Abstract
Eight sesquiterpene quinones: ilimaquinone (1), smenospongidine (3), smenospongiarine (5), smenospongine (7), and their corresponding 5-epimers 2, 4, 6, and 8, isolated from the Palauan marine sponge Hippospongia sp., were examined regarding their effects on TNF-α production in LPS-stimulated RAW 264.7 cells. 5-Epi-smenospongine (8) promoted the production of TNF-α to a level three times greater than the control at 10 μM, but compounds 1–7 did not show apparent activity. The results suggest that the cis-decaline ring and a primary amine in the benzoquinone ring are necessary for activity. This is the first study to report the modulation of TNF-α production by a sesquiterpene quinone.
Keywords: Sesquiterpene quinone, marine sponge, Hippospongia sp., TNF-α production, RAW 264.7 cells
1. Introduction
Marine sponges (Porifera) are well-known as prolific producers of bioactive metabolites. A sesquiterpene quinone, ilimaquinone, was reported in 1979 [1,2], and since then, related compounds have been obtained from several genera of marine sponge [3–9]. We have isolated 11 sesquiterpene quinones: ilimaquinone (1) [1,2] and its 5-epimer (2) [3], smenospongidine (3) [3,4] and its 5-epimer (4) [4], smenospongiarine (5) [4,5] and its 5-epimer (6) [4], smenospongine (7) [4,6] and its 5-epimer (8) [9], dactyloquinone B [7], 18-hydroxy-5-epihyrtiophenol [8], and pelorol [9], from Hippospongia sp. collected in Palau, and reported the effects of these compounds on the production of an inflammatory cytokine, interleukin-8 (IL-8), in tetradecanoyl phorbol acetate (PMA)-stimulated human promyelocytic leukemia HL-60 cells [10]. IL-8 is a member of the superfamily of C-X-C chemokines and a chemotactic factor for T-cells, neutrophils, and basophils [11]. Expression of IL-8 has been detected in a variety of human cancers and is suggested to be a factor involved in tumor progression and metastasis [12–15]. Compounds 3–8 increased the production of IL-8; therefore, we examined the effects of compounds 1–8 on the production of one of the most well-known and important inflammatory cytokines, TNF-α, in lipopolysaccharide (LPS)-stimulated murine monocytic leukemia RAW 264.7 cells.
2. Materials and Methods
2.1. Test Compounds
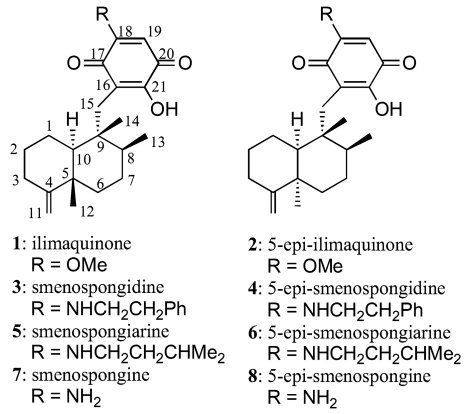
Sesquiterpene quinones 1–8 were isolated as described in the previous paper [10]. The structures of 1–8 are shown in Fig. 1.
Figure 1.
Structures of compounds 1–8.
2.2. Cell lines and culture conditions
The RAW 264.7 cell line was obtained from the Japanese Cancer Research Resources Bank (JCRB, Kamiyoga, Tokyo, Japan). This cell line was maintained in tissue culture dishes in RPMI 1640 medium (Nissui Seiyaku, Tokyo, Japan), supplemented with 10% heat-inactivated FCS, 2 mM glutamine, 100 U/ml of penicillin G and 100 μg/ml of streptomycin.
2.3. Detection of murine TNF-α by ELISA
TNF-α concentrations of the culture supernatants under control and various test conditions were measured by ELISA using a combination of monoclonal and polyclonal antibodies. All samples were assayed at least in duplicate. Data are presented as the mean ± SE of three independent experiments.
2.4. Determination of cell proliferation
Cell proliferation was evaluated by enumerating the viable cells using the MTT formazan production method [16]. RAW264.7 cells (1 × 106 cells/mL) were treated with LPS (with or without test compounds) and then transferred to 96-well microtiter plates. After incubation for 24 h, 20 μL of MTT reagent (5 mg/mL in PBS) was added to each well and further incubated for 3 h. The production of formazan was assessed by measuring optical density (OD570 nm). Data are shown as the values relative (%) to each PMA-stimulated optical density.
3. Results and Discussion
RAW 264.7 cells are known to produce TNF-α in response to the addition of LPS, and this system is used to detect the modulating activities of compounds on TNF-α production.
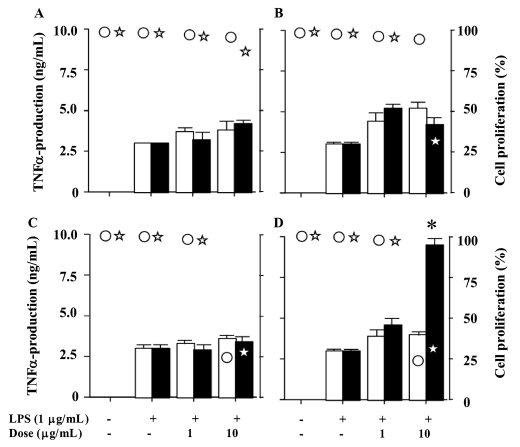
5-Epi-smenospongine (8) stimulated the production of TNF-α to a level three times greater than the control, but compounds 1–7 did not show any apparent activity (Fig. 2).
Figure 2. Effects of compounds 1–8 on TNF-α production and cell proliferation in LPS- stimulated RAW 264.7 cells.
Panels A, B, C, and D show the results of compounds 1 and 2, 3 and 4, 5 and 6, and 7 and 8, respectively. Open and solid bars represent lower and higher numbered compounds, respectively (* P < 0.05). Open circle and star represent lower and higher numbered compounds, respectively.
Compounds 3, 5, and 7 exhibited stronger activity on the promotion of IL-8 production than the corresponding 5-epimers 4, 6, and 8 in PMA-stimulated HL-60 cells [10], which showed the contribution of the trans-decaline ring in enhancing activity. The basic functional group at C-18 increased IL-8 production, since 3–8 exhibited stronger activities than 1 and 2 [10]. It was therefore revealed that the production of TNF-α induced by 8 involves a different mechanism from that involved in IL-8 production.
Ilimaquinone (1) showed antibacterial, cytotoxic, anti-HIV, hemolytic, and antimitotic activities, disruption of the Golgi apparatus, inhibition of the cytotoxicity of ricin and diphtheria toxin, and differentiation-inducing activity [5, 17–22]. Compounds 2–5 and 7 were reported to have cytotoxic, antibacterial, and hemolytic activities [4, 5, 17]. Compounds 2–4, 7, and 8 induced the differentiation of K562 cells into erythroblasts, as did 1 [22].
In this study, we revealed the production of TNF-α production in LPS-stimulated RAW 264.7 cells by one of the eight sesquiterpene quinones, 5-epi-smenospongine (8). This is the first report on the modulation of TNF-α production by sesquiterpene quinones.
From the comparison of structures 1–8, it is suggested that the cis-decaline ring and a primary amine in the benzoquinone ring are necessary for the activity of 8.
As we previously reported, compounds 3 to 8 induced the production of IL-8 by PMA-stimulated HL-60 cells [10]. In this study, we investigated their actions on TNF-α produced by LPS-stimulated mouse macrophage cell lines. Compound 8 induced TNF-α production. However, there was no influence of the other compounds. This may have been associated with the different actions of these compounds related to variations in receptors, signal molecules, and transcription factors in the production process of IL-8 and TNF-α as inflammatory cytokines. Therefore, the results of our experiment suggest that individual inflammatory cytokines can be controlled; these compounds should be further developed in the future.
Acknowledgements
We thank Prof. M. Namikoshi of Tohoku Pharmaceutical University for correcting our manuscript, and Ms. A. Fujita of Tokyo University of Marine Science and Technology for technical assistance.
Footnotes
Samples Availability: Not available.
References
- 1.Luibrand RT, Erdman TR, Vollmer JJ, Scheuer PJ, Finer J, Clardy J. Ilimaquinone, a sesquiterpenoid quinone from a marine sponge. Tetrahedron. 1979;35:609–612. [Google Scholar]
- 2.Capon RJ, MacLeod JK. A revision of the absolute stereochemistry of ilimaquinone. J Org Chem. 1987;52:5059–5060. [Google Scholar]
- 3.Carté B, Rose CB, Faulkner DJ. 5-epi-Ilimaquinone, a metabolite of the sponge Fenestraspongia Sp. J Org Chem. 1985;50:2785–2787. [Google Scholar]
- 4.Rodríguez J, Quiñoá E, Riguera R, Peters BM, Abrell LM, Crews P. The structures and stereochemistry of cytotoxic sesquiterpene quinones from Dactylospongia elegans. Tetrahedron. 1992;48:6667–6680. [Google Scholar]
- 5.Kondracki M-L, Guyot M. Biologically active quinone and hydroquinone sesquiterpenoids from the sponge Smenospongia sp. Tetrahedron. 1989;45:1995–2004. [Google Scholar]
- 6.Kondracki M-L, Guyot M. Smenospongine: a cytotoxic and antimicrobial aminoquinone isolated from Smenospongia sp. Tetrahedron Lett. 1987;28:5815–5818. [Google Scholar]
- 7.Mitome H, Nagasawa T, Miyaoka H, Yamada Y, van Soest RW. Dactyloquinones A and B, new sesquiterpenoid quinones from the Okinawan marine sponge Dactylospongia elegans. J Nat Prod. 2001;64:1506–1508. doi: 10.1021/np010299e. [DOI] [PubMed] [Google Scholar]
- 8.Salmoun M, Devijver C, Daloze D, Braekman JC, Gomez R, de Kluijver M, van Soest RWM. New sesquiterpene/quinones from two sponges of the genus Hyrtios. J Nat Prod. 2000;63:452–456. doi: 10.1021/np9903346. [DOI] [PubMed] [Google Scholar]
- 9.Kwak JH, Schmitz FJ, Kelly M. Sesquiterpene quinols/quinones from the Micronesian sponge Petrosaspongia metachromia. J Nat Prod. 2000;63:1153–1156. doi: 10.1021/np000079l. [DOI] [PubMed] [Google Scholar]
- 10.Oda T, Wang W, Fujita A, Mochizuki M, Ukai K, Namikoshi M. Promotion of IL-8 production in PMA-stimulated HL-60 cells by sesquiterpene quinones from a marine sponge Hippospongia sp. J Nat Med. 2007;61:434–437. [Google Scholar]
- 11.Graves DT, Jiang Y. Chemokines, a family of chemotactic cytokines. Crit Rev Oral Biol Med. 1995;6:109–118. doi: 10.1177/10454411950060020101. [DOI] [PubMed] [Google Scholar]
- 12.di Celle PF, Carbone A, Marchis D, Zhou D, Sozzani S, Zupo S, Pini M, Mantovani A, Foa R. Cytokine gene expression in B-cell chronic lymphocytic leukemia: evidence of constitutive interleukin-8 (IL-8) mRNA expression and secretion of biologically active IL-8 protein. Blood. 1994;84:220–228. [PubMed] [Google Scholar]
- 13.Green AR, Green VL, White MC, Speirs V. Expression of Cytokine messenger RNA in normal and neoplastic human breast tissue: identification of interleukin-8 as a potential regulatory factor in breast tumours. Int J Cancer. 1997;72:937–941. doi: 10.1002/(sici)1097-0215(19970917)72:6<937::aid-ijc3>3.0.co;2-q. [DOI] [PubMed] [Google Scholar]
- 14.Konig B, Steinbach F, Janocha B, Drynda A, Stumm M, Philipp C, Allhoff EP, Konig W. The differential expression of proinflammatory cytokines IL-6, IL-8 and TNF-alpha in renal cell carcinoma. Anticancer Res. 1999;19:1519–1524. [Google Scholar]
- 15.Galffy G, Mohammed KA, Dowling PA, Nasreen N, Ward MJ, Antony VB. Interleukin 8: an autocrine growth factor for malignant mesothelioma. Cancer Res. 1999;59:367–371. [PubMed] [Google Scholar]
- 16.Carmichael J, DeGraff WG, Gazdar AF, Minna JD, Mitchell JB. Evaluation of a tetrazolium-based semiautomated colorimetric assay: assessment of chemosensitivity testing. Cancer Res. 1987;47:936–942. [PubMed] [Google Scholar]
- 17.Prokofeva NG, Utkina NK, Chaikina EL, Makarchenko AE. Biological activities of marine sesquiterpenoid quinones: structure-activity relationships in cytotoxic and hemolytic assays. Comp Biochem Physiol Bio Mol Biol. 2004;139:169–173. doi: 10.1016/j.cbpc.2004.06.017. [DOI] [PubMed] [Google Scholar]
- 18.Tziveleka LA, Vagias C, Roussis V. Natural products with anti-HIV activity from marine organisms. Curr Top Med Chem. 2003;3:1512–1535. doi: 10.2174/1568026033451790. [DOI] [PubMed] [Google Scholar]
- 19.Pous C, Chabin K, Drechou A, Barbot L, Phung-Koskas T, Settegrana C, Bourguet-Kondracki ML, Maurice M, Cassio D, Guyot M, Durand G. Functional specialization of stable and dynamic microtubules in protein traffic in WIF-B cells. J Cell Biol. 1998;142:153–165. doi: 10.1083/jcb.142.1.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cruciani V, Mikalsen SO. Ilimaquinone inhibits gap junctional communication in a connexin isotype-specific manner. Exp Cell Res. 2005;304:136–148. doi: 10.1016/j.yexcr.2004.10.028. [DOI] [PubMed] [Google Scholar]
- 21.Nambiar MP, Wu HC. Ilimaquinone inhibits the cytotoxicities of ricin, diphtheria toxin, and other protein toxins in Vero cells. Exp Cell Res. 1995;219:671–678. doi: 10.1006/excr.1995.1278. [DOI] [PubMed] [Google Scholar]
- 22.Aoki S, Kong D, Matsui K, Rachmat R, Kobayashi M. Sesquiterpene aminoquinones, from a marine sponge, induce erythroid differentiation in human chronic myelogenous leukemia, K562 cells. Chem Pharm Bull. 2004;52:935–937. doi: 10.1248/cpb.52.935. [DOI] [PubMed] [Google Scholar]