Abstract
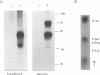
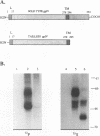
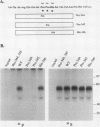
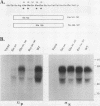
Varicella-zoster virus (VZV) glycoprotein gpIV, to be renamed VZV gI, forms a heterodimer with glycoprotein gpI (gE) which functions as an Fc receptor in virus-infected cells. Like VZV gpI (gE), this viral glycoprotein is phosphorylated in cell culture during biosynthesis. In this report, we investigated the nature and specificity of the phosphorylation event involving VZV gpIV (gI). Phosphoamino acid analysis indicated that gpIV (gI) was modified mainly on serine residues. To identify the precise location of the phosphorylation site on the 64-kDa protein, a step-by-step mutagenesis procedures was followed. Initially a tailless mutant was generated, and this truncated product was no longer phosphorylated. Thereafter, point mutations were made within the cytoplasmic tail of gpIV (gI) at potential phosphorylation sites. The phosphorylation site was localized to the following sequence: Ser-Pro-Pro (amino acids 343 to 345). Examination of the point mutants established that serine 343 in the cytoplasmic tail was the major phosphoacceptor. In addition, we found that the prolines located immediately to the C terminus of serine 343 were an integral part of the kinase recognition sequence. This site was located immediately N terminal to a predicted beta-turn secondary structure. By comparison with known substrate consensus sequences for various protein kinases, these data suggested that the phosphorylation of VZV gpIV (gI) was catalyzed by a proline-directed protein kinase. Computer homology analysis of other alphaherpesviruses demonstrated that a similar potential phosphorylation site was highly conserved in the cytoplasmic tails of herpes simplex virus type 1 gI, equine herpesvirus type 1 gI, and pseudorabies virus gp63.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chou P. Y., Fasman G. D. Prediction of beta-turns. Biophys J. 1979 Jun;26(3):367–383. doi: 10.1016/S0006-3495(79)85259-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark-Lewis I., Sanghera J. S., Pelech S. L. Definition of a consensus sequence for peptide substrate recognition by p44mpk, the meiosis-activated myelin basic protein kinase. J Biol Chem. 1991 Aug 15;266(23):15180–15184. [PubMed] [Google Scholar]
- Davison A. J., Edson C. M., Ellis R. W., Forghani B., Gilden D., Grose C., Keller P. M., Vafai A., Wroblewska Z., Yamanishi K. New common nomenclature for glycoprotein genes of varicella-zoster virus and their glycosylated products. J Virol. 1986 Mar;57(3):1195–1197. doi: 10.1128/jvi.57.3.1195-1197.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davison A. J., Scott J. E. The complete DNA sequence of varicella-zoster virus. J Gen Virol. 1986 Sep;67(Pt 9):1759–1816. doi: 10.1099/0022-1317-67-9-1759. [DOI] [PubMed] [Google Scholar]
- Davison A. J., Waters D. J., Edson C. M. Identification of the products of a varicella-zoster virus glycoprotein gene. J Gen Virol. 1985 Oct;66(Pt 10):2237–2242. doi: 10.1099/0022-1317-66-10-2237. [DOI] [PubMed] [Google Scholar]
- Edson C. M. Phosphorylation of neurotropic alphaherpesvirus envelope glycoproteins: herpes simplex virus type 2 gE2 and pseudorabies virus gI. Virology. 1993 Jul;195(1):268–270. doi: 10.1006/viro.1993.1372. [DOI] [PubMed] [Google Scholar]
- Forghani B., Ni L., Grose C. Neutralization epitope of the varicella-zoster virus gH:gL glycoprotein complex. Virology. 1994 Mar;199(2):458–462. doi: 10.1006/viro.1994.1145. [DOI] [PubMed] [Google Scholar]
- Fuerst T. R., Niles E. G., Studier F. W., Moss B. Eukaryotic transient-expression system based on recombinant vaccinia virus that synthesizes bacteriophage T7 RNA polymerase. Proc Natl Acad Sci U S A. 1986 Nov;83(21):8122–8126. doi: 10.1073/pnas.83.21.8122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez F. A., Raden D. L., Davis R. J. Identification of substrate recognition determinants for human ERK1 and ERK2 protein kinases. J Biol Chem. 1991 Nov 25;266(33):22159–22163. [PubMed] [Google Scholar]
- Grose C. Glycoproteins encoded by varicella-zoster virus: biosynthesis, phosphorylation, and intracellular trafficking. Annu Rev Microbiol. 1990;44:59–80. doi: 10.1146/annurev.mi.44.100190.000423. [DOI] [PubMed] [Google Scholar]
- Grose C., Jackson W., Traugh J. A. Phosphorylation of varicella-zoster virus glycoprotein gpI by mammalian casein kinase II and casein kinase I. J Virol. 1989 Sep;63(9):3912–3918. doi: 10.1128/jvi.63.9.3912-3918.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grose C. The synthesis of glycoproteins in human melanoma cells infected with varicella-zoster virus. Virology. 1980 Feb;101(1):1–9. doi: 10.1016/0042-6822(80)90478-x. [DOI] [PubMed] [Google Scholar]
- Hall F. L., Braun R. K., Mihara K., Fung Y. K., Berndt N., Carbonaro-Hall D. A., Vulliet P. R. Characterization of the cytoplasmic proline-directed protein kinase in proliferative cells and tissues as a heterodimer comprised of p34cdc2 and p58cyclin A. J Biol Chem. 1991 Sep 15;266(26):17430–17440. [PubMed] [Google Scholar]
- Hall F. L., Mitchell J. P., Vulliet P. R. Phosphorylation of synapsin I at a novel site by proline-directed protein kinase. J Biol Chem. 1990 Apr 25;265(12):6944–6948. [PubMed] [Google Scholar]
- Kemp B. E., Pearson R. B. Protein kinase recognition sequence motifs. Trends Biochem Sci. 1990 Sep;15(9):342–346. doi: 10.1016/0968-0004(90)90073-k. [DOI] [PubMed] [Google Scholar]
- Liang W., Johnson J. P. Rapid plasmid insert amplification with polymerase chain reaction. Nucleic Acids Res. 1988 Apr 25;16(8):3579–3579. doi: 10.1093/nar/16.8.3579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Litwin V., Jackson W., Grose C. Receptor properties of two varicella-zoster virus glycoproteins, gpI and gpIV, homologous to herpes simplex virus gE and gI. J Virol. 1992 Jun;66(6):3643–3651. doi: 10.1128/jvi.66.6.3643-3651.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Litwin V., Sandor M., Grose C. Cell surface expression of the varicella-zoster virus glycoproteins and Fc receptor. Virology. 1990 Sep;178(1):263–272. doi: 10.1016/0042-6822(90)90402-d. [DOI] [PubMed] [Google Scholar]
- McVey D., Brizuela L., Mohr I., Marshak D. R., Gluzman Y., Beach D. Phosphorylation of large tumour antigen by cdc2 stimulates SV40 DNA replication. Nature. 1989 Oct 12;341(6242):503–507. doi: 10.1038/341503a0. [DOI] [PubMed] [Google Scholar]
- Meyerson M., Enders G. H., Wu C. L., Su L. K., Gorka C., Nelson C., Harlow E., Tsai L. H. A family of human cdc2-related protein kinases. EMBO J. 1992 Aug;11(8):2909–2917. doi: 10.1002/j.1460-2075.1992.tb05360.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montalvo E. A., Parmley R. T., Grose C. Structural analysis of the varicella-zoster virus gp98-gp62 complex: posttranslational addition of N-linked and O-linked oligosaccharide moieties. J Virol. 1985 Mar;53(3):761–770. doi: 10.1128/jvi.53.3.761-770.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moreno S., Nurse P. Substrates for p34cdc2: in vivo veritas? Cell. 1990 May 18;61(4):549–551. doi: 10.1016/0092-8674(90)90463-o. [DOI] [PubMed] [Google Scholar]
- Moss B., Elroy-Stein O., Mizukami T., Alexander W. A., Fuerst T. R. Product review. New mammalian expression vectors. Nature. 1990 Nov 1;348(6296):91–92. doi: 10.1038/348091a0. [DOI] [PubMed] [Google Scholar]
- Mukhopadhyay N. K., Price D. J., Kyriakis J. M., Pelech S., Sanghera J., Avruch J. An array of insulin-activated, proline-directed serine/threonine protein kinases phosphorylate the p70 S6 kinase. J Biol Chem. 1992 Feb 15;267(5):3325–3335. [PubMed] [Google Scholar]
- Pines J. Cyclins and cyclin-dependent kinases: take your partners. Trends Biochem Sci. 1993 Jun;18(6):195–197. doi: 10.1016/0968-0004(93)90185-p. [DOI] [PubMed] [Google Scholar]
- Pinna L. A., Donella-Deana A., Meggio F. Structural features determining the site specificity of a rat liver cAMP-independent protein kinase. Biochem Biophys Res Commun. 1979 Mar 15;87(1):114–120. doi: 10.1016/0006-291x(79)91654-1. [DOI] [PubMed] [Google Scholar]
- Small D., Chou P. Y., Fasman G. D. Occurrence of phosphorylated residues in predicted beta-turns: implications for beta-turn participation in control mechanisms. Biochem Biophys Res Commun. 1977 Nov 7;79(1):341–346. doi: 10.1016/0006-291x(77)90101-2. [DOI] [PubMed] [Google Scholar]
- Tuazon P. T., Traugh J. A. Casein kinase I and II--multipotential serine protein kinases: structure, function, and regulation. Adv Second Messenger Phosphoprotein Res. 1991;23:123–164. [PubMed] [Google Scholar]
- Vulliet P. R., Hall F. L., Mitchell J. P., Hardie D. G. Identification of a novel proline-directed serine/threonine protein kinase in rat pheochromocytoma. J Biol Chem. 1989 Sep 25;264(27):16292–16298. [PubMed] [Google Scholar]
- Yao Z., Jackson W., Forghani B., Grose C. Varicella-zoster virus glycoprotein gpI/gpIV receptor: expression, complex formation, and antigenicity within the vaccinia virus-T7 RNA polymerase transfection system. J Virol. 1993 Jan;67(1):305–314. doi: 10.1128/jvi.67.1.305-314.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yao Z., Jackson W., Grose C. Identification of the phosphorylation sequence in the cytoplasmic tail of the varicella-zoster virus Fc receptor glycoprotein gpI. J Virol. 1993 Aug;67(8):4464–4473. doi: 10.1128/jvi.67.8.4464-4473.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yao Z., Jones D. H., Grose C. Site-directed mutagenesis of herpesvirus glycoprotein phosphorylation sites by recombination polymerase chain reaction. PCR Methods Appl. 1992 Feb;1(3):205–207. doi: 10.1101/gr.1.3.205. [DOI] [PubMed] [Google Scholar]