Abstract
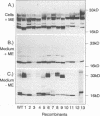
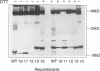
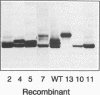
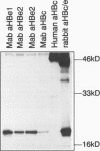
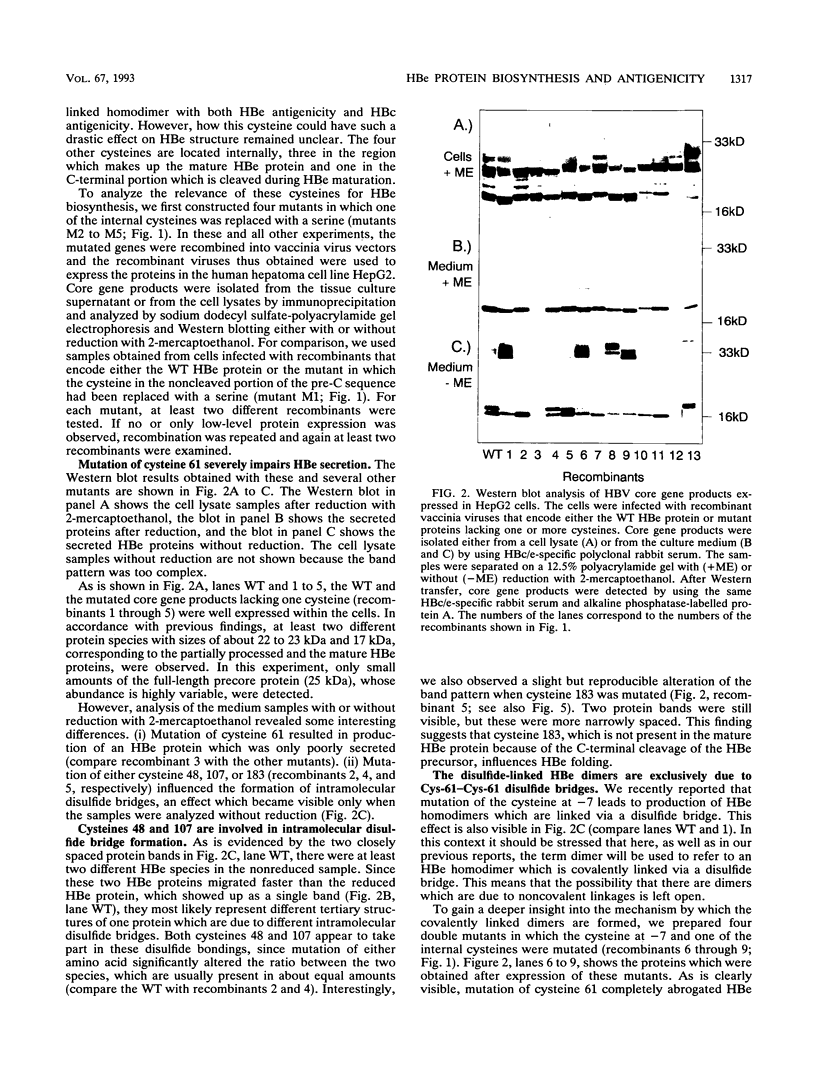
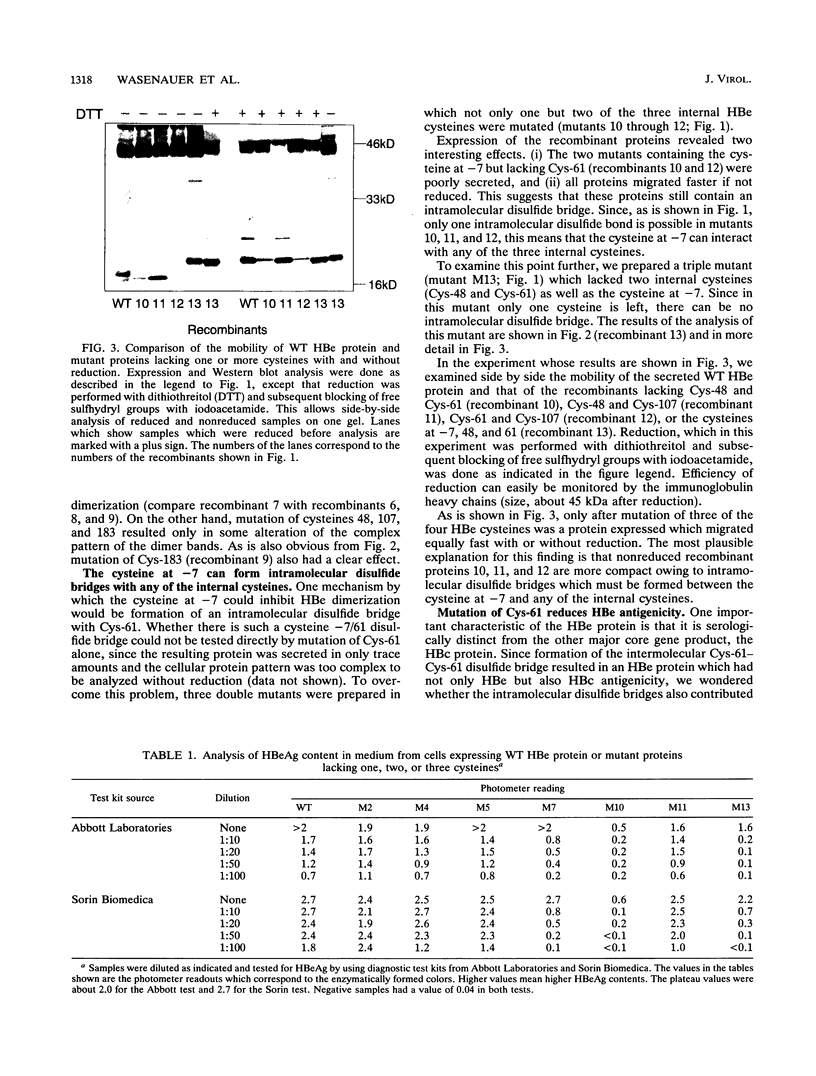
The mature form of the secretory core protein (HBe protein) of human hepatitis B virus contains four cysteines which are located at amino acid positions -7, 48, 61, and 107 relative to the HBc start methionine. In addition, there is a cysteine, Cys-183, located in the C-terminal domain of the HBe precursor, which is cleaved during HBe maturation. Here, the significance of these cysteines for biosynthesis and antigenicity of the HBe protein was examined. The cysteines at positions -7 and 61 were found to be crucial for HBe biosynthesis. As has already been described, if the Cys at position -7 is mutated, disulfide-linked HBe homodimers which have both HBe antigenicity and HBc antigenicity are expressed. Here we show that these dimers are due to Cys-61-Cys-61 disulfide bridges which are formed only if the Cys at position -7 is not present. In the wild-type protein, this dimerization appears to be inhibited by formation of intramolecular disulfide bridges between the Cys at -7 and one of the internal cysteines. Moreover, Cys-61 is important for HBe biosynthesis in general since mutation of this amino acid results in production of HBe proteins which are either only poorly secreted or possess a different antigenicity.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aden D. P., Fogel A., Plotkin S., Damjanov I., Knowles B. B. Controlled synthesis of HBsAg in a differentiated human liver carcinoma-derived cell line. Nature. 1979 Dec 6;282(5739):615–616. doi: 10.1038/282615a0. [DOI] [PubMed] [Google Scholar]
- Argos P., Fuller S. D. A model for the hepatitis B virus core protein: prediction of antigenic sites and relationship to RNA virus capsid proteins. EMBO J. 1988 Mar;7(3):819–824. doi: 10.1002/j.1460-2075.1988.tb02880.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferns R. B., Tedder R. S. Monoclonal antibodies to hepatitis Be antigen (HBeAg) derived from hepatitis B core antigen (HBcAg): their use in characterization and detection of HBeAg. J Gen Virol. 1984 May;65(Pt 5):899–908. doi: 10.1099/0022-1317-65-5-899. [DOI] [PubMed] [Google Scholar]
- Ganem D., Varmus H. E. The molecular biology of the hepatitis B viruses. Annu Rev Biochem. 1987;56:651–693. doi: 10.1146/annurev.bi.56.070187.003251. [DOI] [PubMed] [Google Scholar]
- Garcia P. D., Ou J. H., Rutter W. J., Walter P. Targeting of the hepatitis B virus precore protein to the endoplasmic reticulum membrane: after signal peptide cleavage translocation can be aborted and the product released into the cytoplasm. J Cell Biol. 1988 Apr;106(4):1093–1104. doi: 10.1083/jcb.106.4.1093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoofnagle J. H., Shafritz D. A., Popper H. Chronic type B hepatitis and the "healthy" HBsAg carrier state. Hepatology. 1987 Jul-Aug;7(4):758–763. doi: 10.1002/hep.1840070424. [DOI] [PubMed] [Google Scholar]
- Imai M., Nomura M., Gotanda T., Sano T., Tachibana K., Miyamoto H., Takahashi K., Toyama S., Miyakawa Y., Mayumi M. Demonstration of two distinct antigenic determinants on hepatitis B e antigen by monoclonal antibodies. J Immunol. 1982 Jan;128(1):69–72. [PubMed] [Google Scholar]
- Junker M., Galle P., Schaller H. Expression and replication of the hepatitis B virus genome under foreign promoter control. Nucleic Acids Res. 1987 Dec 23;15(24):10117–10132. doi: 10.1093/nar/15.24.10117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ou J. H., Laub O., Rutter W. J. Hepatitis B virus gene function: the precore region targets the core antigen to cellular membranes and causes the secretion of the e antigen. Proc Natl Acad Sci U S A. 1986 Mar;83(6):1578–1582. doi: 10.1073/pnas.83.6.1578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salfeld J., Pfaff E., Noah M., Schaller H. Antigenic determinants and functional domains in core antigen and e antigen from hepatitis B virus. J Virol. 1989 Feb;63(2):798–808. doi: 10.1128/jvi.63.2.798-808.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlicht H. J., Schaller H. The secretory core protein of human hepatitis B virus is expressed on the cell surface. J Virol. 1989 Dec;63(12):5399–5404. doi: 10.1128/jvi.63.12.5399-5404.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlicht H. J., Wasenauer G. The quaternary structure, antigenicity, and aggregational behavior of the secretory core protein of human hepatitis B virus are determined by its signal sequence. J Virol. 1991 Dec;65(12):6817–6825. doi: 10.1128/jvi.65.12.6817-6825.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Standring D. N., Ou J. H., Masiarz F. R., Rutter W. J. A signal peptide encoded within the precore region of hepatitis B virus directs the secretion of a heterogeneous population of e antigens in Xenopus oocytes. Proc Natl Acad Sci U S A. 1988 Nov;85(22):8405–8409. doi: 10.1073/pnas.85.22.8405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sällberg M., Rudén U., Magnius L. O., Harthus H. P., Noah M., Wahren B. Characterisation of a linear binding site for a monoclonal antibody to hepatitis B core antigen. J Med Virol. 1991 Apr;33(4):248–252. doi: 10.1002/jmv.1890330407. [DOI] [PubMed] [Google Scholar]
- Sällberg M., Rudén U., Wahren B., Noah M., Magnius L. O. Human and murine B-cells recognize the HBeAg/beta (or HBe2) epitope as a linear determinant. Mol Immunol. 1991 Jul;28(7):719–726. doi: 10.1016/0161-5890(91)90114-y. [DOI] [PubMed] [Google Scholar]
- Takahashi K., Machida A., Funatsu G., Nomura M., Usuda S., Aoyagi S., Tachibana K., Miyamoto H., Imai M., Nakamura T. Immunochemical structure of hepatitis B e antigen in the serum. J Immunol. 1983 Jun;130(6):2903–2907. [PubMed] [Google Scholar]
- Wasenauer G., Köck J., Schlicht H. J. A cysteine and a hydrophobic sequence in the noncleaved portion of the pre-C leader peptide determine the biophysical properties of the secretory core protein (HBe protein) of human hepatitis B virus. J Virol. 1992 Sep;66(9):5338–5346. doi: 10.1128/jvi.66.9.5338-5346.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]