Abstract
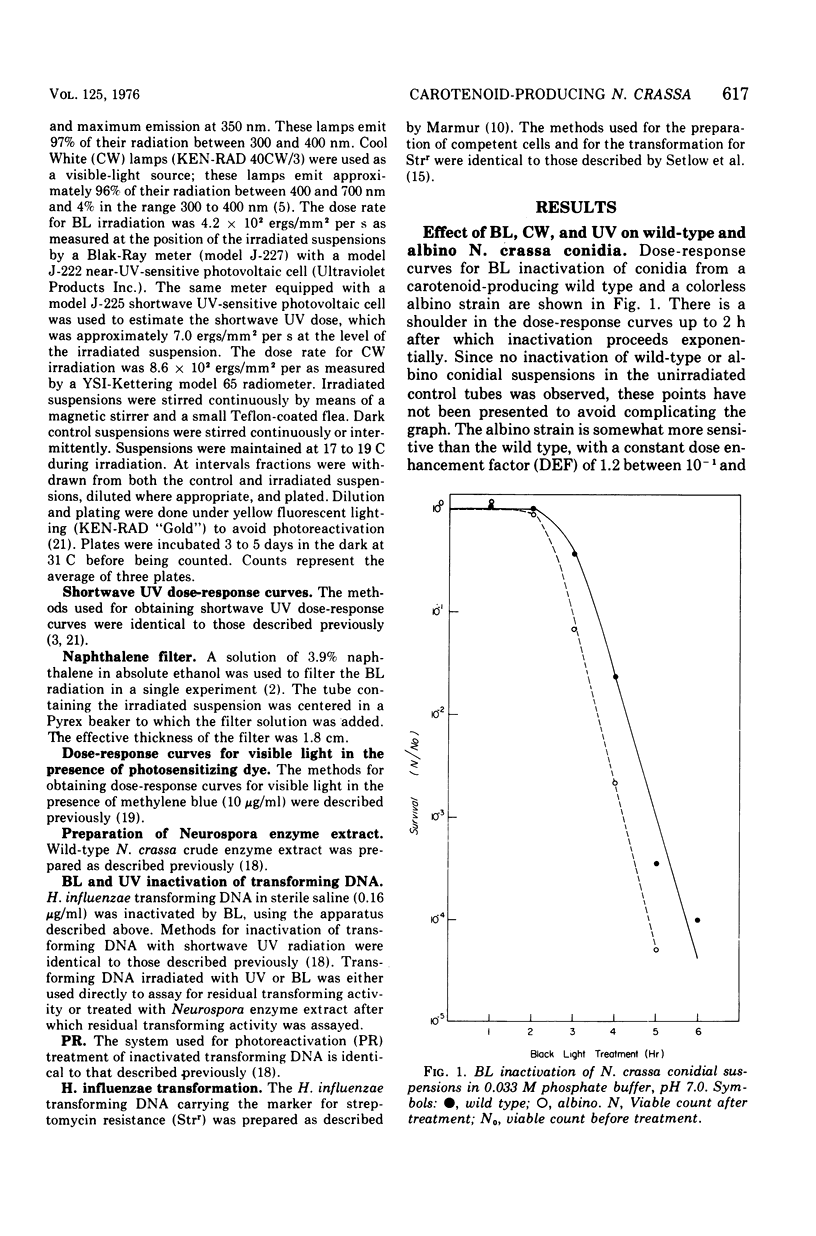
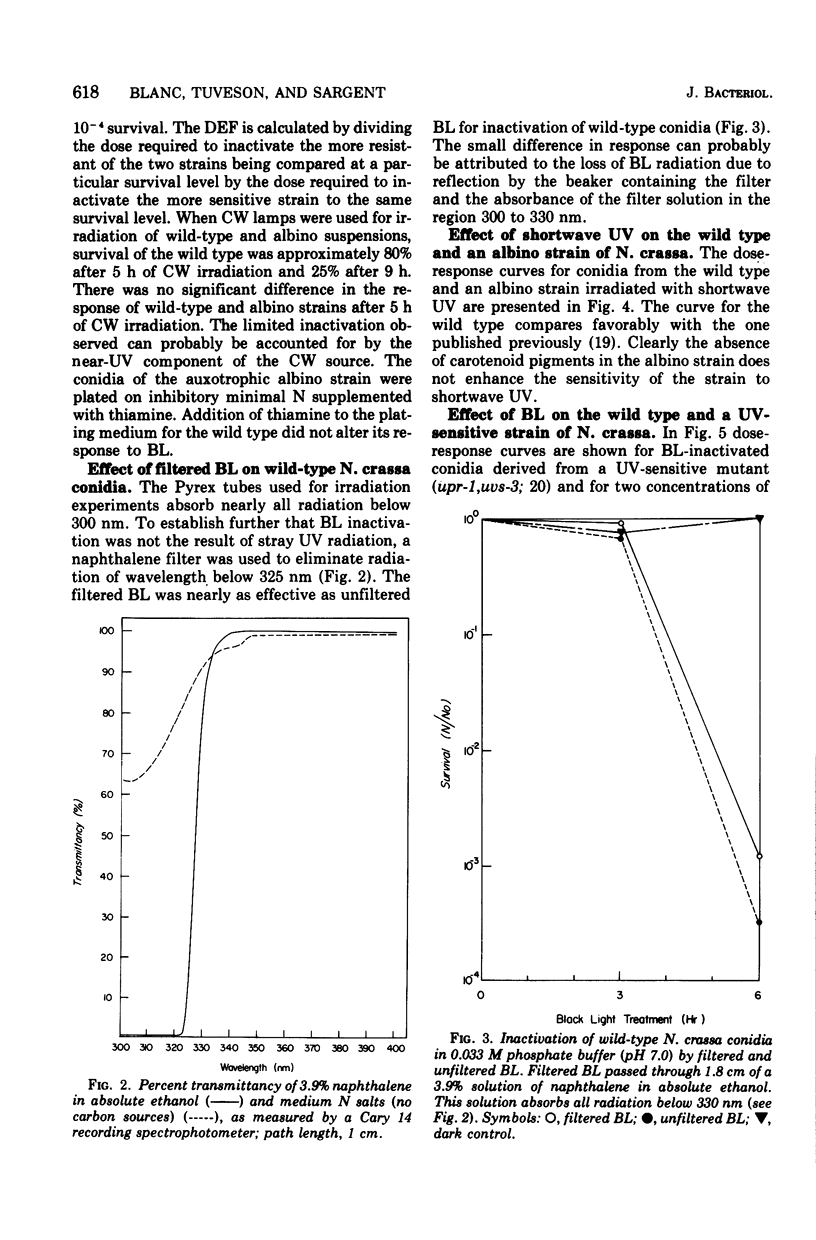
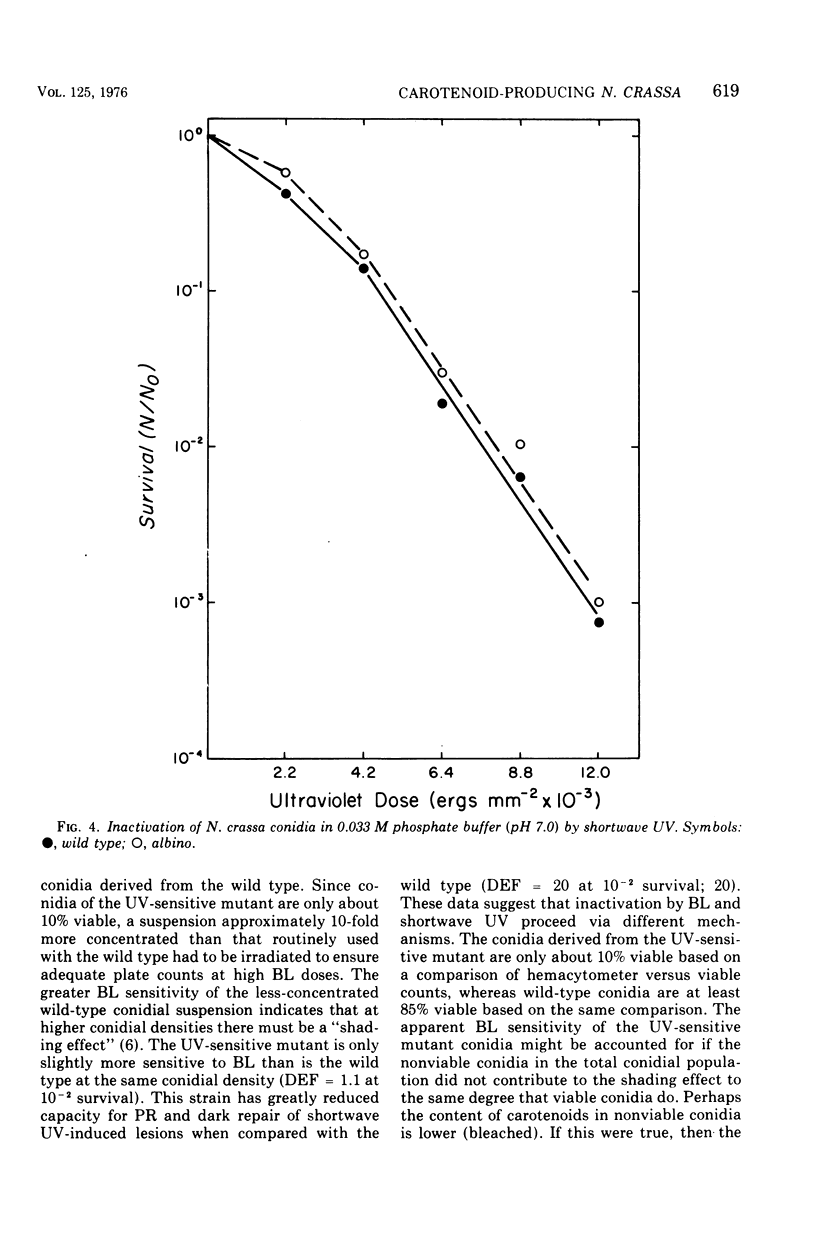
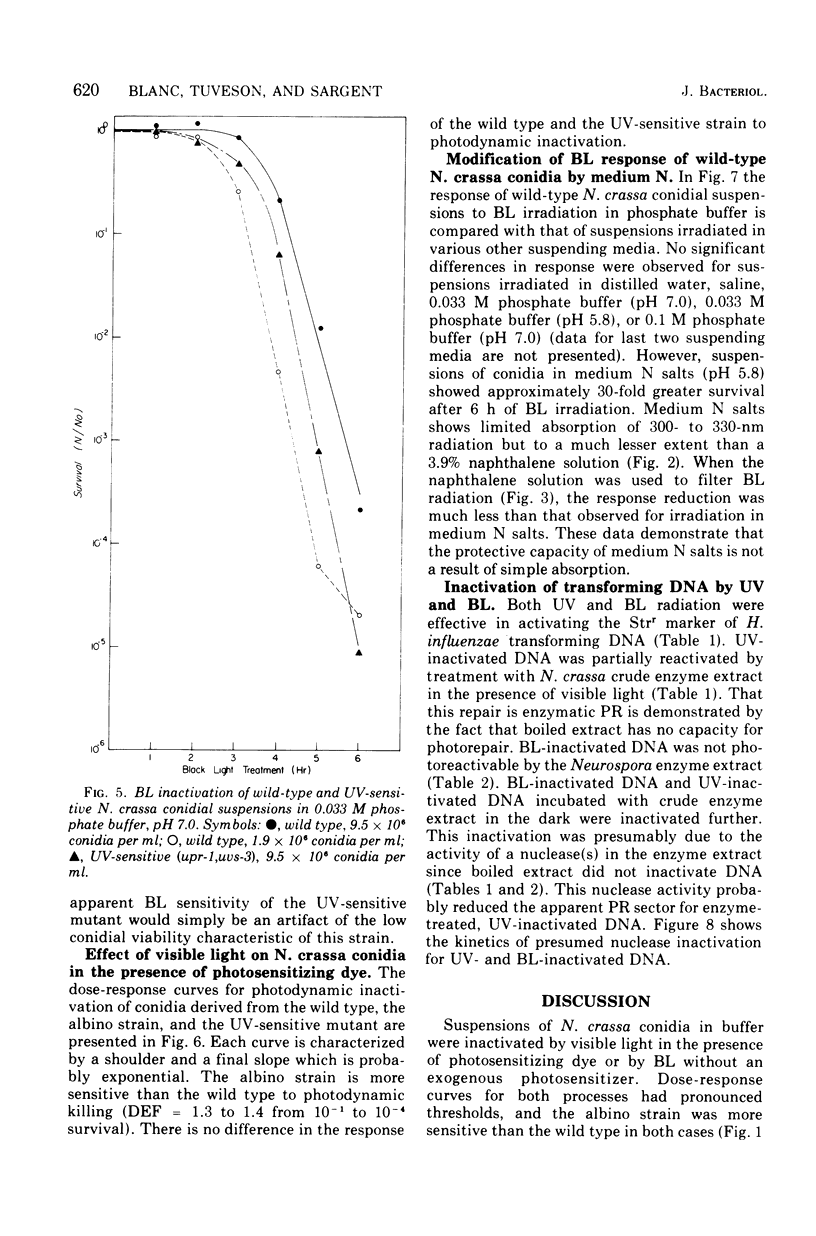
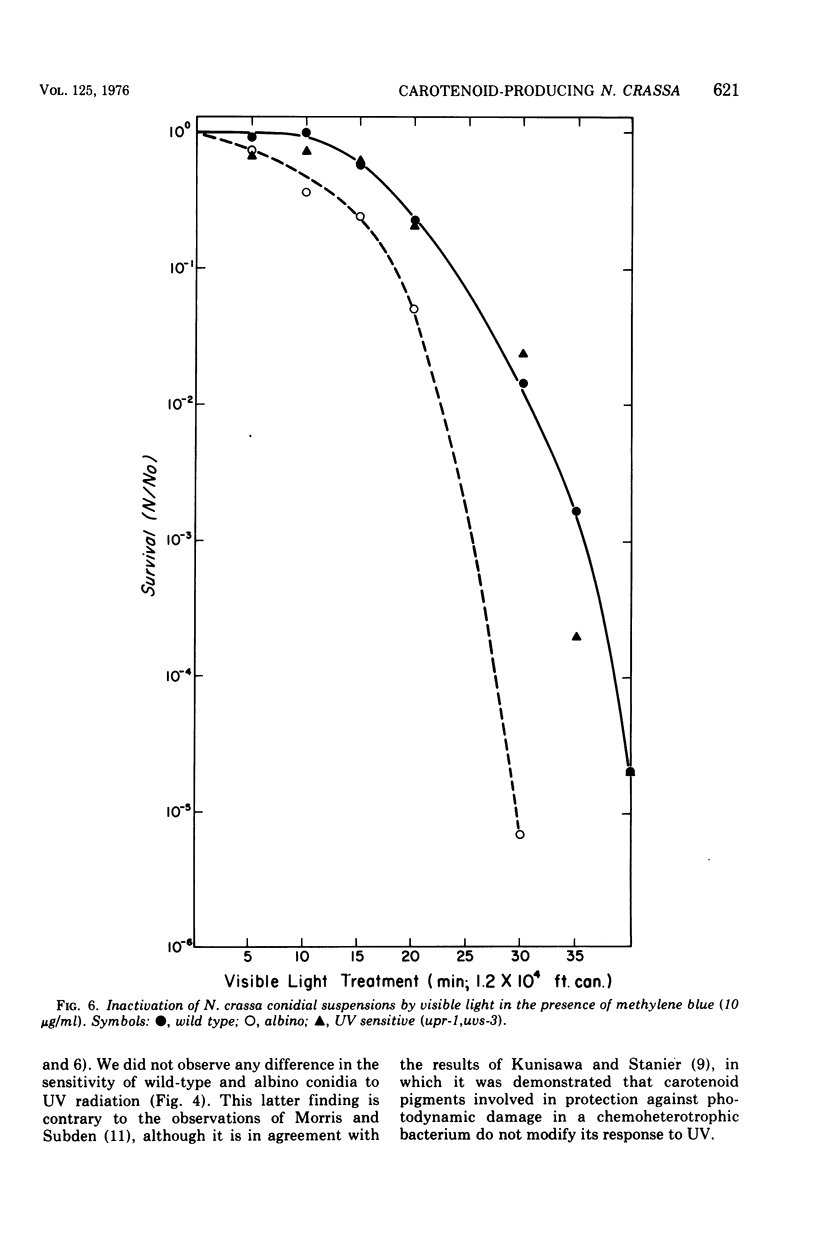
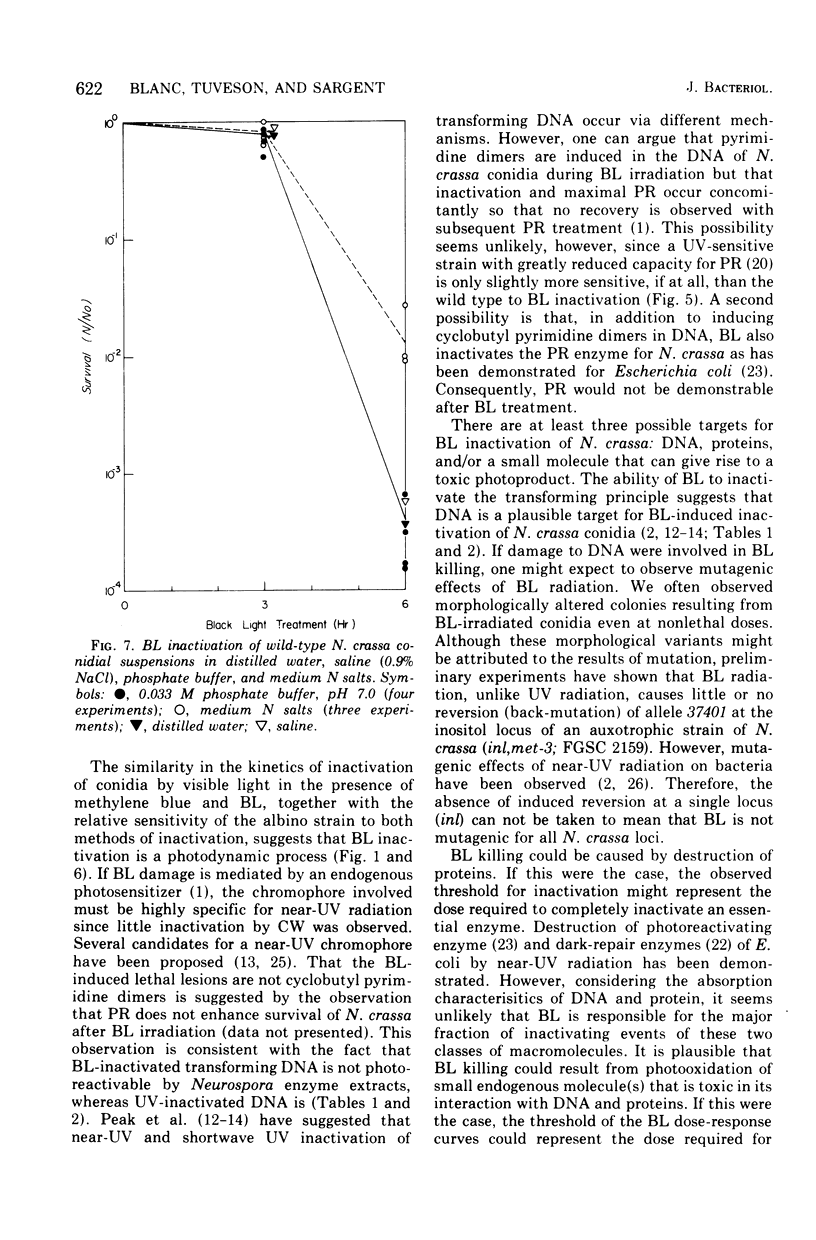
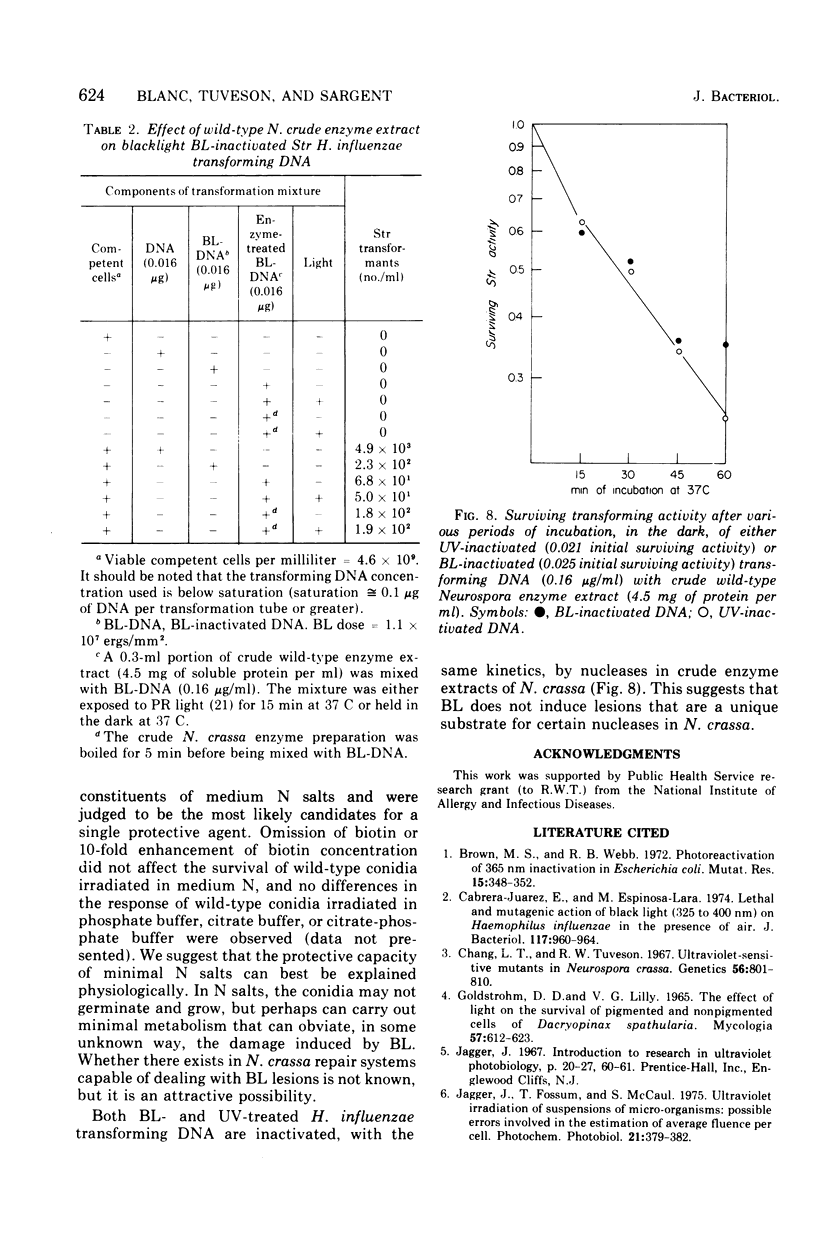
Suspensions of Neurospora crassa conidia were inactivated by blacklight (BL) radiation (300 to 425 nm) in the absence of exogenous photosensitizing compounds. Carotenoid-containing wild-type conidia were less sensitive to BL radiation than albino conidia, showing a dose enhancement factor (DEF) of 1.2 for dose levels resulting in less than 10% survival. The same strains were about equally sensitive to shortwave ultraviolet (UV) inactivation. The kinetics of BL inactivation are similar to those of photodynamic inactivation by visible light in the presence of a photosensitizing dye (methylene blue). Only limited inactivation by visible light in the absence of exogenous photosensitizers was observed. BL and UV inactivations are probably caused by different mechanisms since wild-type conidia are only slightly more resistant to BL radiation (DEF = 1.2 at 1.0% survival) than are conidia from a UV-sensitive strain (upr-1, uvs-3). The BL-induced lethal lesions are probably no cyclobutyl pyrimidine dimers since BL-inactivated Haemophilus influenzae transforming deoxyribonucleic acid is not photoreactivated by N. crassa wild-type enzyme extracts, whereas UV-inactivated transforming deoxyribonucleic acid is photoreactivable with this treatment.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brown M. S., Webb R. B. Photoreactivation of 365 nm inactivation in Escherichia coli. Mutat Res. 1972 Jul;15(3):348–352. doi: 10.1016/0027-5107(72)90080-2. [DOI] [PubMed] [Google Scholar]
- Cabrera-Juarez E., Espinosa-Lara M. Lethal and mutagenic action of black light (325 to 400 nm) on Haemophilus influenzae in the presence of air. J Bacteriol. 1974 Mar;117(3):960–964. doi: 10.1128/jb.117.3.960-964.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang L. T., Tuveson R. W. Ultraviolet-sensitive mutants in Neurospora crassa. Genetics. 1967 Aug;56(4):801–810. doi: 10.1093/genetics/56.4.801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jagger J., Fossum T., McCaul S. Ultraviolet irradiation of suspensions of micro-organisms: possible errors involved in the estimation of average fluence per cell. Photochem Photobiol. 1975 May;21(5):379–382. doi: 10.1111/j.1751-1097.1975.tb06690.x. [DOI] [PubMed] [Google Scholar]
- KAPLAN R. W. Dose-effect curves of s-mutation and killing in Serratia marcescens. Arch Mikrobiol. 1956;24(1):60–79. doi: 10.1007/BF00418422. [DOI] [PubMed] [Google Scholar]
- Morris S. A., Subden R. E. Effects of ultraviolet radiation on carotenoid-containing and albino strains of Neurospora crassa. Mutat Res. 1974 Feb;22(2):105–109. doi: 10.1016/0027-5107(74)90089-x. [DOI] [PubMed] [Google Scholar]
- Peak M. J., Peak J. G., Webb R. B. Inactivation of transforming DNA by ultraviolet light. 3. Further observations on the effects of 365 nm radiation. Mutat Res. 1973 Nov;20(2):143–148. doi: 10.1016/0027-5107(73)90184-x. [DOI] [PubMed] [Google Scholar]
- Peak M. J., Peak J. G., Webb R. B. Inactivation of transforming DNA by ultraviolet light. I. Near-UV action spectrum for marker inactivation. Mutat Res. 1973 Nov;20(2):129–135. doi: 10.1016/0027-5107(73)90182-6. [DOI] [PubMed] [Google Scholar]
- Setlow J. K., Brown D. C., Boling M. E., Mattingly A., Gordon M. P. Repair of deoxyribonucleic acid in Haemophilus influenzae. I. X-ray sensitivity of ultraviolet-sensitive mutants and their behavior as hosts to ultraviolet-irradiated bacteriophage and transforming deoxyribonucleic acid. J Bacteriol. 1968 Feb;95(2):546–558. doi: 10.1128/jb.95.2.546-558.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stoien J. D., Wang R. J. Effect of near-ultraviolet and visible light on mammalian cells in culture II. Formation of toxic photoproducts in tissue culture medium by blacklight. Proc Natl Acad Sci U S A. 1974 Oct;71(10):3961–3965. doi: 10.1073/pnas.71.10.3961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tuveson R. W. Comparison of two transformation systems for the assay of the Neurospora photoreactivating enzyme. Genet Res. 1972 Aug;20(1):9–18. doi: 10.1017/s0016672300013550. [DOI] [PubMed] [Google Scholar]
- Tuveson R. W. Genetic and enzymatic analysis of a gene controlling UV sensitivity in Neurospora crassa. Mutat Res. 1972 Aug;15(4):411–424. doi: 10.1016/0027-5107(72)90005-x. [DOI] [PubMed] [Google Scholar]
- Tuveson R. W. Interaction of genes controlling ultraviolet sensitivity in Neurospora crassa. J Bacteriol. 1972 Oct;112(1):632–634. doi: 10.1128/jb.112.1.632-634.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyrrell R. M., Webb R. B., Brown M. S. Destruction of photoreactivating enzyme by 365 nm radiation. Photochem Photobiol. 1973 Oct;18(4):249–254. doi: 10.1111/j.1751-1097.1973.tb06420.x. [DOI] [PubMed] [Google Scholar]
- Tyrrell R. M., Webb R. B. Reduced dimer excision in bacteria following near ultraviolet (365 nm) radiation. Mutat Res. 1973 Sep;19(3):361–364. doi: 10.1016/0027-5107(73)90238-8. [DOI] [PubMed] [Google Scholar]
- Webb R. B., Lorenz J. R. Toxicity of irradiated medium for repair-deficient strains of Escherichia coli. J Bacteriol. 1972 Oct;112(1):649–652. doi: 10.1128/jb.112.1.649-652.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webb R. B., Malina M. M. Mutagenic effects of near ultraviolet and visible radiant energy on continuous cultures of escherichia coli. Photochem Photobiol. 1970 Dec;12(6):457–468. doi: 10.1111/j.1751-1097.1970.tb06078.x. [DOI] [PubMed] [Google Scholar]
- Yoakum G. H. Tryptophan photoproduct(s): sensitized induction of strand breaks (or alkali-labile bonds) in bacterial deoxyribonucleic acid during near-ultraviolet irradiation. J Bacteriol. 1975 Apr;122(1):199–205. doi: 10.1128/jb.122.1.199-205.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]



