Abstract
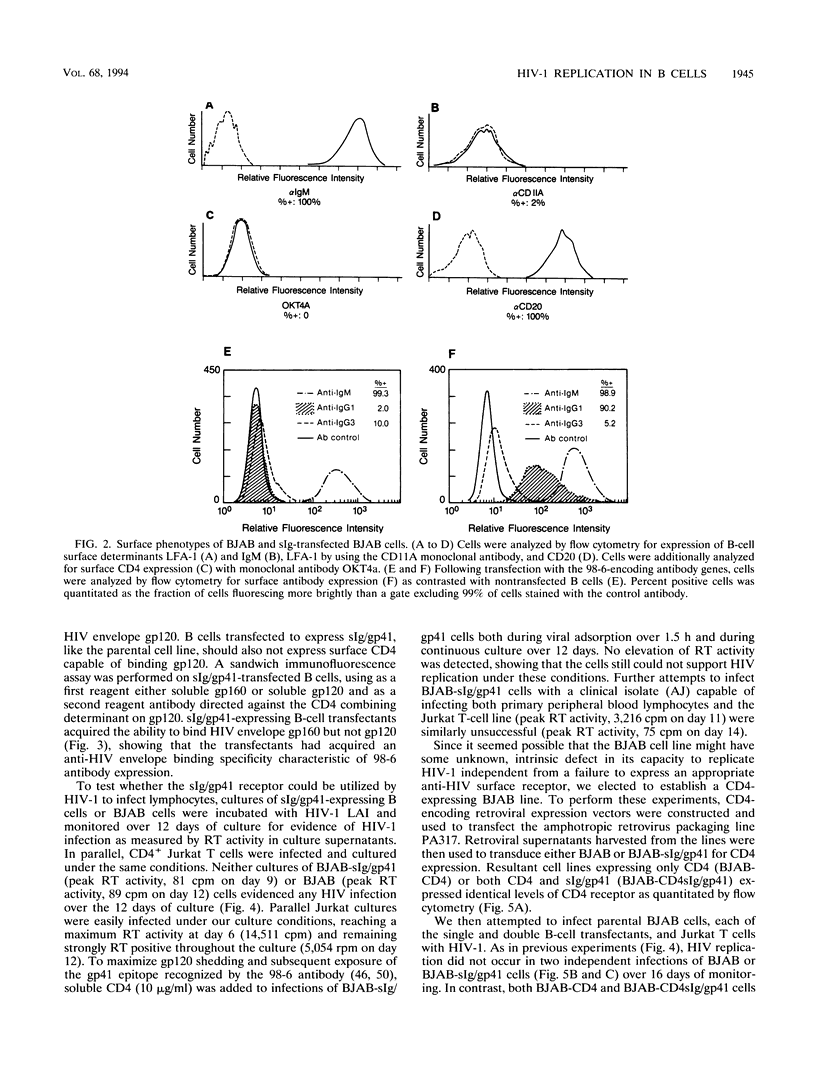
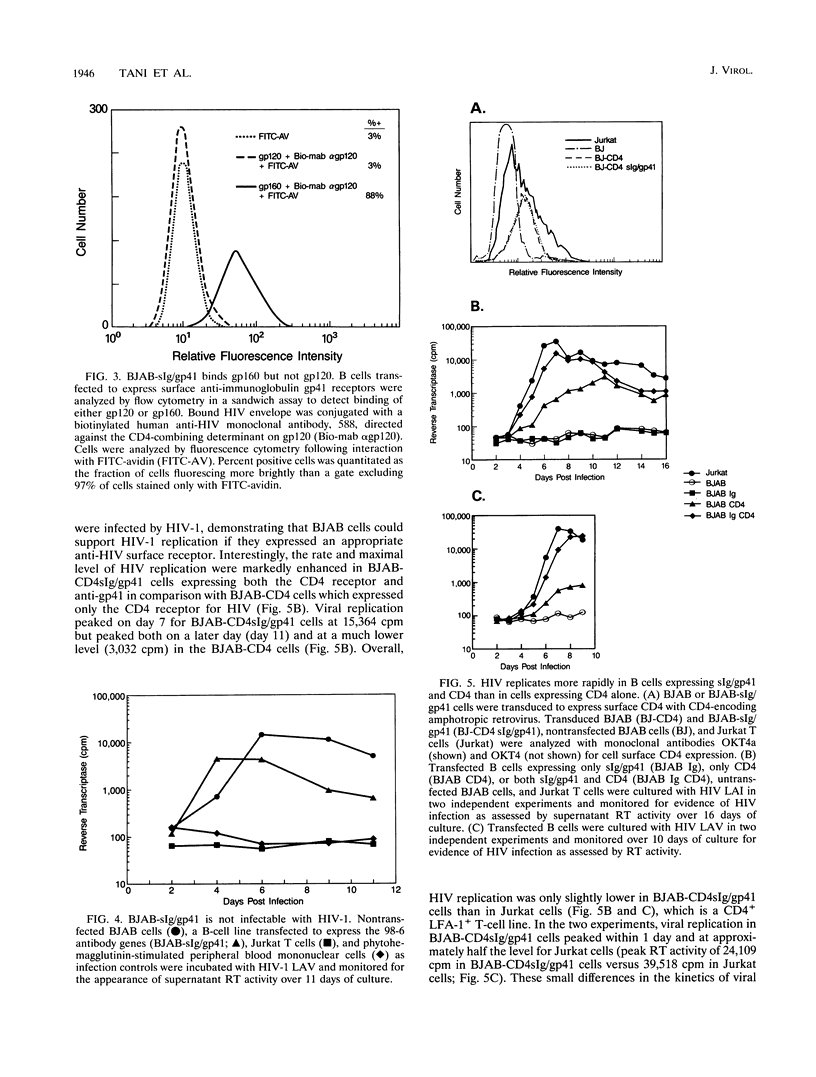
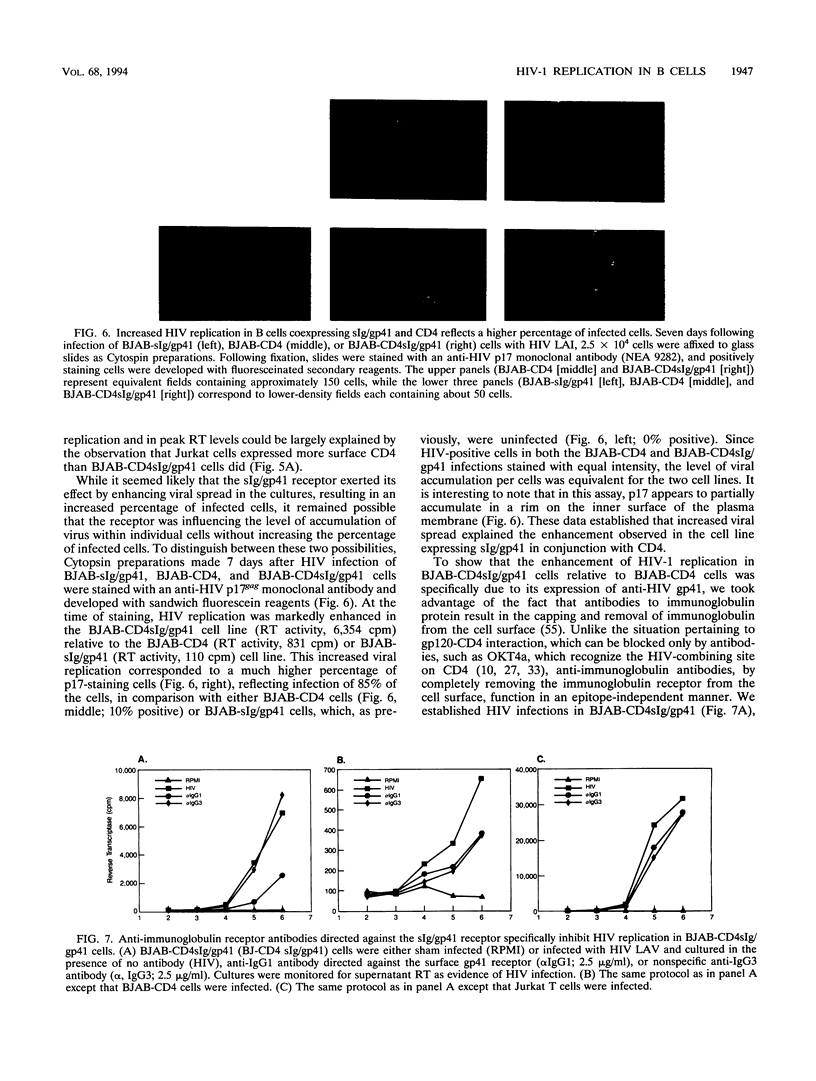
The human immunodeficiency virus type 1 (HIV-1) external envelope glycoprotein gp120 tightly binds CD4 as its principal cellular receptor, explaining the tropism of HIV-1 for CD4+ cells. Nevertheless, reports documenting HIV infection or HIV binding in cells lacking CD4 surface expression have raised the possibility that cellular receptors in addition to CD4 may interact with HIV envelope. Moreover, the lymphocyte adhesion molecule LFA-1 appears to play an important role in augmenting HIV-1 viral spread and cytopathicity in vitro, although the mechanism of this function is still not completely defined. In the course of characterizing a human anti-HIV gp41 monoclonal antibody, we transfected a CD4-negative, LFA-1-negative B-cell line to express an anti-gp41 immunoglobulin receptor (surface immunoglobulin [sIg]/gp41). Despite acquiring the ability to bind HIV envelope, such transfected B cells could not be infected by HIV-1. These cells were not intrinsically defective for supporting HIV-1 infection, because when directed to produce surface CD4 by using retroviral constructs, they acquired the ability to replicate HIV-1. Interestingly, transfected cells expressing both surface CD4 and sIg/gp41 receptors replicated HIV much better than cells expressing only CD4. The enhancement resided specifically in sIg/gp41, because isotype-specific, anti-IgG1 antibodies directed against sIg/gp41 blocked the enhancement. These data directly establish the ability of a cell surface anti-gp41 receptor to enhance HIV-1 replication.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amadori A., Zamarchi R., Ciminale V., Del Mistro A., Siervo S., Alberti A., Colombatti M., Chieco-Bianchi L. HIV-1-specific B cell activation. A major constituent of spontaneous B cell activation during HIV-1 infection. J Immunol. 1989 Oct 1;143(7):2146–2152. [PubMed] [Google Scholar]
- Andris J. S., Johnson S., Zolla-Pazner S., Capra J. D. Molecular characterization of five human anti-human immunodeficiency virus type 1 antibody heavy chains reveals extensive somatic mutation typical of an antigen-driven immune response. Proc Natl Acad Sci U S A. 1991 Sep 1;88(17):7783–7787. doi: 10.1073/pnas.88.17.7783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arthur L. O., Bess J. W., Jr, Sowder R. C., 2nd, Benveniste R. E., Mann D. L., Chermann J. C., Henderson L. E. Cellular proteins bound to immunodeficiency viruses: implications for pathogenesis and vaccines. Science. 1992 Dec 18;258(5090):1935–1938. doi: 10.1126/science.1470916. [DOI] [PubMed] [Google Scholar]
- Berger E. A., Lifson J. D., Eiden L. E. Stimulation of glycoprotein gp120 dissociation from the envelope glycoprotein complex of human immunodeficiency virus type 1 by soluble CD4 and CD4 peptide derivatives: implications for the role of the complementarity-determining region 3-like region in membrane fusion. Proc Natl Acad Sci U S A. 1991 Sep 15;88(18):8082–8086. doi: 10.1073/pnas.88.18.8082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blobel C. P., Wolfsberg T. G., Turck C. W., Myles D. G., Primakoff P., White J. M. A potential fusion peptide and an integrin ligand domain in a protein active in sperm-egg fusion. Nature. 1992 Mar 19;356(6366):248–252. doi: 10.1038/356248a0. [DOI] [PubMed] [Google Scholar]
- Celada F., Cambiaggi C., Maccari J., Burastero S., Gregory T., Patzer E., Porter J., McDanal C., Matthews T. Antibody raised against soluble CD4-rgp120 complex recognizes the CD4 moiety and blocks membrane fusion without inhibiting CD4-gp120 binding. J Exp Med. 1990 Oct 1;172(4):1143–1150. doi: 10.1084/jem.172.4.1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clapham P. R., Weber J. N., Whitby D., McIntosh K., Dalgleish A. G., Maddon P. J., Deen K. C., Sweet R. W., Weiss R. A. Soluble CD4 blocks the infectivity of diverse strains of HIV and SIV for T cells and monocytes but not for brain and muscle cells. Nature. 1989 Jan 26;337(6205):368–370. doi: 10.1038/337368a0. [DOI] [PubMed] [Google Scholar]
- Clements G. J., Price-Jones M. J., Stephens P. E., Sutton C., Schulz T. F., Clapham P. R., McKeating J. A., McClure M. O., Thomson S., Marsh M. The V3 loops of the HIV-1 and HIV-2 surface glycoproteins contain proteolytic cleavage sites: a possible function in viral fusion? AIDS Res Hum Retroviruses. 1991 Jan;7(1):3–16. doi: 10.1089/aid.1991.7.3. [DOI] [PubMed] [Google Scholar]
- Curtis B. M., Scharnowske S., Watson A. J. Sequence and expression of a membrane-associated C-type lectin that exhibits CD4-independent binding of human immunodeficiency virus envelope glycoprotein gp120. Proc Natl Acad Sci U S A. 1992 Sep 1;89(17):8356–8360. doi: 10.1073/pnas.89.17.8356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalgleish A. G., Beverley P. C., Clapham P. R., Crawford D. H., Greaves M. F., Weiss R. A. The CD4 (T4) antigen is an essential component of the receptor for the AIDS retrovirus. Nature. 1984 Dec 20;312(5996):763–767. doi: 10.1038/312763a0. [DOI] [PubMed] [Google Scholar]
- Dubay J. W., Roberts S. J., Brody B., Hunter E. Mutations in the leucine zipper of the human immunodeficiency virus type 1 transmembrane glycoprotein affect fusion and infectivity. J Virol. 1992 Aug;66(8):4748–4756. doi: 10.1128/jvi.66.8.4748-4756.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Earl P. L., Doms R. W., Moss B. Multimeric CD4 binding exhibited by human and simian immunodeficiency virus envelope protein dimers. J Virol. 1992 Sep;66(9):5610–5614. doi: 10.1128/jvi.66.9.5610-5614.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foltz K. R., Partin J. S., Lennarz W. J. Sea urchin egg receptor for sperm: sequence similarity of binding domain and hsp70. Science. 1993 Mar 5;259(5100):1421–1425. doi: 10.1126/science.8383878. [DOI] [PubMed] [Google Scholar]
- Freed E. O., Delwart E. L., Buchschacher G. L., Jr, Panganiban A. T. A mutation in the human immunodeficiency virus type 1 transmembrane glycoprotein gp41 dominantly interferes with fusion and infectivity. Proc Natl Acad Sci U S A. 1992 Jan 1;89(1):70–74. doi: 10.1073/pnas.89.1.70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freed E. O., Myers D. J., Risser R. Identification of the principal neutralizing determinant of human immunodeficiency virus type 1 as a fusion domain. J Virol. 1991 Jan;65(1):190–194. doi: 10.1128/jvi.65.1.190-194.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Golding H., Dimitrov D. S., Blumenthal R. LFA-1 adhesion molecules are not involved in the early stages of HIV-1 env-mediated cell membrane fusion. AIDS Res Hum Retroviruses. 1992 Sep;8(9):1593–1598. doi: 10.1089/aid.1992.8.1593. [DOI] [PubMed] [Google Scholar]
- Gorny M. K., Gianakakos V., Sharpe S., Zolla-Pazner S. Generation of human monoclonal antibodies to human immunodeficiency virus. Proc Natl Acad Sci U S A. 1989 Mar;86(5):1624–1628. doi: 10.1073/pnas.86.5.1624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harouse J. M., Kunsch C., Hartle H. T., Laughlin M. A., Hoxie J. A., Wigdahl B., Gonzalez-Scarano F. CD4-independent infection of human neural cells by human immunodeficiency virus type 1. J Virol. 1989 Jun;63(6):2527–2533. doi: 10.1128/jvi.63.6.2527-2533.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasunuma T., Tsubota H., Watanabe M., Chen Z. W., Lord C. I., Burkly L. C., Daley J. F., Letvin N. L. Regions of the CD4 molecule not involved in virus binding or syncytia formation are required for HIV-1 infection of lymphocytes. J Immunol. 1992 Mar 15;148(6):1841–1846. [PubMed] [Google Scholar]
- Hattori T., Koito A., Takatsuki K., Kido H., Katunuma N. Involvement of tryptase-related cellular protease(s) in human immunodeficiency virus type 1 infection. FEBS Lett. 1989 May 8;248(1-2):48–52. doi: 10.1016/0014-5793(89)80429-6. [DOI] [PubMed] [Google Scholar]
- Healey D., Dianda L., Moore J. P., McDougal J. S., Moore M. J., Estess P., Buck D., Kwong P. D., Beverley P. C., Sattentau Q. J. Novel anti-CD4 monoclonal antibodies separate human immunodeficiency virus infection and fusion of CD4+ cells from virus binding. J Exp Med. 1990 Oct 1;172(4):1233–1242. doi: 10.1084/jem.172.4.1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hildreth J. E., Orentas R. J. Involvement of a leukocyte adhesion receptor (LFA-1) in HIV-induced syncytium formation. Science. 1989 Jun 2;244(4908):1075–1078. doi: 10.1126/science.2543075. [DOI] [PubMed] [Google Scholar]
- Homsy J., Meyer M., Levy J. A. Serum enhancement of human immunodeficiency virus (HIV) infection correlates with disease in HIV-infected individuals. J Virol. 1990 Apr;64(4):1437–1440. doi: 10.1128/jvi.64.4.1437-1440.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikeuchi K., Kim S., Byrn R. A., Goldring S. R., Groopman J. E. Infection of nonlymphoid cells by human immunodeficiency virus type 1 or type 2. J Virol. 1990 Sep;64(9):4226–4231. doi: 10.1128/jvi.64.9.4226-4231.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karwowska S., Gorny M. K., Buchbinder A., Gianakakos V., Williams C., Fuerst T., Zolla-Pazner S. Production of human monoclonal antibodies specific for conformational and linear non-V3 epitopes of gp120. AIDS Res Hum Retroviruses. 1992 Jun;8(6):1099–1106. doi: 10.1089/aid.1992.8.1099. [DOI] [PubMed] [Google Scholar]
- Kido H., Fukutomi A., Katunuma N. A novel membrane-bound serine esterase in human T4+ lymphocytes immunologically reactive with antibody inhibiting syncytia induced by HIV-1. Purification and characterization. J Biol Chem. 1990 Dec 15;265(35):21979–21985. [PubMed] [Google Scholar]
- Klatzmann D., Champagne E., Chamaret S., Gruest J., Guetard D., Hercend T., Gluckman J. C., Montagnier L. T-lymphocyte T4 molecule behaves as the receptor for human retrovirus LAV. Nature. 1984 Dec 20;312(5996):767–768. doi: 10.1038/312767a0. [DOI] [PubMed] [Google Scholar]
- Kowalski M., Bergeron L., Dorfman T., Haseltine W., Sodroski J. Attenuation of human immunodeficiency virus type 1 cytopathic effect by a mutation affecting the transmembrane envelope glycoprotein. J Virol. 1991 Jan;65(1):281–291. doi: 10.1128/jvi.65.1.281-291.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozlowski M. R., Sandler P., Lin P. F., Watson A. Brain-derived cells contain a specific binding site for Gp120 which is not the CD4 antigen. Brain Res. 1991 Jul 12;553(2):300–304. doi: 10.1016/0006-8993(91)90838-m. [DOI] [PubMed] [Google Scholar]
- Kunsch C., Hartle H. T., Wigdahl B. Infection of human fetal dorsal root ganglion glial cells with human immunodeficiency virus type 1 involves an entry mechanism independent of the CD4 T4A epitope. J Virol. 1989 Dec;63(12):5054–5061. doi: 10.1128/jvi.63.12.5054-5061.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X. L., Moudgil T., Vinters H. V., Ho D. D. CD4-independent, productive infection of a neuronal cell line by human immunodeficiency virus type 1. J Virol. 1990 Mar;64(3):1383–1387. doi: 10.1128/jvi.64.3.1383-1387.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markowitz D., Goff S., Bank A. A safe packaging line for gene transfer: separating viral genes on two different plasmids. J Virol. 1988 Apr;62(4):1120–1124. doi: 10.1128/jvi.62.4.1120-1124.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDougal J. S., Kennedy M. S., Sligh J. M., Cort S. P., Mawle A., Nicholson J. K. Binding of HTLV-III/LAV to T4+ T cells by a complex of the 110K viral protein and the T4 molecule. Science. 1986 Jan 24;231(4736):382–385. doi: 10.1126/science.3001934. [DOI] [PubMed] [Google Scholar]
- Miller A. D., Buttimore C. Redesign of retrovirus packaging cell lines to avoid recombination leading to helper virus production. Mol Cell Biol. 1986 Aug;6(8):2895–2902. doi: 10.1128/mcb.6.8.2895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller A. D., Rosman G. J. Improved retroviral vectors for gene transfer and expression. Biotechniques. 1989 Oct;7(9):980-2, 984-6, 989-90. [PMC free article] [PubMed] [Google Scholar]
- Montefiori D. C., Robinson W. E., Jr, Hirsch V. M., Modliszewski A., Mitchell W. M., Johnson P. R. Antibody-dependent enhancement of simian immunodeficiency virus (SIV) infection in vitro by plasma from SIV-infected rhesus macaques. J Virol. 1990 Jan;64(1):113–119. doi: 10.1128/jvi.64.1.113-119.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore J. P., McKeating J. A., Huang Y. X., Ashkenazi A., Ho D. D. Virions of primary human immunodeficiency virus type 1 isolates resistant to soluble CD4 (sCD4) neutralization differ in sCD4 binding and glycoprotein gp120 retention from sCD4-sensitive isolates. J Virol. 1992 Jan;66(1):235–243. doi: 10.1128/jvi.66.1.235-243.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore J. P., McKeating J. A., Norton W. A., Sattentau Q. J. Direct measurement of soluble CD4 binding to human immunodeficiency virus type 1 virions: gp120 dissociation and its implications for virus-cell binding and fusion reactions and their neutralization by soluble CD4. J Virol. 1991 Mar;65(3):1133–1140. doi: 10.1128/jvi.65.3.1133-1140.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Page K. A., Stearns S. M., Littman D. R. Analysis of mutations in the V3 domain of gp160 that affect fusion and infectivity. J Virol. 1992 Jan;66(1):524–533. doi: 10.1128/jvi.66.1.524-533.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pantaleo G., Butini L., Graziosi C., Poli G., Schnittman S. M., Greenhouse J. J., Gallin J. I., Fauci A. S. Human immunodeficiency virus (HIV) infection in CD4+ T lymphocytes genetically deficient in LFA-1: LFA-1 is required for HIV-mediated cell fusion but not for viral transmission. J Exp Med. 1991 Feb 1;173(2):511–514. doi: 10.1084/jem.173.2.511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinter A., Honnen W. J., Tilley S. A., Bona C., Zaghouani H., Gorny M. K., Zolla-Pazner S. Oligomeric structure of gp41, the transmembrane protein of human immunodeficiency virus type 1. J Virol. 1989 Jun;63(6):2674–2679. doi: 10.1128/jvi.63.6.2674-2679.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson W. E., Jr, Gorny M. K., Xu J. Y., Mitchell W. M., Zolla-Pazner S. Two immunodominant domains of gp41 bind antibodies which enhance human immunodeficiency virus type 1 infection in vitro. J Virol. 1991 Aug;65(8):4169–4176. doi: 10.1128/jvi.65.8.4169-4176.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson W. E., Jr, Kawamura T., Gorny M. K., Lake D., Xu J. Y., Matsumoto Y., Sugano T., Masuho Y., Mitchell W. M., Hersh E. Human monoclonal antibodies to the human immunodeficiency virus type 1 (HIV-1) transmembrane glycoprotein gp41 enhance HIV-1 infection in vitro. Proc Natl Acad Sci U S A. 1990 Apr;87(8):3185–3189. doi: 10.1073/pnas.87.8.3185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson W. E., Jr, Kawamura T., Lake D., Masuho Y., Mitchell W. M., Hersh E. M. Antibodies to the primary immunodominant domain of human immunodeficiency virus type 1 (HIV-1) glycoprotein gp41 enhance HIV-1 infection in vitro. J Virol. 1990 Nov;64(11):5301–5305. doi: 10.1128/jvi.64.11.5301-5305.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson W. E., Jr, Montefiori D. C., Mitchell W. M., Prince A. M., Alter H. J., Dreesman G. R., Eichberg J. W. Antibody-dependent enhancement of human immunodeficiency virus type 1 (HIV-1) infection in vitro by serum from HIV-1-infected and passively immunized chimpanzees. Proc Natl Acad Sci U S A. 1989 Jun;86(12):4710–4714. doi: 10.1073/pnas.86.12.4710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sattentau Q. J., Moore J. P. Conformational changes induced in the human immunodeficiency virus envelope glycoprotein by soluble CD4 binding. J Exp Med. 1991 Aug 1;174(2):407–415. doi: 10.1084/jem.174.2.407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnittman S. M., Psallidopoulos M. C., Lane H. C., Thompson L., Baseler M., Massari F., Fox C. H., Salzman N. P., Fauci A. S. The reservoir for HIV-1 in human peripheral blood is a T cell that maintains expression of CD4. Science. 1989 Jul 21;245(4915):305–308. doi: 10.1126/science.2665081. [DOI] [PubMed] [Google Scholar]
- Shioda T., Levy J. A., Cheng-Mayer C. Macrophage and T cell-line tropisms of HIV-1 are determined by specific regions of the envelope gp120 gene. Nature. 1991 Jan 10;349(6305):167–169. doi: 10.1038/349167a0. [DOI] [PubMed] [Google Scholar]
- Shioda T., Levy J. A., Cheng-Mayer C. Small amino acid changes in the V3 hypervariable region of gp120 can affect the T-cell-line and macrophage tropism of human immunodeficiency virus type 1. Proc Natl Acad Sci U S A. 1992 Oct 15;89(20):9434–9438. doi: 10.1073/pnas.89.20.9434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spear G. T., Takefman D. M., Sullivan B. L., Landay A. L., Zolla-Pazner S. Complement activation by human monoclonal antibodies to human immunodeficiency virus. J Virol. 1993 Jan;67(1):53–59. doi: 10.1128/jvi.67.1.53-59.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Springer T. A. Adhesion receptors of the immune system. Nature. 1990 Aug 2;346(6283):425–434. doi: 10.1038/346425a0. [DOI] [PubMed] [Google Scholar]
- Takeuchi Y., Akutsu M., Murayama K., Shimizu N., Hoshino H. Host range mutant of human immunodeficiency virus type 1: modification of cell tropism by a single point mutation at the neutralization epitope in the env gene. J Virol. 1991 Apr;65(4):1710–1718. doi: 10.1128/jvi.65.4.1710-1718.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tateno M., Gonzalez-Scarano F., Levy J. A. Human immunodeficiency virus can infect CD4-negative human fibroblastoid cells. Proc Natl Acad Sci U S A. 1989 Jun;86(11):4287–4290. doi: 10.1073/pnas.86.11.4287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyler D. S., Stanley S. D., Zolla-Pazner S., Gorny M. K., Shadduck P. P., Langlois A. J., Matthews T. J., Bolognesi D. P., Palker T. J., Weinhold K. J. Identification of sites within gp41 that serve as targets for antibody-dependent cellular cytotoxicity by using human monoclonal antibodies. J Immunol. 1990 Nov 15;145(10):3276–3282. [PubMed] [Google Scholar]
- Willey R. L., Smith D. H., Lasky L. A., Theodore T. S., Earl P. L., Moss B., Capon D. J., Martin M. A. In vitro mutagenesis identifies a region within the envelope gene of the human immunodeficiency virus that is critical for infectivity. J Virol. 1988 Jan;62(1):139–147. doi: 10.1128/jvi.62.1.139-147.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J. Y., Gorny M. K., Palker T., Karwowska S., Zolla-Pazner S. Epitope mapping of two immunodominant domains of gp41, the transmembrane protein of human immunodeficiency virus type 1, using ten human monoclonal antibodies. J Virol. 1991 Sep;65(9):4832–4838. doi: 10.1128/jvi.65.9.4832-4838.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada M., Zurbriggen A., Oldstone M. B., Fujinami R. S. Common immunologic determinant between human immunodeficiency virus type 1 gp41 and astrocytes. J Virol. 1991 Mar;65(3):1370–1376. doi: 10.1128/jvi.65.3.1370-1376.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]



