Abstract
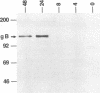
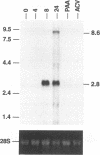
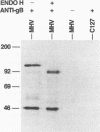
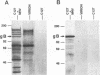
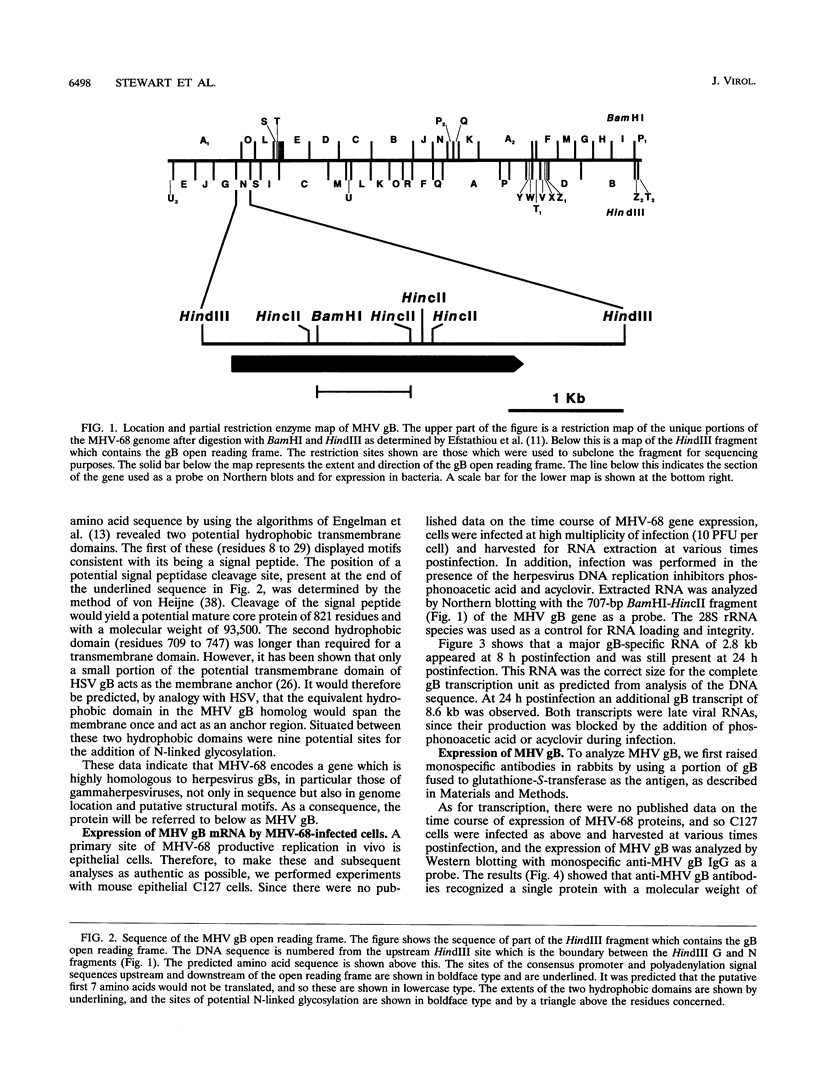
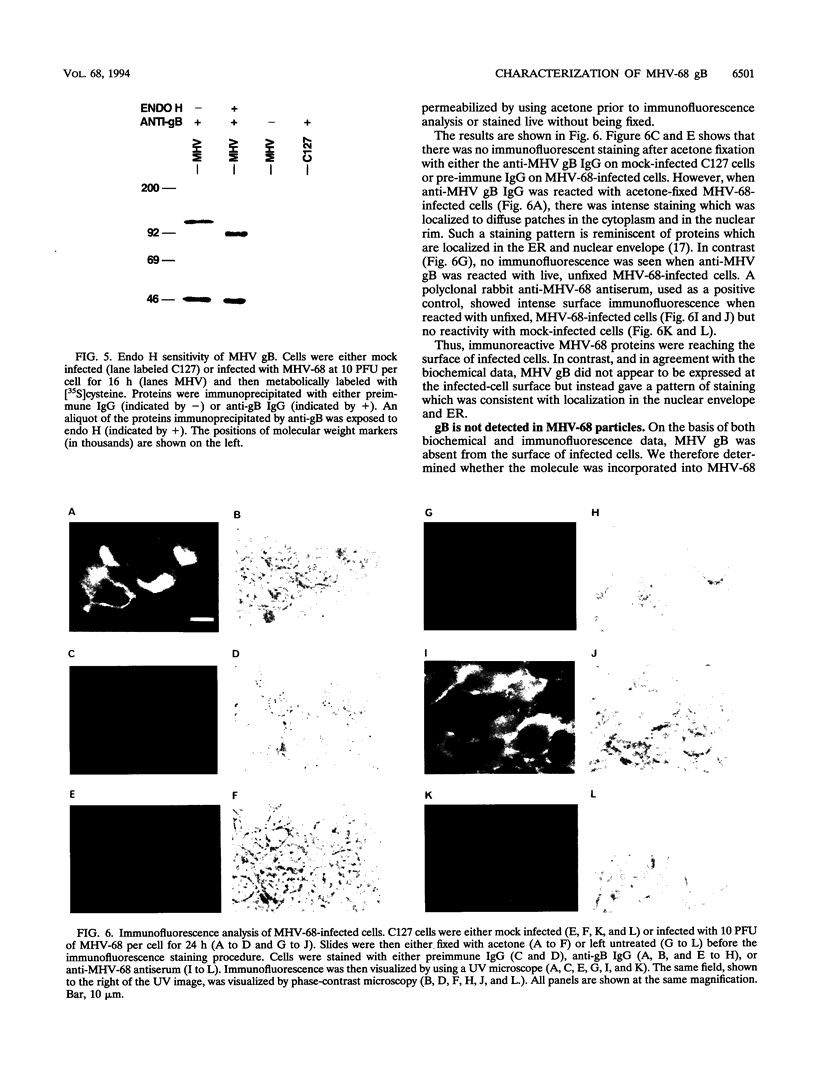
Murine gammaherpesvirus 68 (MHV-68) is a natural pathogen of murid rodents and displays similar pathobiological characteristics to those of the human gammaherpesvirus Epstein-Barr virus (EBV). However, in contrast to EBV, MHV-68 will replicate in epithelial cells in vitro. It has therefore been proposed that MHV-68 may be of use as a model for the study of gammaherpesviruses, EBV in particular, both in vitro and in vivo. The EBV homolog of herpes simplex virus glycoprotein B (gB), termed gp110, is somewhat unusual compared with those of many other herpesviruses. We therefore decided to characterize the homolog of gB encoded by MHV-68 (termed MHV gB) to observe the properties of a gammaherpesvirus gB produced in epithelial cells and also to test the relatedness of MHV-68 and EBV. The MHV gB-coding sequence was determined from cloned DNA. The predicted amino acid sequence shared closest homology with gammaherpesvirus gB homologs. Biochemical analysis showed that MHV gB was a glycoprotein with a molecular weight of 105,000. However, the glycans were of the N-linked, high-mannose type, indicating retention in the endoplasmic reticulum. In line with this, MHV gB was localized to the cytoplasm and nuclear margins of infected cells but was not detected on the cell surface or in virions. Additionally, anti-MHV gB antisera were nonneutralizing. Thus, the MHV gB was unlike many other herpesvirus gBs but was extremely similar to the EBV gB. This highlights the close relationship between MHV-68 and EBV and underlines the potential of MHV-68 as a model for EBV in epithelial cells both in vitro and in vivo.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Albrecht J. C., Nicholas J., Biller D., Cameron K. R., Biesinger B., Newman C., Wittmann S., Craxton M. A., Coleman H., Fleckenstein B. Primary structure of the herpesvirus saimiri genome. J Virol. 1992 Aug;66(8):5047–5058. doi: 10.1128/jvi.66.8.5047-5058.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaskovic D., Sekeyová Z., Turna J., Kúdelová M., Slávik I., Mucha V. Purification of murine alpha-herpesvirus and some properties of its DNA. Acta Virol. 1988 Jul;32(4):329–333. [PubMed] [Google Scholar]
- Blaskovic D., Stanceková M., Svobodová J., Mistríková J. Isolation of five strains of herpesviruses from two species of free living small rodents. Acta Virol. 1980 Dec;24(6):468–468. [PubMed] [Google Scholar]
- Bzik D. J., Fox B. A., DeLuca N. A., Person S. Nucleotide sequence specifying the glycoprotein gene, gB, of herpes simplex virus type 1. Virology. 1984 Mar;133(2):301–314. doi: 10.1016/0042-6822(84)90397-0. [DOI] [PubMed] [Google Scholar]
- Cai W. H., Gu B., Person S. Role of glycoprotein B of herpes simplex virus type 1 in viral entry and cell fusion. J Virol. 1988 Aug;62(8):2596–2604. doi: 10.1128/jvi.62.8.2596-2604.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavener D. R., Ray S. C. Eukaryotic start and stop translation sites. Nucleic Acids Res. 1991 Jun 25;19(12):3185–3192. doi: 10.1093/nar/19.12.3185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dayhoff M. O., Barker W. C., Hunt L. T. Establishing homologies in protein sequences. Methods Enzymol. 1983;91:524–545. doi: 10.1016/s0076-6879(83)91049-2. [DOI] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Efstathiou S., Ho Y. M., Hall S., Styles C. J., Scott S. D., Gompels U. A. Murine herpesvirus 68 is genetically related to the gammaherpesviruses Epstein-Barr virus and herpesvirus saimiri. J Gen Virol. 1990 Jun;71(Pt 6):1365–1372. doi: 10.1099/0022-1317-71-6-1365. [DOI] [PubMed] [Google Scholar]
- Efstathiou S., Ho Y. M., Minson A. C. Cloning and molecular characterization of the murine herpesvirus 68 genome. J Gen Virol. 1990 Jun;71(Pt 6):1355–1364. doi: 10.1099/0022-1317-71-6-1355. [DOI] [PubMed] [Google Scholar]
- Ehtisham S., Sunil-Chandra N. P., Nash A. A. Pathogenesis of murine gammaherpesvirus infection in mice deficient in CD4 and CD8 T cells. J Virol. 1993 Sep;67(9):5247–5252. doi: 10.1128/jvi.67.9.5247-5252.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engelman D. M., Steitz T. A., Goldman A. Identifying nonpolar transbilayer helices in amino acid sequences of membrane proteins. Annu Rev Biophys Biophys Chem. 1986;15:321–353. doi: 10.1146/annurev.bb.15.060186.001541. [DOI] [PubMed] [Google Scholar]
- Gilbert R., Ghosh H. P. Immunoelectron microscopic localization of herpes simplex virus glycoprotein gB in the nuclear envelope of infected cells. Virus Res. 1993 Jun;28(3):217–231. doi: 10.1016/0168-1702(93)90023-g. [DOI] [PubMed] [Google Scholar]
- Gilbert R., Ghosh K., Rasile L., Ghosh H. P. Membrane anchoring domain of herpes simplex virus glycoprotein gB is sufficient for nuclear envelope localization. J Virol. 1994 Apr;68(4):2272–2285. doi: 10.1128/jvi.68.4.2272-2285.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gong M., Kieff E. Intracellular trafficking of two major Epstein-Barr virus glycoproteins, gp350/220 and gp110. J Virol. 1990 Apr;64(4):1507–1516. doi: 10.1128/jvi.64.4.1507-1516.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gong M., Ooka T., Matsuo T., Kieff E. Epstein-Barr virus glycoprotein homologous to herpes simplex virus gB. J Virol. 1987 Feb;61(2):499–508. doi: 10.1128/jvi.61.2.499-508.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Highlander S. L., Cai W. H., Person S., Levine M., Glorioso J. C. Monoclonal antibodies define a domain on herpes simplex virus glycoprotein B involved in virus penetration. J Virol. 1988 Jun;62(6):1881–1888. doi: 10.1128/jvi.62.6.1881-1888.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson D. C., Spear P. G. O-linked oligosaccharides are acquired by herpes simplex virus glycoproteins in the Golgi apparatus. Cell. 1983 Mar;32(3):987–997. doi: 10.1016/0092-8674(83)90083-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornfeld R., Kornfeld S. Assembly of asparagine-linked oligosaccharides. Annu Rev Biochem. 1985;54:631–664. doi: 10.1146/annurev.bi.54.070185.003215. [DOI] [PubMed] [Google Scholar]
- Little S. P., Jofre J. T., Courtney R. J., Schaffer P. A. A virion-associated glycoprotein essential for infectivity of herpes simplex virus type 1. Virology. 1981 Nov;115(1):149–160. doi: 10.1016/0042-6822(81)90097-0. [DOI] [PubMed] [Google Scholar]
- Lowy D. R., Rands E., Scolnick E. M. Helper-independent transformation by unintegrated Harvey sarcoma virus DNA. J Virol. 1978 May;26(2):291–298. doi: 10.1128/jvi.26.2.291-298.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pellett P. E., Biggin M. D., Barrell B., Roizman B. Epstein-Barr virus genome may encode a protein showing significant amino acid and predicted secondary structure homology with glycoprotein B of herpes simplex virus 1. J Virol. 1985 Dec;56(3):807–813. doi: 10.1128/jvi.56.3.807-813.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pellett P. E., Kousoulas K. G., Pereira L., Roizman B. Anatomy of the herpes simplex virus 1 strain F glycoprotein B gene: primary sequence and predicted protein structure of the wild type and of monoclonal antibody-resistant mutants. J Virol. 1985 Jan;53(1):243–253. doi: 10.1128/jvi.53.1.243-253.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasile L., Ghosh K., Raviprakash K., Ghosh H. P. Effects of deletions in the carboxy-terminal hydrophobic region of herpes simplex virus glycoprotein gB on intracellular transport and membrane anchoring. J Virol. 1993 Aug;67(8):4856–4866. doi: 10.1128/jvi.67.8.4856-4866.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarmiento M., Haffey M., Spear P. G. Membrane proteins specified by herpes simplex viruses. III. Role of glycoprotein VP7(B2) in virion infectivity. J Virol. 1979 Mar;29(3):1149–1158. doi: 10.1128/jvi.29.3.1149-1158.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart J. P., Arrand J. R. Expression of the Epstein-Barr virus latent membrane protein in nasopharyngeal carcinoma biopsy specimens. Hum Pathol. 1993 Mar;24(3):239–242. doi: 10.1016/0046-8177(93)90032-c. [DOI] [PubMed] [Google Scholar]
- Stewart J. P., Behm F. G., Arrand J. R., Rooney C. M. Differential expression of viral and human interleukin-10 (IL-10) by primary B cell tumors and B cell lines. Virology. 1994 May 1;200(2):724–732. doi: 10.1006/viro.1994.1236. [DOI] [PubMed] [Google Scholar]
- Stewart J. P., Hampson I. N., Heinrich H. W., Mackett M., Arrand J. R. Enhanced expression of the Epstein-Barr virus latent membrane protein by a recombinant vaccinia virus. J Gen Virol. 1989 May;70(Pt 5):1231–1237. doi: 10.1099/0022-1317-70-5-1231. [DOI] [PubMed] [Google Scholar]
- Stewart J. P., Rooney C. M. The interleukin-10 homolog encoded by Epstein-Barr virus enhances the reactivation of virus-specific cytotoxic T cell and HLA-unrestricted killer cell responses. Virology. 1992 Dec;191(2):773–782. doi: 10.1016/0042-6822(92)90253-l. [DOI] [PubMed] [Google Scholar]
- Sunil-Chandra N. P., Efstathiou S., Arno J., Nash A. A. Virological and pathological features of mice infected with murine gamma-herpesvirus 68. J Gen Virol. 1992 Sep;73(Pt 9):2347–2356. doi: 10.1099/0022-1317-73-9-2347. [DOI] [PubMed] [Google Scholar]
- Sunil-Chandra N. P., Efstathiou S., Nash A. A. Interactions of murine gammaherpesvirus 68 with B and T cell lines. Virology. 1993 Apr;193(2):825–833. doi: 10.1006/viro.1993.1191. [DOI] [PubMed] [Google Scholar]
- Sunil-Chandra N. P., Efstathiou S., Nash A. A. Murine gammaherpesvirus 68 establishes a latent infection in mouse B lymphocytes in vivo. J Gen Virol. 1992 Dec;73(Pt 12):3275–3279. doi: 10.1099/0022-1317-73-12-3275. [DOI] [PubMed] [Google Scholar]
- Wenske E. A., Bratton M. W., Courtney R. J. Endo-beta-N-acetylglucosaminidase H sensitivity of precursors to herpes simplex virus type 1 glycoproteins gB and gC. J Virol. 1982 Oct;44(1):241–248. doi: 10.1128/jvi.44.1.241-248.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Heijne G. A new method for predicting signal sequence cleavage sites. Nucleic Acids Res. 1986 Jun 11;14(11):4683–4690. doi: 10.1093/nar/14.11.4683. [DOI] [PMC free article] [PubMed] [Google Scholar]