manufacturer (R&D Systems, UK). Recombinant human TGFβ1 (R&D Systems, UK) was used as standard at 0–1000 pg ml−1. A preliminary evaluation was performed to assess the buffer compatibility and the parallelism of sample dilutions. The inter- and intra-assay (n=10) CVs of a pool of tumour extracts with mean value of 134.1 pg TGFβ1 per mg protein were 7.5 and 3.9% respectively.
Statistical analysis
The strength of the associations of TGFβ1 with other variables was tested with Spearman rank correlation. The associations of TGFβ1 (used as continuous variables) with other variables (used as grouping variables) were examined using Mann–Whitney U test (two categories), or in the case of more than two ordered categories by Kruskal–Wallis test. Survival curves were generated using the method of Kaplan and Meier and the log-rank test for trend was used to examine survival data. For the univariate survival analysis, DFS time (the interval between date of surgery and primary failure defined as a locoregional and/or distant recurrence) and OS time (the interval between date of surgery and death by any cause) were used as follow-up parameters. P-values ⩽0.05 were considered as significant.
Cox multivariate regression analysis was used to evaluate the prognostic value of TGFβ1 in the overall or N+ or N− populations. Multivariate analysis was performed with variables eliminated in a step-down fashion. Variables with a P⩽0.05 were retained in the final multivariate models. Hazard ratios (HR) derived from the estimated regression coefficients, are presented with their 95% confidence intervals (CI).
Variables were categorised as follows: age (50 or younger, and older than 50), pathological tumour size (⩽20 mm or >20 mm), menopausal status (premenopausal vs postmenopausal), pathological nodal status (N−, none; N+, 1 to 3; N++, more than 3), histologic grade (SBR grade I, II or III), and histologic type (ductal vs lobular).
In regard to the variations of ER levels observed in premenopausal vs postmenopausal patients (39), all tumours were considered to be estrogen receptor-negative (ER−) if ER values <15 fmol mg−1 protein; for the premenopausal population, tumours with ER 15–205 fmol mg−1 protein (75th percentile) were classified ER+, whereas tumours of postmenopausal patients were considered as ER+ when ER level was 15–377 fmol mg−1 protein (75th percentile). In both pre- and post-menopausal populations, ER++ represents tumours with ER values exceeding the 75th percentile. In all cases, the tumours were considered to be PR-positive if values exceeded 20 fmol mg−1 protein. For all others biological parameters, cutpoints corresponded to the 25th and 75th percentiles of the distribution (see Table 1).
Table 1. Patient characteristics.
| Feature | Category | No. of patients | Percentage |
|---|---|---|---|
| Total population | 193 | ||
| Age (years) | ⩽50 | 62 | 32.1 |
| >50 | 131 | 67.9 | |
| Hormonal status | Premenopausal | 72 | 37.3 |
| Menopausal | 121 | 62.7 | |
| Receptor statusa | ER− PR− | 21 | 10.9 |
| ER− PR+ | 7 | 3.6 | |
| ER+ PR− | 39 | 20.2 | |
| ER+ PR+ | 126 | 65.3 | |
| Histology | Invasive duct | 145 | 75.1 |
| Invasive lobular | 48 | 24.9 | |
| Tumour size | T1 | 83 | 43.0 |
| T2 | 110 | 57.0 | |
| SBR grade | I | 49 | 25.5 |
| II | 102 | 52.8 | |
| III | 42 | 21.7 | |
| Nodal status | N− | 94 | 48.7 |
| N+ | 57 | 29.6 | |
| N++ | 42 | 21.7 |
The subgroups ER+ include the patients with ER+ and ER++ (ER>15 fmol mg−1 protein), as defined in Materials and Methods section.
RESULTS
Clinicopathological characteristics
The clinicopathological characteristics of the patients are presented in Table 1. Patients were characterised according to their age, hormonal (menopausal) and steroid receptor status, tumour grade according the SBR grading system, histology and size of the tumour, and the axillary nodal status.
Biological characteristics of the breast cancer samples analyzed
The distribution of biological factors in breast cancer samples are listed in Table 2. A wide inter-patient variability in the levels of all the parameters measured could be observed. ER, PR and TK levels were previously determined in our laboratory and integrated elsewhere in other published study (Romain et al, 1995, 2000). uPA and PAi-1 levels ranged from 0.01 to 1.39 ng mg−1 protein (median, 0.20) and from 0.27 to 54 ng mg−1 protein (median, 6.0), respectively. TGFβ1 was detectable in 94.3% of samples and its concentration ranged from 0 to 684 pg mg−1 protein, with a median at 86.7 pg mg−1 protein.
Table 2. Biological characteristics of the breast cancer population.
| Variables | Range | Mean (IC) a | Q25 | Q50 | Q75 |
|---|---|---|---|---|---|
| ER (fmol mg−1 protein) | |||||
| Overall population | 0–654 | 197 (23.9) | 33 | 149 | 325 |
| Premenopausal | 1–455 | 122 (26.8) | 22 | 87 | 205 |
| Postmenopausal | 0–654 | 218 (22.4) | 67 | 254 | 377 |
| PR (fmol mg−1 protein) | 0–1000 | 156 (26.3) | 8.7 | 67 | 274 |
| TK (mU mg−1 protein) | 4–2804 | 349 (66.9) | 70 | 138 | 388 |
| uPA (ng mg−1 protein) | 0.01–1.39 | 0.28 (0.03) | 0.11 | 0.20 | 0.38 |
| PAi-1 (ng mg−1 protein) | 0.27–54 | 8.73 (1.19) | 3.7 | 6.0 | 10 |
| TGFβ1 (pg mg−1 protein) | 0–684 | 121 (16.6) | 42 | 86.7 | 148 |
95% confidence interval, CI.
Relationships between TGFβ1 and clinicopathological and biological parameters
When the correlation between TGFβ1 and each of the others parameters was examined in the overall population, no significant correlation could be observed between TGFβ1 and the biological and clinicopathological variables, except the hormonal status. Thus, premenopausal patients were found to express higher TGFβ1 levels than postmenopausal patients (114 vs 86 pg mg−1 protein, P=0.0006) (not shown). When patient population was subdivided according to pathological nodal status, TGFβ1 remained correlated to the hormonal status in both node-negative (P=0.012) and node-positive (P=0.008) subgroups (Table 3). Moreover, a positive association between TGFβ1 and PAi-1 (P=0.040) was observed in the node-negative population.
Table 3. Relationship between TGFβ1 and biological and clinicopathological variables.
|
Node-negative patients
|
|||||
|---|---|---|---|---|---|
| n | Range | Q50 | P | ρ | |
| Hormonal status | |||||
| Premenopausal | 35 | 8–567 | 132 | ||
| Postmenopausal | 59 | 0–349 | 74 | 0.012 | −0.259 |
| Tumour size | |||||
| T1 | 61 | 0–349 | 74 | ||
| T2 | 33 | 3–367 | 91 | 0.725 | 0.036 |
| SBR grade | |||||
| I | 30 | 3–349 | 100 | ||
| II | 48 | 0–367 | 82 | ||
| III | 16 | 3–337 | 79 | 0.885 | 0.015 |
| ER | |||||
| Negative | 17 | 17–259 | 75 | ||
| Low | 56 | 0–367 | 85 | ||
| High | 21 | 9–349 | 117 | 0.906 | 0.094 |
| PgR | |||||
| Negative | 30 | 3–341 | 72 | ||
| Positive | 64 | 0–349 | 104 | 0.696 | 0.041 |
| TK | |||||
| Negative | 26 | 3–349 | 102 | ||
| Low | 50 | 0–337 | 69 | ||
| High | 18 | 15–684 | 132 | 0.293 | 0.108 |
| uPA | |||||
| Negative | 28 | 0–303 | 72 | ||
| Low | 42 | 7–349 | 107 | ||
| High | 24 | 4–337 | 79 | 0.380 | 0.091 |
| PAI-1 | |||||
| Negative | 26 | 0–167 | 72 | ||
| Now | 47 | 3–366 | 81 | ||
| High | 21 | 3–684 | 149 | 0.040 | 0.211 |
|
Node-positive patients
|
|||||
|---|---|---|---|---|---|
| n | Range | Q50 | P | ρ | |
| Hormonal status | |||||
| Premenopausal | 37 | 13–684 | 120 | ||
| Postmenopausal | 62 | 2–405 | 71 | 0.008 | −0.265 |
| Tumour size | |||||
| T1 | 22 | 5–679 | 88 | ||
| T2 | 77 | 2–684 | 76 | 0.943 | −0.007 |
| SBR grade | |||||
| I | 19 | 4–346 | 76 | ||
| II | 54 | 2–643 | 98 | ||
| III | 26 | 2–684 | 62 | 0.680 | −0.041 |
| ER | |||||
| Negative | 10 | 14–433 | 107 | ||
| Low | 62 | 3–643 | 83 | ||
| High | 27 | 3–684 | 89 | 0.728 | 0.035 |
| PgR | |||||
| Negative | 32 | 3–684 | 101 | ||
| Positive | 67 | 2–678 | 87 | 0.929 | 0.009 |
| TK | |||||
| Negative | 20 | 3–257 | 83 | ||
| Low | 42 | 2–387 | 107 | ||
| High | 37 | 10–325 | 82 | 0.749 | 0.032 |
| uPA | |||||
| Negative | 17 | 3–348 | 87 | ||
| Low | 56 | 2–684 | 96 | ||
| High | 26 | 6–151 | 90 | 0.793 | −0.027 |
| PAI-1 | |||||
| Negative | 21 | 3–643 | 82 | ||
| Now | 50 | 4–341 | 89 | ||
| High | 28 | 6–676 | 100 | 0.977 | −0.003 |
n=number of patients; Q50, median values.
Prognostic relevance
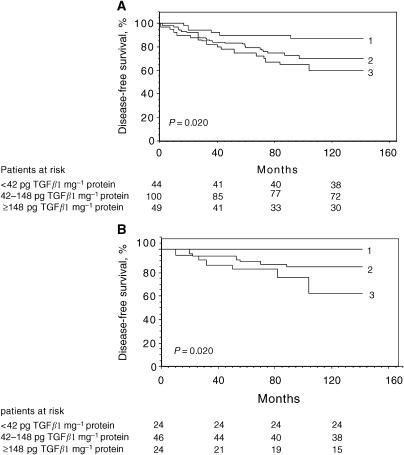
The impact of TGFβ1 on OS and DFS was determined in the overall population and node-negative/node-positive subsets. When 25th and 75th percentiles of the distribution were used as cutoff values, TGFβ1 appeared significant (P=0.020) for DFS in the overall population (Figure 1A). The 10-year probability of DFS was 86.4% for patients with low TGFβ1 levels (<42 pg mg−1 protein), 72% for the intermediate group (42–148 pg TGFβ1/mg protein) and 61% for patients with TGFβ1⩾148 pg mg−1 protein. The patients were then dichotomised according to their nodal status. While TGFβ1 was found to have no significant impact on DFS in the node-positive subgroup (not shown), high TGFβ1 levels were significantly associated with poor DFS in the node-negative population (P=0.02) (Figure 1B). Thus, among the node-negative patients with low TGFβ1 level, no relapse (DFS=100%) were observed, whereas 17 and 38% relapses were observed for patients with intermediate and high TGFβ1 expression levels, respectively. Unlikely, the level of TGFβ1 had no impact on OS, neither in the overall population nor in the node-positive/node-negative groups (not shown).
Figure 1.
The probability of DFS for overall (A) and node-negative (B) populations according to TGFβ1 levels. 1, negative (TGFβ1<42 pg mg−1 protein); 2, low (42 pg mg−1 protein ⩽TGFβ1 <148 pg mg−1 protein); 3, high (TGFβ1⩾148 pg mg−1 protein).
A Cox multivariate analysis was performed to evaluate whether TGFβ1 might significantly add to the contribution of the traditional prognostic factors. A significance level of 5% in the univariate analysis was chosen as the criterion for entering variables (SBR grade, nodal status, ER, uPA, PAi-1, TK and TGFβ1) (not shown). The analysis was conducted in the overall population and node-negative/node-positive subgroups (Table 4). The analysis performed for OS revealed nodal status (P=0.0003) and SBR grade (P=0.0008) as independent parameters in the overall population. The prominent predictor for OS was SBR grade (P=0.004) and ER (P=0.020) in the node-positive population, and uPA (P=0.010) in the node-negative subgroup. In addition of nodal status (P=0.0002) and uPA (P=0.004), TGFβ1 was independently associated to poor DFS in the overall population (P=0.005). Whereas PAi-1 appeared as the prominent independent predictor for the node-positive patients (P=0.019), the parameter associated with DFS in the population without node-infiltration was TGFβ1 (P=0.010).
Table 4. Cox multivariate analysis of OS and DFS, in overall population (n=193), and in node-positive (n=99) and node-negative patients (n=94).
|
DFS
|
OS
|
||||
|---|---|---|---|---|---|
| Variable category | Coding | HR (CI) | P | HR (CI) | P |
| Overall population | |||||
| Nodal status | |||||
| N− | 0 | 1.00 | 1.00 | ||
| N+ | 1 | 1.84 (1.33–2.54) | 2.31 (1.46–3.66) | ||
| N++ | 2 | 3.39 (1.77–6.45) | 0.0002 | 5.34 (2.13–13.4) | 0.0003 |
| SBR grade | |||||
| I | 0 | 1.00 | |||
| II | 1 | 2.89 (1.55–5.37) | |||
| III | 2 | 8.35 (2.42–28.8) | 0.0008 | ||
| UPA | |||||
| Negative | 0 | ||||
| Low | 1 | 2.00 (1.25–3.21) | |||
| High | 2 | 4.02 (1.57–10.3) | 0.004 | ||
| TGFβ1 | |||||
| Negative | 0 | 1.00 | |||
| Low | 1 | 1.83 (1.19–2.81) | |||
| High | 2 | 3.36 (1.43–7.91) | 0.005 | ||
| Node-positive patients | |||||
| PAi-1 | |||||
| Negative | 0 | 1.00 | |||
| Low | 1 | 2.09 (1.26–3.46) | |||
| High | 2 | 4.36 (1.58–11.7) | 0.019 | ||
| SBR grade | |||||
| I | 0 | 1.00 | |||
| II | 1 | 3.07 (1.43–6.62) | |||
| III | 2 | 9.46 (2.47–45.2) | 0.004 | ||
| ER | |||||
| Negative | 0 | 1.00 | |||
| Low | 1 | 0.45 (0.22–0.9) | |||
| High | 2 | 0.20 (0.05–0.81) | 0.020 | ||
| Node-negative patients | |||||
| UPA | |||||
| Negative | 0 | 1.00 | |||
| Low | 1 | 3.75 (0.83–16.9) | |||
| High | 2 | 14.1 (0.69–285.5) | 0.010 | ||
| TGFβ1 | |||||
| Negative | 0 | 1.00 | |||
| Low | 1 | 2.90 (1.19–7.10) | |||
| High | 2 | 8.41 (1.41–50.4) | 0.010 | ||
Candidate variables in the Cox model are listed in Results. HR, hazard ratio; CI, 95% confidence interval.
DISCUSSION
The reduced response to TGFβ in some tumour systems appears to involve multiple mechanisms, including loss of functional TGF-β receptor proteins (Grady et al, 1999; Fukai et al, 2003). In addition, mutations of downstream TGF-β signalling pathway genes have also been shown to result in a loss of responsiveness to TGF-β1 (Wang et al, 2000; Maliekal et al, 2003). In contrast to many other tumours, structural lesions of TGFβ signal transducers appear to be rare in breast cancers (Chen et al, 1998; Xie et al, 2002; Jeruss et al, 2003). This suggests that, in a number of circumstances such as cell dedifferentiation, the normal function of TGFβ1 in breast epithelial cells might be abrogated on behalf of oncogenic function.
Whereas TGFβ1 seems to be confirmed as a marker of bad prognostic in a number of human tumours such as colorectal (Tsushima et al, 1996; Picon et al, 1998) and prostatic (Ivanovic et al, 1995; Shariat et al, 2004) cancers, the impact of TGFβ1 on the progression of breast cancer remains uncertain. As for carcinomas in other organs, TGFβ1 expression is often increased locally and systemically in advanced breast cancers, particularly at the leading invasive edge of the tumour and in metastasis (Dalal et al, 1993; Walker et al, 1994; Chakravarthy et al, 1999). Nevertheless, whereas the elevated expression of TGFβ1 is described to associate with disease progression in a number of studies (Gorsch et al, 1992; Dalal et al, 1993; Walker et al, 1994), others studies failed to reveal diagnostic or predictive value of TGFβ1 for breast cancer patients (Dublin et al, 1993; Murray et al, 1993; Kesari et al, 1999; Ghellal et al, 2000).
It is notable that, except when measured in plasma, most of the authors employed semiquantitative immunohistochemical staining to evaluate TGFβ1 protein level in breast samples. In this study, we have measured for the first time the level of TGFβ1 protein in breast tumour samples by ELISA, in order to examine potent correlations with clinical features. Using this assay, we found 94.3% of patients expressing TGFβ1 with a range of 0–684 pg mg−1 protein and a median value of 86.7 pg mg−1 of protein.
In agreement with a number of previous studies, we show in the overall population that TGFβ1 was correlated only with menopausal status. Thus, a moderate but significant increase of tumoral TGFβ1 level was observed in premenopausal patients when compared to postmenopausal subgroup (P=0.0006). These data are in apparent opposition with some published studies, indicating that estradiol decreased the production of TGFβ1 by breast cancer epithelial cells in vitro (Knabbe et al, 1987; Philips and McFadden, 2004). Nevertheless, the diminution of TGFβ1 in postmenopausal patients might reflect adaptability process of tumoral cells to the profound hormonal modifications, which occur during menopause.
The increased expression of uPA has been reported to be associated with poor prognostic for patients with breast cancer (Duffy et al, 1998). Paradoxically, its inhibitor PAi-1 has also been described to contribute to the malignant phenotype of tumour cells (Look et al, 2002; Schrohl et al, 2004). Thus, PAi-1 might promote the development of tumoral angiogenesis through the stabilisation and maturation of new vessels (Bajou et al, 2004). Interestingly, TGFβ1 was also found to be positively correlated to PAi-1 in the node-negative subgroup (P=0.040). The activity of PAi-1 is tightly regulated on the transcriptional level, and TGFβ1 is the major regulator of PAi-1 expression and in turn of local PAi-1 activity (Westerhausen et al, 1991). Thus, in the node-negative population, the upregulation of PAi-1 by TGFβ1 might constitute an early event that promotes further progression of breast tumours. This is in agreement with our data indicating that TGFβ1 is an indicator of bad prognostic for breast cancer patients. Thus, multivariate analysis revealed that, after lymph node status (P=0.0002) and uPA (P=0.004), TGFβ1 was an independent prognostic marker for DFS (P=0.005) in the overall population. Furthermore, TGFβ1 remained the prominent prognostic factor in the node-negative population (P=0.010). In this late population, Kaplan–Meier curves further demonstrated that high level of TGFβ1 was correlated with a shorter disease-free survival (P=0.020). Conversely, TGFβ was not a prognostic factor for OS in the node-negative population. However, it has to be mentioned that at the cutoff date of the study, three deaths had been recorded in the node-negative subgroup. This is probably insufficient to distinguish a potential influence of TGFβ on overall survival, in this population. Whereas clinical studies in breast cancers have led to conflicting results, our data suggest that TGFβ1 has the potential to promote metastasis and recurrence for patients with breast carcinomas. It has to be noted that patients included in this study have not received modern chemotherapy, which could influence the outcomes. The fact that prognostic value of TGFβ1 was observed in node-negative population strongly suggests that TGFβ1 interferes at early stages of tumour progression, probably by making cell environment favorable for metastatic spread.
Although the lymph node status is one of the best prognostic factors in breast cancer, it is not sufficiently accurate to predict the clinical course of the disease. Indeed, 20–30% of node-negative breast cancer patients will experience disease recurrence and metastatic dissemination. Whereas numerous predictive factors have been characterised thus far, early prognostic markers that interfere at the beginning of tumour progression are scarce. The prognostic significance of high TGFβ1 level on DFS observed in node-negative breast cancer patients suggest that the determination of tumoral TGFβ1 status might help to identify a high-risk population early in tumour progression, for which a more appropriate therapy should be established. In this context, it appears fundamental to confirm the prognostic value of TGFβ in a large cohort of node-negative patients. Furthermore, as total TGFβ (active plus latent forms) has been measured in our study, it would be helpful to determine the respective role for latent and active TGFβ as prognostic markers in breast cancers.
Footnotes
Grant sponsor: Ministère Français de la Santé (1995), on behalf of P Bonnier, JM Brandone, C Bressac, JM Blanc, L Cals, C Charpin, J Del Grande, J Guidon, A Lachard, P Nouyrigat, L Piana, and M Pizzi-Anselme.
References
- Alexander MG, Moses HL (1995) Transforming growth factor β and cell cycle regulation. Cancer Res 55: 1452–1457 [PubMed] [Google Scholar]
- Bajou K, Maillard C, Jost M, Lijnen RH, Gils A, Declerck P, Carmeliet P, Foidart JM, Noel A (2004) Host-derived plasminogen activator inhibitor-1 (PAi-1) concentration is critical for in vivo tumoral angiogenesis and growth. Oncogene 23: 6986–6990 [DOI] [PubMed] [Google Scholar]
- Bottinger EP, Jakubczak JL, Haines DC, Bagnall K, Wakefield LM (1997) Transgenic mice overexpressing a dominant-negative mutant type II transforming growth factor receptor show enhanced tumorigenesis in the mammary gland and lung in response to the carcinogen 7,12-dimethylbenz-[a]-anthracene. Cancer Res 57: 5564–5570 [PubMed] [Google Scholar]
- Bristow RE, Baldwin R, Yamada SD, Korc M, Karlan BY (1999) Altered expression of transforming growth factor-beta ligands and receptors in primary and recurrent ovarian carcinoma. Cancer 85: 658–668 [DOI] [PubMed] [Google Scholar]
- Chakravarthy D, Green AR, Green VL, Kerin MJ, Speirs V (1999) Expression and secretion of TGF-beta isoforms and expression of TGF-beta-receptors I, II and III in normal and neoplastic human breast. Int J Oncol 15: 187–194 [DOI] [PubMed] [Google Scholar]
- Chen T, Carter D, Garrigue-Antar L, Reiss M (1998) Transforming growth factor-β type I receptor kinase mutant associated with metastatic breast cancer. Cancer Res 58: 4805–4810 [PubMed] [Google Scholar]
- Dalal BI, Keown PA, Greenberg AH (1993) Immunocytochemical localization of secreted transforming growth factor-beta 1 to the advancing edges of primary tumors and to lymph node matastases of human mammary carcinoma. Am J Pathol 143: 381–389 [PMC free article] [PubMed] [Google Scholar]
- Dublin EA, Barnes DM, Wang DY, King RJ, Levison DA (1993) TGF alpha and TGF beta expression in mammary carcinomas. J Pathol 170: 15–22 [DOI] [PubMed] [Google Scholar]
- Duffy MJ, Duggan C, Mulcahy HE, McDermott EW, O’Higgins NJ (1998) Urokinase plasminogen activator: a prognostic marker in breast cancer including patients with axillary node-negative disease. Clin Chem 44: 1177–1183 [PubMed] [Google Scholar]
- Foekens JA, Portengen H, van Putten WL, Trapman AM, Reubi JC, Alexieva-Figusch J, Klijn JG (1989) Prognostic value of estrogen and progesterone receptors measured by enzyme immunoassays in human breast cancer tumor cytosol. Cancer Res 49: 5823–5828 [PubMed] [Google Scholar]
- Foekens JA, Romain S, Look MP, Martin PM, Klijn JGM (2001) Thymidine kinase and thymidylate synthase in advanced breast cancer: response to tamoxifen and chemotherapy. Cancer Res 61: 1421–1425 [PubMed] [Google Scholar]
- Fukai Y, Fukuchi M, Masuda N, Osawa H, Kato H, Nakajima T, Kuwano H (2003) Reduced expression of transforming growth factor-beta receptors is an unfavorable prognostic factor in human oesophageal squamous cell carcinoma. Int J Cancer 20: 161–166 [DOI] [PubMed] [Google Scholar]
- Ghellal A, Li C, Hayes M, Byrne G, Bundred N, Kumar S (2000) Prognostic significance of TGF beta 1 and TGF beta 3 in human breast carcinoma. Anticancer Res 20: 4413–4418 [PubMed] [Google Scholar]
- Gobbi H, Arteaga CL, Jensen RA, Simpson JF, Dupont WD, Olson SJ, Schuyler PA, Plummer Jr WD, Page DL (2000) Loss of expression of transforming growth factor β type II receptor correlates with high tumour grade in human breast in-situ and invasive carcinomas. Histopathology (Oxford) 36: 168–177 [DOI] [PubMed] [Google Scholar]
- Gorsch SM, Memoli VA, Stukel TA, Golg LI, Arrick BA (1992) Immunohistochemical staining for transforming growth factor beta 1 associates with disease progression in human breast cancer. Cancer Res 52: 6949–6952 [PubMed] [Google Scholar]
- Grady WM, Myeroff LL, Swinler SE, Rajput A, Thiagalingam S, Lutterbaugh JD, Neumann A, Brattain MG, Chang J, Kim SJ, Kinzler KW, Vogelstein B, Willson JK, Markowitz S (1999) Mutational inactivation of transforming growth factor β receptor type II in microsatellite stable cancer colon. Cancer Res 59: 320–324 [PubMed] [Google Scholar]
- Ivanovic V, Melman A, Davis-Joseph B, Valvic M, Geliebter J (1995) Elevated plasma levels of TGF-beta 1 in patients with invasive prostate cancer. Nat Med 1: 282–284 [DOI] [PubMed] [Google Scholar]
- Ivanovic V, Todorovic-Rakovic N, Demajo M, Neskovic-Konstantinovic Z, Subota V, Ivanisevic-Milovanovic O, Nikolic-Vukosavljevic D (2003) Elevated plasma levels of transforming growth factor-beta 1 (TGF-beta 1) in patients with advanced breast cancer: association with disease progression. Eur J Cancer 39: 454–461 [DOI] [PubMed] [Google Scholar]
- Jeruss JS, Sturgis CD, Rademaker AW, Woodruff TK (2003) Down-regulation of activin, activin receptors, and smads in high-grade breast cancer. Cancer Res 63: 3783–3790 [PubMed] [Google Scholar]
- Jhappan C, Geiser AG, Kordon EC, Bagheri D, Hennighausen L, Roberts AB, Smith GH, Merlino G (1993) Targeting expression of a transforming growth factor β1 transgene to the pregnant mammary gland inhibits alveolar development and lactation. EMBO J 12: 1835–1845 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kesari AL, Chellam VG, Mathew BS, Nair MK, Pillai MR (1999) Transforming growth factor beta related to extent of tumo angiogenesis but not apoptosis or proliferation in breast carcinoma. Breast Cancer 25: 29–36 [DOI] [PubMed] [Google Scholar]
- Knabbe C, Lippman ME, Wakefield LM, Flanders KC, Kasid A, Derynck R, Dickson RB (1987) Evidence that transforming growth factor-beta is a hormonally regulated negative growth factor in human breast cancer cells. Cell 48: 417–428 [DOI] [PubMed] [Google Scholar]
- Kong FM, Anscher MS, Murase T, Abbott BD, Iglehart JD, Jirtle RL (1995) Elevated plasma transforming growth factor-beta 1 levels in breast cancer patients decrease after surgical removal of the tumor. Ann Surg 222: 155–162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lebrecht A, Grimm C, Euller G, Ludwig E, Ulbrich E, Lantzsch T, Hefler L, Koelbl H (2004) Transforming growth factor beta 1 serum levels in patients with preinvasive and invasive lesions of the breast. Int J Biol Markers 19: 236–239 [DOI] [PubMed] [Google Scholar]
- Letterio JJ, Roberts AB (1998) Regulation of immune responses by TGFβ. Annu Rev Immunol 16: 137–161 [DOI] [PubMed] [Google Scholar]
- Look MP, van Putten WL, Duffy MJ, Harbeck N, Christensen IJ, Thomssen C, Kates R, Spyratos F, Ferno M, Eppenberger-Castori S, Sweep CG, Ulm K, Peyrat JP, Martin PM, Magdelenat H, Brunner N, Duggan C, Lisboa BW, Bendahl PO, Quillien V, Daver A, Ricolleau G, Meijer-van Gelder ME, Manders P, Fiets WE, Blankenstein MA, Broet P, Romain S, Daxenbichler G, Windbichler G, Cufer T, Borstnar S, Kueng W, Beex LV, Klijn JG, O'Higgins N, Eppenberger U, Janicke F, Schmitt M, Foekens JA (2002) Pooled analysis of prognostic impact of urokinase-type plasminogen activator and its inhibitor PAI-1 in 8377 breast cancer patients. J Natl Cancer Inst 94: 116–128 [DOI] [PubMed] [Google Scholar]
- Maliekal TT, Antony ML, Nair A, Paulmurugan R, Karunagaran D (2003) Loss of expression and mutations of Smad2 and Smad4 in human cervical cancer. Oncogene 22: 4889–4897 [DOI] [PubMed] [Google Scholar]
- Moretti S, Pinzi C, Spallanzani A, Berti E, Chiarugi A, Mazzoli S, Fabiani M, Vallecchi C, Herlyn M (1999) Immunohistochemical evidence of cytokine networks during progression of human melanocytic lesions. Int J Cancer 20: 160–168 [DOI] [PubMed] [Google Scholar]
- Murray PA, Barrett-Lee P, Travers M, Luqmani Y, Powles T, Coombes RC (1993) The prognostic significance of transforming growth factors in human breast cancer. Br J Cancer 67: 1408–1412 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oft M, Peli J, Rudaz C, Schwarz H, Beug H, Reichmann E (1996) TGF-beta1 and Ha-Ras collaborate in modulating the phenotypic plasticity and invasiveness of epithelial tumor cells. Genes Dev 10: 2462–2477 [DOI] [PubMed] [Google Scholar]
- Oh SP, Seki T, Goss KA, Imamura T, Yi Y, Donahoe PK, Li L, Miyazono K, ten Dijke P, Kim S, Li E (2000) Activin receptor-like kinase 1 modulates transforming growth factor-β1 signaling in the regulation of angiogenesis. Proc Natl Acad Sci USA 97: 2626–2631 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perlman R, Schiemann WP, Brooks MW, Lodish HF, Weinberg RA (2001) TGF-β-induced apoptosis is mediated by the adaptor protein DAXX that facilitates JNK activation. Nat Cell Biol 13: 708–714 [DOI] [PubMed] [Google Scholar]
- Philips N, McFadden K (2004) Inhibition of transforming growth factor-beta and matrix metalloproteinases by estrogen and prolactin in breast cancer cells. Cancer Lett 206: 63–68 [DOI] [PubMed] [Google Scholar]
- Picon A, Gold LI, Wang J, Cohen A, Friedman E (1998) A subset of metastasis human colon cancers expresses elevated levels of transforming growth factor beta1. Cancer Epidemiol Biomarkers Prev 7: 497–504 [PubMed] [Google Scholar]
- Pierce Jr DF, Gorska AE, Chytil A, Meise KS, Page DL, Coffey Jr RJ, Moses HL (1995) Mammary tumor suppression by transforming growth factor β1 transgene expression. Proc Natl Acad Sci USA 92: 4254–4258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts AB, Sporn MB (1990) The transforming growth factor-βs. In Peptide Growth Factors and their Receptors, Part I Sprone MB, Roberts AB (eds). pp 419–472. Berlin: Springer-Verlag [Google Scholar]
- Romain S, Lainé-Bidron C, Martin P-M, Magdelenat H (1995) Steroid receptor distribution in 47892 breast cancers. A collaborative study of 7 European laboratories. Eur J Cancer 31A: 411–417 [DOI] [PubMed] [Google Scholar]
- Romain S, Spyratos F, Descotes F, Daver A, Rostaing-Puissant B, Bougnoux P, Colonna M, Bolla M, Martin PM (2000) Prognostic of DNA-synthesizing enzyme activities (thymidine kinase and thymidylate synthase) in 908 T1-T2, N0-N1, M0 breast cancers: a retrospective multicenter study. Int J Cancer 87: 860–868 [PubMed] [Google Scholar]
- Schrohl AS, Holten-Andersen MN, Peters HA, Look MP, Meijer-van Gelder ME, Klijn JG, Brunner N, Foekens JA (2004) Tumor tissue levels of tissue inhibitor of metalloproteinase-1 as a prognostic marker in primary breast cancer. Clin Cancer Res 10: 2289–2298 [DOI] [PubMed] [Google Scholar]
- Schwarte-Waldhoff I, Volpert OV, Bouck NP, Sipos B, Hahn SA, Klein-Scory S, Luttges J, Kloppel G, Graeven U, Eilert-Micus C, Hintelmann A, Schmiegel W (2000) Smad4/DPC4-mediated tumor suppression through suppression of angiogenesis. Proc Natl Acad Sci USA 97: 9624–9629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shariat SF, Kattan MW, Traxel E, Andrews B, Zhu K, Wheeler TM, Slawin KM (2004) Association of pre- and postoperative plasma levels of transforming growth factor β1 and interleukin 6 and its soluble receptor with prostate cancer progression. Clin Cancer Res 10: 1992–1999 [DOI] [PubMed] [Google Scholar]
- Strange R, Li F, Saurer S, Burkhardt A, Friis RR (1992) Apoptotic cell death and tissue remodelling during mouse mammary gland involution. Development 115: 49–58 [DOI] [PubMed] [Google Scholar]
- Tsushima H, Kawata S, Tamura S, Ito N, Shirai Y, Kiso S, Imai Y, Shimomukai H, Nomura Y, Matsuda Y, Matsuzawa Y (1996) High levels of transforming growth factor beta 1 in patients with colorectal cancer: association with disease progression. Gastroenterology 110: 375–382 [DOI] [PubMed] [Google Scholar]
- Wakefield LM, Letterio JJ, Chen T, Danielpour D, Allison RS, Pai LH, Denicoff AM, Noone MH, Cowan KH, O'Shaughnessy JA (1995) Transforming growth factor-beta1 circulates in normal human plasma and is unchanged in advanced metastatic breast cancer. Clin Cancer Res 1: 129–136 [PubMed] [Google Scholar]
- Wakefield LM, Piek E, Bottinger EP (2000) TGF-β signaling in mammary gland development and tumorigenesis. J Mammary Gland Biol Neoplasia 6: 67–82 [DOI] [PubMed] [Google Scholar]
- Walker RA, Dearing SJ, Gallacher B (1994) Relationship of transforming growth factor beta 1 to extracellular matrix and stromal infiltrates in invasive breast carcinoma. Br J Cancer 69: 1160–1165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D, Kanuma T, Mizunuma H, Takama F, Ibuki Y, Wake N, Mogi A, Shitara Y, Takenoshita S (2000) Analysis of specific gene mutations in the transforming growth factor-β signal transduction pathway in human ovarian cancer. Cancer Res 60: 4507–4512 [PubMed] [Google Scholar]
- Westerhausen Jr DR, Hopkins WE, Billadello JJ (1991) Multiple transforming growth factor-beta-inducible elements regulate expression of the plasminogen activator inhibitor type-1 gene in Hep G2 cells. J Biol Chem 266: 1092–1100 [PubMed] [Google Scholar]
- Xie W, Mertens JC, Reiss DJ, Rimm DL, Camp RL, Haffty BG, Reiss M (2002) Alterations of smad signaling in human breast carcinoma are associated with poor outcome. A tissue microarray study. Cancer Res 62: 497–505 [PubMed] [Google Scholar]