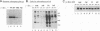Abstract
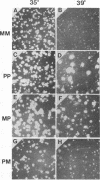
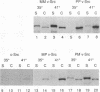
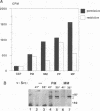
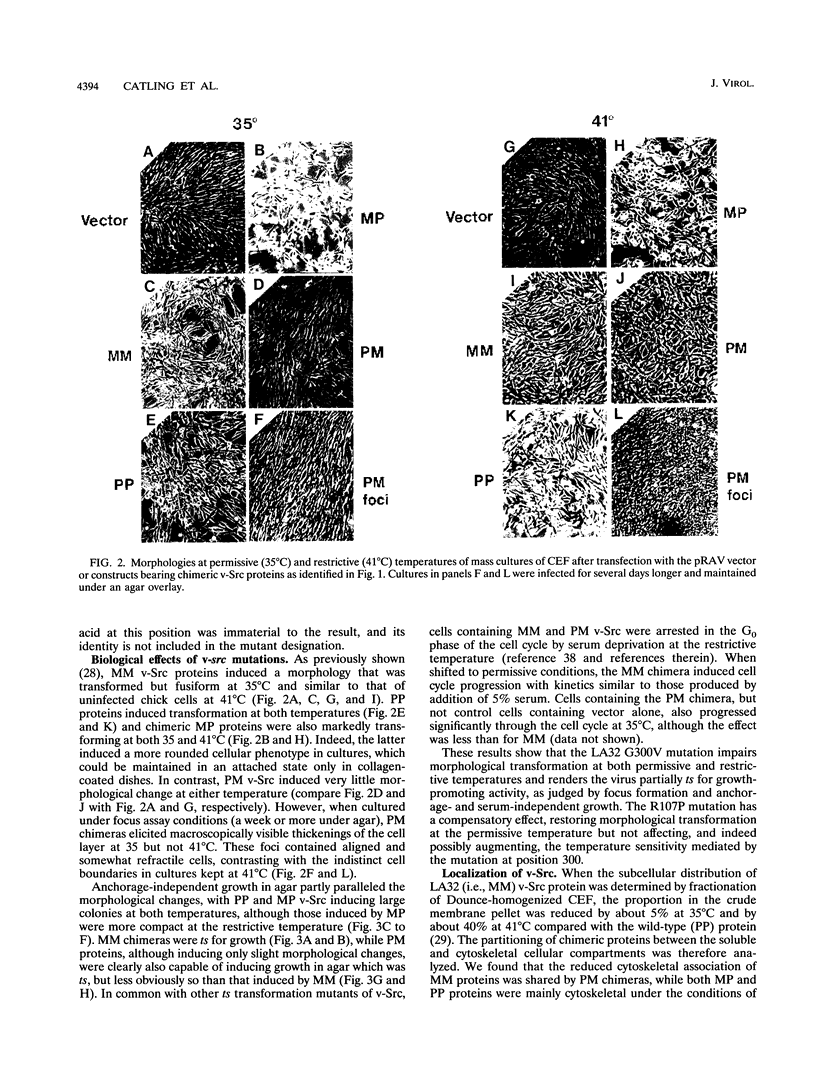
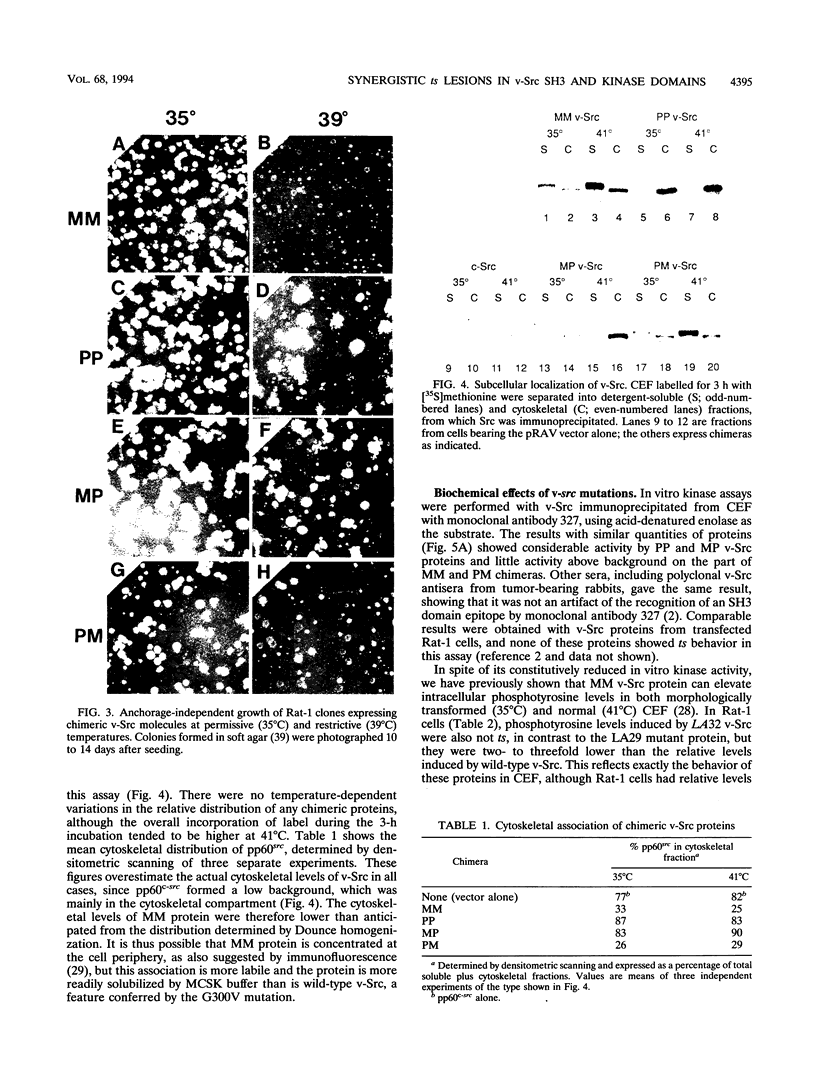
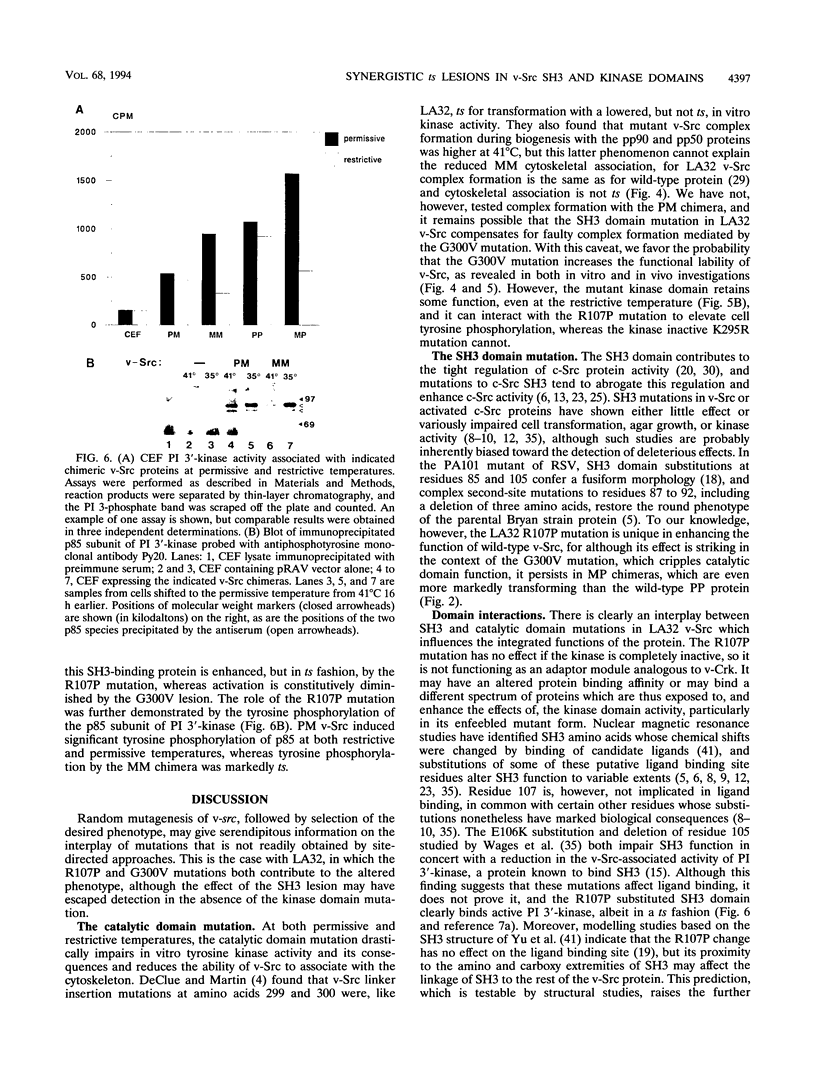
We have analyzed two functionally significant amino acid alterations encoded by the temperature-sensitive (ts) v-src mutant of Rous sarcoma virus, LA32. The G-to-V change at residue 300 in the catalytic domain nonconditionally impairs morphological transformation, in vitro kinase activity, in vivo tyrosine phosphorylation, and the cytoskeletal association of v-Src while rendering anchorage- and serum-independent growth ts. The R-to-P mutation in the SH3 domain subtly enhances morphological transformation but has no phenotype if the catalytic domain is inactivated. In the presence of the G-300-to-V mutation, this SH3 domain lesion does not affect v-Src in vitro kinase activity and cytoskeletal association, but it nonconditionally enhances cellular tyrosine phosphorylation and restores morphological transformation at the permissive temperature only. This ability to induce a ts transformed morphology, in concert with nonconditional elevations of cellular phosphotyrosine, suggest that a subset of v-Src targets that are crucial to transformation may be affected in ts fashion by the SH3 mutation. Consistent with this, we find that the R-107-to-P mutation confers ts activity and tyrosine phosphorylation on the SH3-binding enzyme phosphatidylinositol 3'-kinase. Thus, both the SH3 and catalytic domain mutations in LA32 have some ts attributes and they cooperate in determining the mutant's behavior. The ts SH3 mutation is unique and offers the potential for deeper understanding of the function of this domain.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Birge R. B., Hanafusa H. Closing in on SH2 specificity. Science. 1993 Dec 3;262(5139):1522–1524. doi: 10.1126/science.7504323. [DOI] [PubMed] [Google Scholar]
- Cooper J. A., Sefton B. M., Hunter T. Detection and quantification of phosphotyrosine in proteins. Methods Enzymol. 1983;99:387–402. doi: 10.1016/0076-6879(83)99075-4. [DOI] [PubMed] [Google Scholar]
- DeClue J. E., Martin G. S. Linker insertion-deletion mutagenesis of the v-src gene: isolation of host- and temperature-dependent mutants. J Virol. 1989 Feb;63(2):542–554. doi: 10.1128/jvi.63.2.542-554.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dezélée P., Barnier J. V., Hampe A., Laugier D., Marx M., Galibert F., Calothy G. Small deletion in v-src SH3 domain of a transformation defective mutant of Rous sarcoma virus restores wild type transforming properties. Virology. 1992 Aug;189(2):556–567. doi: 10.1016/0042-6822(92)90579-e. [DOI] [PubMed] [Google Scholar]
- Espino P. C., Harvey R., Schweickhardt R. L., White G. A., Smith A. E., Cheng S. H. The amino-terminal region of pp60c-src has a modulatory role and contains multiple sites of tyrosine phosphorylation. Oncogene. 1990 Mar;5(3):283–293. [PubMed] [Google Scholar]
- Fincham V. J., Wyke J. A. Localization of temperature-sensitive transformation mutations and back mutations in the Rous sarcoma virus src gene. J Virol. 1986 May;58(2):694–699. doi: 10.1128/jvi.58.2.694-699.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirai H., Varmus H. E. Mutations in src homology regions 2 and 3 of activated chicken c-src that result in preferential transformation of mouse or chicken cells. Proc Natl Acad Sci U S A. 1990 Nov;87(21):8592–8596. doi: 10.1073/pnas.87.21.8592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirai H., Varmus H. E. Site-directed mutagenesis of the SH2- and SH3-coding domains of c-src produces varied phenotypes, including oncogenic activation of p60c-src. Mol Cell Biol. 1990 Apr;10(4):1307–1318. doi: 10.1128/mcb.10.4.1307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jove R., Mayer B. J., Iba H., Laugier D., Poirier F., Calothy G., Hanafusa T., Hanafusa H. Genetic analysis of p60v-src domains involved in the induction of different cell transformation parameters. J Virol. 1986 Dec;60(3):840–848. doi: 10.1128/jvi.60.3.840-848.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamps M. P., Sefton B. M. Neither arginine nor histidine can carry out the function of lysine-295 in the ATP-binding site of p60src. Mol Cell Biol. 1986 Mar;6(3):751–757. doi: 10.1128/mcb.6.3.751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanner S. B., Reynolds A. B., Wang H. C., Vines R. R., Parsons J. T. The SH2 and SH3 domains of pp60src direct stable association with tyrosine phosphorylated proteins p130 and p110. EMBO J. 1991 Jul;10(7):1689–1698. doi: 10.1002/j.1460-2075.1991.tb07693.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato J. Y., Takeya T., Grandori C., Iba H., Levy J. B., Hanafusa H. Amino acid substitutions sufficient to convert the nontransforming p60c-src protein to a transforming protein. Mol Cell Biol. 1986 Dec;6(12):4155–4160. doi: 10.1128/mcb.6.12.4155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kellie S., Patel B., Wigglesworth N. M., Critchley D. R., Wyke J. A. The use of Rous sarcoma virus transformation mutants with differing tyrosine kinase activities to study the relationships between vinculin phosphorylation, pp60v-src location and adhesion plaque integrity. Exp Cell Res. 1986 Jul;165(1):216–228. doi: 10.1016/0014-4827(86)90546-x. [DOI] [PubMed] [Google Scholar]
- Liu X., Marengere L. E., Koch C. A., Pawson T. The v-Src SH3 domain binds phosphatidylinositol 3'-kinase. Mol Cell Biol. 1993 Sep;13(9):5225–5232. doi: 10.1128/mcb.13.9.5225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayer B. J., Baltimore D. Signalling through SH2 and SH3 domains. Trends Cell Biol. 1993 Jan;3(1):8–13. doi: 10.1016/0962-8924(93)90194-6. [DOI] [PubMed] [Google Scholar]
- Mayer B. J., Hanafusa H. Association of the v-crk oncogene product with phosphotyrosine-containing proteins and protein kinase activity. Proc Natl Acad Sci U S A. 1990 Apr;87(7):2638–2642. doi: 10.1073/pnas.87.7.2638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayer B. J., Jove R., Krane J. F., Poirier F., Calothy G., Hanafusa H. Genetic lesions involved in temperature sensitivity of the src gene products of four Rous sarcoma virus mutants. J Virol. 1986 Dec;60(3):858–867. doi: 10.1128/jvi.60.3.858-867.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okada M., Howell B. W., Broome M. A., Cooper J. A. Deletion of the SH3 domain of Src interferes with regulation by the phosphorylated carboxyl-terminal tyrosine. J Biol Chem. 1993 Aug 25;268(24):18070–18075. [PubMed] [Google Scholar]
- Pawson T., Gish G. D. SH2 and SH3 domains: from structure to function. Cell. 1992 Oct 30;71(3):359–362. doi: 10.1016/0092-8674(92)90504-6. [DOI] [PubMed] [Google Scholar]
- Pawson T., Schlessingert J. SH2 and SH3 domains. Curr Biol. 1993 Jul 1;3(7):434–442. doi: 10.1016/0960-9822(93)90350-w. [DOI] [PubMed] [Google Scholar]
- Potts W. M., Reynolds A. B., Lansing T. J., Parsons J. T. Activation of pp60c-src transforming potential by mutations altering the structure of an amino terminal domain containing residues 90-95. Oncogene Res. 1988;3(4):343–355. [PubMed] [Google Scholar]
- Seidel-Dugan C., Meyer B. E., Thomas S. M., Brugge J. S. Effects of SH2 and SH3 deletions on the functional activities of wild-type and transforming variants of c-Src. Mol Cell Biol. 1992 Apr;12(4):1835–1845. doi: 10.1128/mcb.12.4.1835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snyder M. A., Bishop J. M., McGrath J. P., Levinson A. D. A mutation at the ATP-binding site of pp60v-src abolishes kinase activity, transformation, and tumorigenicity. Mol Cell Biol. 1985 Jul;5(7):1772–1779. doi: 10.1128/mcb.5.7.1772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stoker A. W., Enrietto P. J., Wyke J. A. Functional domains of the pp60v-src protein as revealed by analysis of temperature-sensitive Rous sarcoma virus mutants. Mol Cell Biol. 1984 Aug;4(8):1508–1514. doi: 10.1128/mcb.4.8.1508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stoker A. W., Kellie S., Wyke J. A. Intracellular localization and processing of pp60v-src proteins expressed by two distinct temperature-sensitive mutants of Rous sarcoma virus. J Virol. 1986 Jun;58(3):876–883. doi: 10.1128/jvi.58.3.876-883.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Superti-Furga G., Fumagalli S., Koegl M., Courtneidge S. A., Draetta G. Csk inhibition of c-Src activity requires both the SH2 and SH3 domains of Src. EMBO J. 1993 Jul;12(7):2625–2634. doi: 10.1002/j.1460-2075.1993.tb05923.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tato F., Beamand J. A., Wyke J. A. A mutant of Rous sarcoma virus with a thermolabile defect in the virus envelope. Virology. 1978 Jul 1;88(1):71–81. doi: 10.1016/0042-6822(78)90111-3. [DOI] [PubMed] [Google Scholar]
- Vallette F., Mege E., Reiss A., Adesnik M. Construction of mutant and chimeric genes using the polymerase chain reaction. Nucleic Acids Res. 1989 Jan 25;17(2):723–733. doi: 10.1093/nar/17.2.723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vennström B., Fanshier L., Moscovici C., Bishop J. M. Molecular cloning of the avian erythroblastosis virus genome and recovery of oncogenic virus by transfection of chicken cells. J Virol. 1980 Nov;36(2):575–585. doi: 10.1128/jvi.36.2.575-585.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verderame M. F., Varmus H. E. Highly conserved amino acids in the SH2 and catalytic domains of v-src are altered in naturally occurring, transformation-defective alleles. Oncogene. 1994 Jan;9(1):175–182. [PubMed] [Google Scholar]
- Wages D. S., Keefer J., Rall T. B., Weber M. J. Mutations in the SH3 domain of the src oncogene which decrease association of phosphatidylinositol 3'-kinase activity with pp60v-src and alter cellular morphology. J Virol. 1992 Apr;66(4):1866–1874. doi: 10.1128/jvi.66.4.1866-1874.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waksman G., Kominos D., Robertson S. C., Pant N., Baltimore D., Birge R. B., Cowburn D., Hanafusa H., Mayer B. J., Overduin M. Crystal structure of the phosphotyrosine recognition domain SH2 of v-src complexed with tyrosine-phosphorylated peptides. Nature. 1992 Aug 20;358(6388):646–653. doi: 10.1038/358646a0. [DOI] [PubMed] [Google Scholar]
- Welham M. J., Wyke J. A. A single point mutation has pleiotropic effects on pp60v-src function. J Virol. 1988 Jun;62(6):1898–1906. doi: 10.1128/jvi.62.6.1898-1906.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welham M. J., Wyke J. A., Lang A., Wyke A. W. Mitogenesis induced by pp60v-src is not accompanied by increased expression of immediate early response genes. Oncogene. 1990 Feb;5(2):161–169. [PubMed] [Google Scholar]
- Wyke J. A., Linial M. Temperature-sensitive avian sarcoma viruses: a physiological comparison of twenty mutants. Virology. 1973 May;53(1):152–161. doi: 10.1016/0042-6822(73)90474-1. [DOI] [PubMed] [Google Scholar]
- Wyke J. A., Stoker A. W. Genetic analysis of the form and function of the viral src oncogene product. Biochim Biophys Acta. 1987 Apr 20;907(1):47–69. doi: 10.1016/0304-419x(87)90018-7. [DOI] [PubMed] [Google Scholar]
- Yu H., Rosen M. K., Shin T. B., Seidel-Dugan C., Brugge J. S., Schreiber S. L. Solution structure of the SH3 domain of Src and identification of its ligand-binding site. Science. 1992 Dec 4;258(5088):1665–1668. doi: 10.1126/science.1280858. [DOI] [PubMed] [Google Scholar]