Abstract
Exercise in the heat can pose a severe challenge to human cardiovascular control, and thus the provision of oxygen to exercising muscles and vital organs, because of enhanced thermoregulatory demand for skin blood flow coupled with dehydration and hyperthermia. Cardiovascular strain, typified by reductions in cardiac output, skin and locomotor muscle blood flow and systemic and muscle oxygen delivery accompanies marked dehydration and hyperthermia during prolonged and intense exercise characteristic of many summer Olympic events. This review focuses on how the cardiovascular system is regulated when exercising in the heat and how restrictions in locomotor skeletal muscle and/or skin perfusion might limit athletic performance in hot environments.
Exercise in the heat
The demands of dynamic exercise at intensities up to maximum oxygen consumption 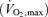 distill down to demands for blood flow. Blood flow to active muscle (and the myocardium) is required to meet the energetic demands for muscular activity (principally the demand for oxygen), while blood flow to skin is required to meet the demands of temperature regulation. These combined demands for blood flow can result in a competition for the available cardiac output (Rowell, 1974), which has, as a bottom line, a limit to the ability to meet the dual demands of exercise per se and of temperature regulation, particularly during intense dynamic exercise. The focus of this review pertains to how this limitation becomes manifest during exercise in the heat: reduced muscle blood flow, limited skin blood flow or both? Reduced muscle blood flow will limit the intensity and duration of exercise, while reduced skin blood flow will limit the disposal of heat resulting in adverse effects of elevated internal temperature, including that of the central nervous system. This topic, which has been periodically reviewed over the past 35 years or more (e.g. Thauer, 1965; Rowell, 1974; Brengelmann, 1983; Johnson, 1992; Kenny & Johnson, 1992; Rowell et al. 1996) has received renewed interest as some prior speculation has been replaced by data and the study of special populations has led to a further evolution of our understanding of the processes involved.
distill down to demands for blood flow. Blood flow to active muscle (and the myocardium) is required to meet the energetic demands for muscular activity (principally the demand for oxygen), while blood flow to skin is required to meet the demands of temperature regulation. These combined demands for blood flow can result in a competition for the available cardiac output (Rowell, 1974), which has, as a bottom line, a limit to the ability to meet the dual demands of exercise per se and of temperature regulation, particularly during intense dynamic exercise. The focus of this review pertains to how this limitation becomes manifest during exercise in the heat: reduced muscle blood flow, limited skin blood flow or both? Reduced muscle blood flow will limit the intensity and duration of exercise, while reduced skin blood flow will limit the disposal of heat resulting in adverse effects of elevated internal temperature, including that of the central nervous system. This topic, which has been periodically reviewed over the past 35 years or more (e.g. Thauer, 1965; Rowell, 1974; Brengelmann, 1983; Johnson, 1992; Kenny & Johnson, 1992; Rowell et al. 1996) has received renewed interest as some prior speculation has been replaced by data and the study of special populations has led to a further evolution of our understanding of the processes involved.
Blood flow to active muscle and cardiac output
The magnitude of the increase in blood flow to working muscle is potentially huge. Earlier estimates of 80 ml (100 g−1) min−1 as maximal muscle blood flow have been replaced by values severalfold greater (Laughlin & Armstrong, 1983; Andersen & Saltin, 1985; Rowell et al. 1986). Thus, blood flow as high as 250–400 ml (100 g−1) min−1 or more can occur when the exercise is limited to a small muscle mass. Maximal exercise, i.e. levels of whole-body dynamic exercise associated with 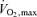 , however, cannot be associated with such high levels of blood flow if all or a large fraction of the total muscle mass is simultaneously recruited. For example, maximal aerobic exercise involving 15 kg of skeletal muscle (an estimate of the active muscle mass during maximal running exercise from a total arm and leg muscle mass of 25–28 kg) would require a cardiac output of almost 40 l min−1 to meet the degree of vasodilatation indicated above. This is well beyond the limits of cardiac pumping capacity in persons of average physical training, but might be approached by elite athletes. This suggests that the greater cardiac pumping capacity allows the athlete to achieve a greater fractional muscle recruitment, peak aerobic power production and
, however, cannot be associated with such high levels of blood flow if all or a large fraction of the total muscle mass is simultaneously recruited. For example, maximal aerobic exercise involving 15 kg of skeletal muscle (an estimate of the active muscle mass during maximal running exercise from a total arm and leg muscle mass of 25–28 kg) would require a cardiac output of almost 40 l min−1 to meet the degree of vasodilatation indicated above. This is well beyond the limits of cardiac pumping capacity in persons of average physical training, but might be approached by elite athletes. This suggests that the greater cardiac pumping capacity allows the athlete to achieve a greater fractional muscle recruitment, peak aerobic power production and 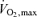 than sedentary individuals – assuming that muscle recruitment increases with power production within the full range of human exercise intensities.
than sedentary individuals – assuming that muscle recruitment increases with power production within the full range of human exercise intensities.
An important point to the above is that there is a significant disparity between the potential demand for blood flow by exercising muscle and the ability of the heart to meet that demand, even during brief exercise in which additional blood flow to skin is not a concern. Instead, circulatory control elements prevent muscle vascular conductance from reaching such high levels. Otherwise blood pressure could be severely threatened. Such a compromised blood pressure regulation is, in fact, seen in conditions wherein sympathetic vasoconstrictor function is compromised (see Puvi-Rajasingham et al. 1997; Krediet et al. 2004).
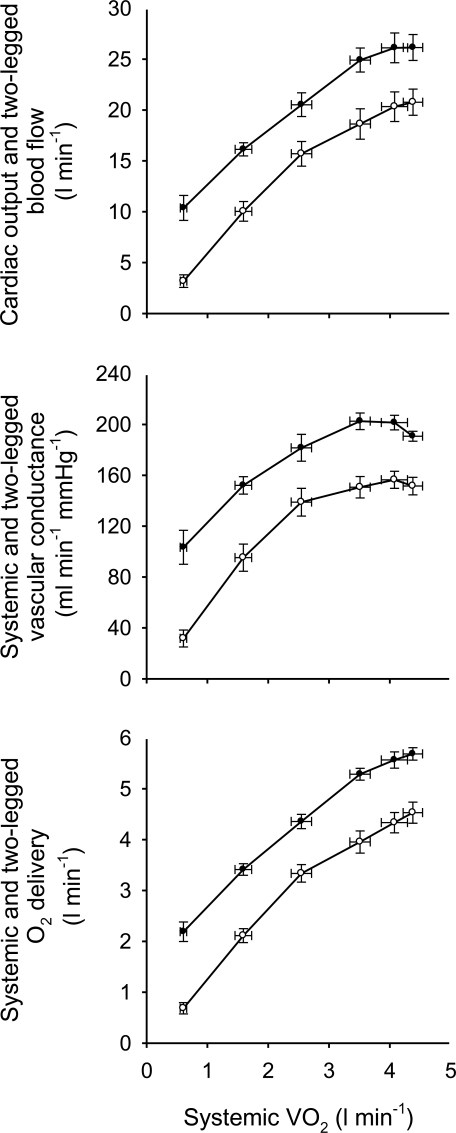
Both the baroreflex and metaboreflexes originating in active muscle individually have been implicated in this limitation to muscle vasodilatation during intense dynamic exercise (Rowell et al. 1996). In both cases, a neural reflex link is acting in competition with the local control mechanisms of functional hyperaemia to limit the increase in muscle blood flow with exercise, particularly when engaging a large muscle mass (e.g. whole-body exercise such as cycling, running or rowing). In support of this, Mortensen et al. (2005) showed an attenuation in the rate of rise in leg muscle blood flow and O2 delivery with increases in cycling exercise intensity above 50% of 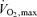 and a plateau in cardiac output above 90% of
and a plateau in cardiac output above 90% of 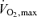 , accompanying a levelling off in limb muscle and systemic vascular conductance (see Fig. 1; Mortensen et al. 2005). They also showed in the same individuals that cardiac output and muscle blood flow increased linearly during incremental small muscle mass exercise to exhaustion (i.e. one-legged knee-extensor exercise). Together, these observations support the possibility that the restrictions in active muscle blood flow might be at least in part related to an insufficient cardiac output as the limits of cardiac pumping capacity are reached.
, accompanying a levelling off in limb muscle and systemic vascular conductance (see Fig. 1; Mortensen et al. 2005). They also showed in the same individuals that cardiac output and muscle blood flow increased linearly during incremental small muscle mass exercise to exhaustion (i.e. one-legged knee-extensor exercise). Together, these observations support the possibility that the restrictions in active muscle blood flow might be at least in part related to an insufficient cardiac output as the limits of cardiac pumping capacity are reached.
Figure 1. Systemic and exercising legs haemodynamics during incremental cycling to exhaustion.
Note the attenuation in the rate of rise in blood flow and O2 delivery to the exercising legs above 50% 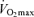 and the plateau in cardiac output above 90% of
and the plateau in cardiac output above 90% of 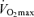 (Mortensen et al. 2005). Filled symbols depict the cardiac output, systemic vascular conductance and systemic O2 delivery for the upper, middle, and lower panels, respectively. Open symbols depict the blood flow, vascular conductance and O2 delivery to the exercising legs for the respective panels.
(Mortensen et al. 2005). Filled symbols depict the cardiac output, systemic vascular conductance and systemic O2 delivery for the upper, middle, and lower panels, respectively. Open symbols depict the blood flow, vascular conductance and O2 delivery to the exercising legs for the respective panels.
In the context of exercise in a hot environment, the above development raises the consideration that other reflexes, notably thermoregulatory reflexes and associated increases in skin blood flow, might also contribute to limiting skeletal muscle blood flow. In humans, body heating at rest causes significant vasoconstriction in the splanchnic and renal circulations (see Rowell, 1974). The sympathetically mediated thermoregulatory reduction in visceral blood flows also occurs during exercise in the heat. There is also an increased sympathetic nerve activity directed to skeletal muscle in resting hyperthermic humans, with perhaps a modest reduction in blood flow to resting muscle (Rowell, 1974; Niimi et al. 1997; Keller et al. 2006). In the present context, it is interesting that the stimulation of muscle sympathetic nerve activity by mechanosensitive afferents is exaggerated if the muscle is warmed by 4°C (Ray & Gracey, 1997).
Thus, the requisite elements appear to be in place for thermoregulatory reflexes to limit blood flow to active skeletal muscle during exercise in the heat: (i) generally increased sympathetic vasoconstrictor nerve activity with hyperthermia, (ii) evidence that other reflexes can limit the increase in active skeletal muscle blood flow, and (iii) a large disparity between the ability of the heart to pump sufficient volume of blood and the potential capacity for vascular conductance in active muscle. In this third point, adding cutaneous vasodilatation to the mix compounds that problem.
In keeping with that possibility, Ekelund (1967) found cardiac output to be quite stable between 10 and 60 min of exercise, while in a separate series, skin blood flow progressively increased over that same time interval (Johnson & Rowell, 1975). Extrapolation of that increased skin blood flow from the forearm to the entire body surface area would represent an increase of around 3 l min−1. Such a redistribution of blood flow would not be possible through further vasoconstriction in visceral vascular beds. Indeed, the logical source for blood flow redistribution to skin would be active skeletal muscle. A small reduction in skeletal muscle blood flow, relative to the level early in exercise, would represent a substantial redistribution to skin, relative to early levels in that region.
The notion that thermoregulatory vasodilatation in skin causes a reflex vasoconstriction in active muscle was tested directly in several studies (Savard et al. 1988; Nielsen et al. 1990, 1993; González-Alonso et al. 1998). The consistent conclusion was that even extreme body heating had no measurable effect on blood flow to active muscle. This was true when the exercise was limited to a small muscle group (Savard et al. 1988), but was also found to be the case when more muscle groups were active (Nielsen et al. 1990, 1993; González-Alonso et al. 1998). It was also seen to be the case when low intensity cycling exercise was performed in the heat and the cardiovascular system responded adequately to the additional demand for an elevated skin blood flow by increasing cardiac output by 1–3 l min−1 and reducing visceral blood flow (Savard et al. 1988; Nielsen et al. 1990, 1993).
Therefore, when exercise is performed in the heat and skin and muscle are competing for blood flow, muscle wins; thermoregulatory demands do not lead to a diversion of blood flow from active muscle acutely. However, it is also the case that hyperthermia can significantly affect active muscle blood flow and performance negatively in several ways. When exercise in a hot environment is prolonged without fluid replenishment and thus significant dehydration develops, progressive reductions in cardiac output and arterial pressure accompany elevations in systemic vascular resistance, cutaneous vascular resistance (despite much higher internal temperature) and plasma noradrenaline levels (González-Alonso et al. 1995, 1998, 1999b). The reduction in cardiac output of nearly 4 l min−1 is accompanied by a reduction in active muscle blood flow; however, reduced blood flow to active muscle does not appear to be the primary source of fatigue in prolonged moderate exercise in the heat in as much as muscle O2 delivery is for the most part maintained (due to a parallel increase in O2 content with dehydration-induced haemoconcentration) and active muscle 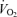 only tends to be depressed at exhaustion (González-Alonso et al. 1998). Instead, a second, central neural contributor appears to be operative. This conclusion follows from studies of exercise in warm to hot environments with varying degrees of initial internal temperatures and rates of heat storae (e.g. González-Alonso et al. 1999a). The common feature to the studies was that fatigue occurred at the same internal temperature (slightly above 40°C), regardless of the initial core temperature, the rate of heat storage or the level of mean skin temperature ranging from 35 to 38°C. This feature was seen typically with higher than normal skin temperature. What effect, if any, of cooler (or warmer) skin temperature on the internal temperature at fatigue is unknown. The observation of a similar upper level of internal temperature during uncompensable hot environments is interpreted as an indication that the limit to submaximal exercise performance (fatigue) in hyperthermia is the temperature of central nervous structures, an interpretation supported by EEG findings in conditions of heat stress, exercise and fatigue (Nielsen et al. 2001; Nybo & Nielsen, 2001; Nielsen & Nybo, 2003).
only tends to be depressed at exhaustion (González-Alonso et al. 1998). Instead, a second, central neural contributor appears to be operative. This conclusion follows from studies of exercise in warm to hot environments with varying degrees of initial internal temperatures and rates of heat storae (e.g. González-Alonso et al. 1999a). The common feature to the studies was that fatigue occurred at the same internal temperature (slightly above 40°C), regardless of the initial core temperature, the rate of heat storage or the level of mean skin temperature ranging from 35 to 38°C. This feature was seen typically with higher than normal skin temperature. What effect, if any, of cooler (or warmer) skin temperature on the internal temperature at fatigue is unknown. The observation of a similar upper level of internal temperature during uncompensable hot environments is interpreted as an indication that the limit to submaximal exercise performance (fatigue) in hyperthermia is the temperature of central nervous structures, an interpretation supported by EEG findings in conditions of heat stress, exercise and fatigue (Nielsen et al. 2001; Nybo & Nielsen, 2001; Nielsen & Nybo, 2003).
In contrast to low intensity exercise (where active muscle 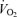 is preserved and core hyperthermia is the primary issue), fatigue during high-intensity exercise is preceded by significant restrictions in muscle metabolism (González-Alonso & Calbet, 2003; Mortensen et al. 2005). In a hot environment, the lower
is preserved and core hyperthermia is the primary issue), fatigue during high-intensity exercise is preceded by significant restrictions in muscle metabolism (González-Alonso & Calbet, 2003; Mortensen et al. 2005). In a hot environment, the lower 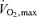 and the subsequent early fatigue are indeed strongly associated with an accelerated blunting in active muscle aerobic energy production owing to the reduced blood flow and O2 delivery. Importantly, vascular conductance in the active legs is not reduced; rather, blood flow is reduced secondary to a falling blood pressure at workloads equivalent to
and the subsequent early fatigue are indeed strongly associated with an accelerated blunting in active muscle aerobic energy production owing to the reduced blood flow and O2 delivery. Importantly, vascular conductance in the active legs is not reduced; rather, blood flow is reduced secondary to a falling blood pressure at workloads equivalent to 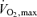 (González-Alonso & Calbet, 2003). This also suggests that there is little or no role for actual vasoconstriction of active muscle in this reduced blood flow, but it is a passive event brought on more rapidly in a hot environment. Hence, although thermoregulatory reflexes apparently do not significantly reduce blood flow to active muscle, hyperthermia does act directly or indirectly to limit the intensity and duration of exercise.
(González-Alonso & Calbet, 2003). This also suggests that there is little or no role for actual vasoconstriction of active muscle in this reduced blood flow, but it is a passive event brought on more rapidly in a hot environment. Hence, although thermoregulatory reflexes apparently do not significantly reduce blood flow to active muscle, hyperthermia does act directly or indirectly to limit the intensity and duration of exercise.
Skin circulation
The general control scheme for the skin circulation includes a variety of thermoregulatory and non-thermoregulatory reflexes (see Johnson & Proppe, 1996). The former include skin and internal (principally brain) temperatures and the latter include blood pressure control and the responses to exercise, including central command, muscle metaboreceptor stimulation and perhaps muscle mechanoreceptor stimulation (Shibasaki et al. 2005). These reflex controllers are subject to modification by a variety of other conditions. The efferent portion of neural control is brought about by noradrenergic vasoconstrictor nerves in both glabrous and hairy skin and, in hairy skin, by a sympathetic active vasodilator system that involves cholinergic cotransmitters (Kellogg et al. 1995; Johnson & Proppe, 1996).
The potential for overall cutaneous vascular conductance is second only to skeletal muscle in the degree of vasodilatation that can be and frequently is achieved in healthy humans. As mentioned above, significant body heat stress leads both to large increases in cardiac output and to reductions in vascular conductance to splanchnic and renal circulations (Rowell, 1974). Combined, this increase in cardiac output and the redistribution from other regions lead to a maximal skin blood flow of around 8 l min−1. Some of that increase is via withdrawal of vasoconstrictor activity. In non-glabrous skin, most of the vasodilatation is due to activation of the vasodilator system. Any reflex modification by exercise of thermoregulatory control of the cutaneous circulation must occur through an effect on one or both of those sympathetic systems.
When dynamic exercise is performed in a hot environment, the thermoregulatory demands to increase skin blood flow and the metabolic demands to increase muscle blood flow would both be high – so high that the limits to cardiac pumping capacity and the limits to further redistribution would usually fall short of meeting the demands for both. The consequence is either (a) a mismatch between cardiac output and total vascular conductance and a loss of blood pressure, or (b) a mismatch between muscle blood flow and metabolism leading to a reduction in muscle 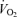 , or (c) a compromised skin blood flow resulting in higher body temperature. The possibility of reduced muscle blood flow was discussed above and the evidence does not support a reflex thermoregulatory reduction in active muscle blood flow. Although blood pressure may suffer somewhat during exercise in the heat, it is generally maintained at reasonable levels (Rowell, 1974), suggesting that the third option of a compromised skin circulation is the key to understanding the circulatory consequences of work in a hot environment.
, or (c) a compromised skin blood flow resulting in higher body temperature. The possibility of reduced muscle blood flow was discussed above and the evidence does not support a reflex thermoregulatory reduction in active muscle blood flow. Although blood pressure may suffer somewhat during exercise in the heat, it is generally maintained at reasonable levels (Rowell, 1974), suggesting that the third option of a compromised skin circulation is the key to understanding the circulatory consequences of work in a hot environment.
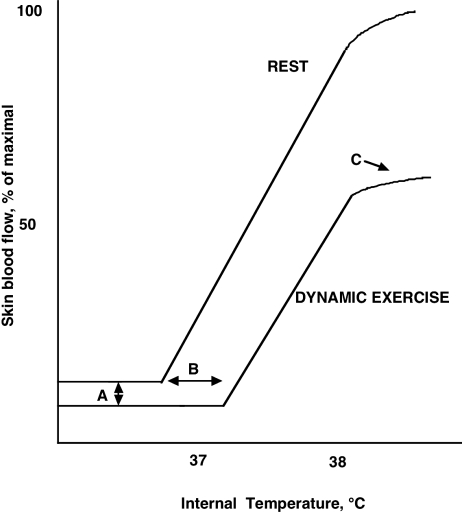
Dynamic exercise is known to affect skin blood flow through several modifications of the vasoconstrictor and active vasodilator systems (Johnson, 1992; Kenney & Johnson, 1992). At the onset of exercise, there is an initial cutaneous vasoconstriction that occurs both in normothermic conditions and in hyperthermia (see Fig. 2). In both cases, this acute reduction in skin blood flow is brought about by increased vasoconstrictor system activity. As exercise continues, especially in the heat, body temperature rises and the vasodilator system is activated. This second role for exercise arises from its thermogenic nature, but the degree of vasodilatation is tempered in two ways. First, the threshold internal temperature at which vasodilatation begins is shifted to a higher level during exercise relative to rest (Kellogg et al. 1991; Smolander et al. 1991; Johnson, 1992; Kenney & Johnson, 1992). This is accomplished by a delay in initiation of active vasodilatation (Kellogg et al. 1991), resulting in skin blood flow during exercise being lower than at rest for the same thermal conditions (Fig. 2). The core temperature threshold for the initial increase in skin blood flow is dependent on the level of exercise. Secondly, as exercise in the heat continues, internal temperature continues to rise steadily, but skin blood flow reaches an apparent upper limit at an internal temperature of around 38°C (Brengelmann et al. 1977; González-Alonso et al. 1999a). Above that temperature, the rate of rise in skin blood flow is sharply curtailed, in some cases failing to increase further despite an additional increase in internal temperature of 1°C or more. This occurs at a skin blood flow estimated to be around 50% of maximal whereas, at rest, blood flow would continue to rise with internal temperature until a true maximal level was achieved. This upper limit is apparently achieved by exercise limiting active vasodilator system activity because pharmacological blockade of the vasoconstrictor system tended to make the plateau phenomenon more apparent (Kellogg et al. 1993).
Figure 2. Schematic description of the thermoregulatory control of skin blood flow as modified by moderately intense exercise.
The relation of skin blood flow to internal temperature is affected, relative to resting conditions, in at least three ways by exercise: a vasoconstrictor response at the onset of dynamic exercise (A), an increase in the internal temperature threshold at which skin blood flow begins to increase (B), and a levelling off, or plateau, in skin blood flow above an internal temperature of 38°C at a level well below maximal (C). Exercise exerts these effects through the vasoconstrictor system for the initial vasoconstriction and through inhibiting the active vasodilator system for the increased threshold and for the plateau. At rest, the plateau only occurs as skin blood flow approaches maximal vasodilatation.
To summarize, dynamic exercise has a collection of effects on the skin circulation, most of which act to keep skin blood flow below levels that would be expected on the basis of thermoregulatory control alone. The implication is that, while the metabolic demands of the active muscle may be met, the consequence is that the regulation of body temperature will suffer. This does, in fact, appear to be the case during light exercise. As discussed above, the limiting factor for continuing exercise in the heat at low intensity is not compromised muscle blood flow, nor reduced blood pressure, but mainly the elevation in central temperature. However, during intense exercise typical of summer sport competitions including the Olympic Games, significant cardiovascular strain accompanies hyperthermia and, as will be discussed below, the combination of both stressors plays a major role in the development of fatigue.
Cardiovascular limitations to athletic performance
The summer Olympic Games includes a wide array of events that can generally be classified on the basis of the relative 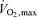 intensity as submaximal, maximal and supramaximal and on the basis of their duration as endurance, middle distance and sprinting events. The focus of this section is on how restrictions in locomotor skeletal muscle and/or skin perfusion with dehydration and/or hyperthermia might limit performance in the endurance, middle distance and sprinting athlete.
intensity as submaximal, maximal and supramaximal and on the basis of their duration as endurance, middle distance and sprinting events. The focus of this section is on how restrictions in locomotor skeletal muscle and/or skin perfusion with dehydration and/or hyperthermia might limit performance in the endurance, middle distance and sprinting athlete.
The cardiovascular challenge to the endurance athlete
The endurance athlete performs at intensities below 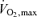 (∼65–85 ml kg−1 min−1) in events lasting from > 10 min to several hours. For instance, elite marathoners can run the 42.2 km marathon race in ∼2.2 h at speeds eliciting 80–90% of
(∼65–85 ml kg−1 min−1) in events lasting from > 10 min to several hours. For instance, elite marathoners can run the 42.2 km marathon race in ∼2.2 h at speeds eliciting 80–90% of 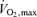 whereas elite cyclists can complete a 47 km time-trial in ∼1 h at 85–95% of
whereas elite cyclists can complete a 47 km time-trial in ∼1 h at 85–95% of 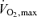 . To sustain these astonishing exercise intensities for 1–2 h, the heart needs to continuously pump 22–27 l min−1 of blood, the locomotor muscles to produce 19–24 kcal min−1 of energy and, as discussed above, the thermoregulatory system to dissipate the excess heat that is not converted into mechanical power. The regulatory demands on the cardiovascular, thermoregulatory, metabolic, nervous and endocrine systems to sustain such extreme responses during competition are undoubtedly very severe, particularly if endurance athletes have to cope with the stress of dehydration and hyperthermia.
. To sustain these astonishing exercise intensities for 1–2 h, the heart needs to continuously pump 22–27 l min−1 of blood, the locomotor muscles to produce 19–24 kcal min−1 of energy and, as discussed above, the thermoregulatory system to dissipate the excess heat that is not converted into mechanical power. The regulatory demands on the cardiovascular, thermoregulatory, metabolic, nervous and endocrine systems to sustain such extreme responses during competition are undoubtedly very severe, particularly if endurance athletes have to cope with the stress of dehydration and hyperthermia.
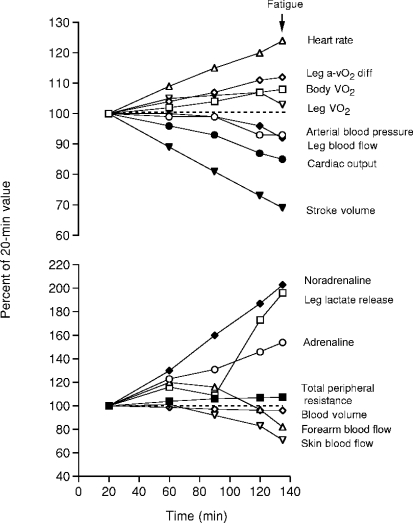
Summer sport competitions normally take place in environmental conditions favouring evaporative cooling where skin temperatures are kept around 31–32°C instead of 37–38°C or higher as seen with severe heat stress. In these conditions, dehydration rather than heat stress accounts for the majority of the cardiovascular, thermoregulatory and metabolic strain that endurance athletes may experience. Well-controlled laboratory studies have consistently demonstrated that progressive dehydration to 4% of body weight loss during moderate exercise in the heat results in gradual reductions in systemic, muscle and skin blood flow, progressive increases in core and muscle temperature, greater reliance on muscle glycogen and cellular metabolism and a trend for a suppressed exercising muscle 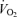 at exhaustion (Sawka et al. 1979; Montain & Coyle, 1992; Hargreaves et al. 1996; González-Alonso et al. 1998, 1999b). Figure 3 illustrates the marked cardiovascular and metabolic strain and the augmented vasoconstrictor sympathetic drive preceding exhaustion with dehydration and hyperthermia. Therefore, dehydration and concomitant hyperthermia can lead to early fatigue in the endurance athlete, partly by reducing locomotor muscle blood flow, secondary to reductions in cardiac output and perfusion pressure, and by reducing O2 supply and oxidative metabolism.
at exhaustion (Sawka et al. 1979; Montain & Coyle, 1992; Hargreaves et al. 1996; González-Alonso et al. 1998, 1999b). Figure 3 illustrates the marked cardiovascular and metabolic strain and the augmented vasoconstrictor sympathetic drive preceding exhaustion with dehydration and hyperthermia. Therefore, dehydration and concomitant hyperthermia can lead to early fatigue in the endurance athlete, partly by reducing locomotor muscle blood flow, secondary to reductions in cardiac output and perfusion pressure, and by reducing O2 supply and oxidative metabolism.
Figure 3. Effects of progressive dehydration and hyperthermia on cardiovascular haemodynamics, metabolism and circulating catecholamines during exhaustive prolonged cycling in the heat.
The seven endurance-trained, heat acclimated subjects (VO2max = 4.9 ± 0.61 min−1) became exhausted after 135 ± 4 min of cycling at 208 ± 21 W in a 35°C, 40–50% relative humidity and 2–3 m s−1 wind speed environment with skin, core and exercising muscle temperatures reaching ∼35, ∼40 and ∼41°C, respectively. (From González-Alonso 2007 with permission, which was redrawn from González-Alonso et al. 1998, 1999). A week after the dehydration trial, the subjects performed a control trial in the heat for the same duration where cardiovascular, metabolic and thermoregulatory haemostasis was maintained throughout exercise by replacing fluid losses with fluid ingestion (data not shown).
The reduction in stroke volume and cardiac output beyond the regulatory adjustments to increases in blood oxygen content will result in a fall in systemic oxygen delivery. The lower stroke volume could therefore be a critical factor in the development of fatigue with dehydration and concomitant hyperthermia during exercise in the heat (Montain & Coyle, 1992; González-Alonso et al. 1997, 1999c, 2000). Interestingly, when thermoregulatory demands of exercise are minimized in a cold environment and the rise in core temperature is blunted, the decline in stroke volume and the increase in heart rate are significantly attenuated. In these conditions the significant drop in cardiac output normally observed with dehydration and hyperthermia in the heat is prevented (González-Alonso et al. 2000). Plasma volume expansion in dehydrated and normothermic subjects completely reverses the otherwise small reduction in stroke volume during exercise in the cold, despite the persistent 3–4 l extravascular dehydration (González-Alonso et al. 1997). Similarly, hyperthermia without dehydration induces a small decline in stroke volume during exercise in the heat, which is associated with an increased heart rate, whereas preventing dehydration and hyperthermia via fluid ingestion fully ameliorates these alterations in both stroke volume and heart rate (González-Alonso et al. 1995, 1997, 1998). Thus, the reductions in cardiac stroke volume that could ultimately set a limit to local muscle perfusion and O2 supply in the dehydrated and hyperthermic endurance athlete might be largely related to diminished intravascular volume (or related haematological changes) and the hyperthermia-induced tachycardia.
The cardiovascular challenge to the middle distance and sprinting athlete
The middle distance athletes perform at work rates equal to or slightly above 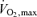 in events lasting from ∼2 to 10 min whereas the sprinting athlete competes in events lasting from 6–10 s to ∼1 min at intensities requiring up to ∼2.5 times the energy equivalent of
in events lasting from ∼2 to 10 min whereas the sprinting athlete competes in events lasting from 6–10 s to ∼1 min at intensities requiring up to ∼2.5 times the energy equivalent of 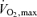 (Bangsbo, 1998; Weyand & Bundle, 2005). The number of human studies of the cardiovascular, thermoregulatory and metabolic responses to maximal and supramaximal whole-body exercise is very limited due principally to methodological limitations. However, it is reasonable to think that the maintenance of cardiovascular function and intracellular metabolism during such competition is at least as important for the middle distance as the endurance athlete. For instance, the capacity to maintain maximal systemic and locomotor muscle perfusion, O2 supply and
(Bangsbo, 1998; Weyand & Bundle, 2005). The number of human studies of the cardiovascular, thermoregulatory and metabolic responses to maximal and supramaximal whole-body exercise is very limited due principally to methodological limitations. However, it is reasonable to think that the maintenance of cardiovascular function and intracellular metabolism during such competition is at least as important for the middle distance as the endurance athlete. For instance, the capacity to maintain maximal systemic and locomotor muscle perfusion, O2 supply and 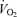 is critical for optimal performance during middle distance races (e.g. 800 and 1500 m in Athletics). In the present context of exercise in the heat, it is noteworthy that superimposition of severe heat stress and dehydration upset cardiovascular function and locomotor muscle metabolism during maximal exercise (Nybo et al. 2001; González-Alonso & Calbet, 2003; González-Alonso et al. 2004).
is critical for optimal performance during middle distance races (e.g. 800 and 1500 m in Athletics). In the present context of exercise in the heat, it is noteworthy that superimposition of severe heat stress and dehydration upset cardiovascular function and locomotor muscle metabolism during maximal exercise (Nybo et al. 2001; González-Alonso & Calbet, 2003; González-Alonso et al. 2004).
During events performed at intensities eliciting 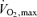 , the superimposition of heat stress causes an earlier decline in exercising muscle blood flow, presumably due to lower cardiac output and perfusion pressures, leading to a more rapid suppression in systemic and locomotor muscles O2 delivery and
, the superimposition of heat stress causes an earlier decline in exercising muscle blood flow, presumably due to lower cardiac output and perfusion pressures, leading to a more rapid suppression in systemic and locomotor muscles O2 delivery and 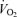 compared to non-heat stressed conditions (Figs 4 and 5; González-Alonso & Calbet, 2003; González-Alonso et al. 2004). It therefore seems that heat stress more quickly pushes the cardiovascular system to its regulatory limit, where cardiac output and subsequently perfusion pressure cannot be maintained resulting in reduced muscle blood flow. Importantly, fatigue during maximal exercise does not appear to be associated with an impaired brain aerobic metabolism, even though brain blood flow and O2 delivery decline. Rather, brain aerobic metabolism is enhanced because the increase in O2 extraction outstrips the decline in blood flow (Fig. 4) (González-Alonso et al. 2004). Hence, the reductions in O2 delivery to and aerobic metabolism in primary locomotor muscles could also be a crucial component in the development of fatigue for the middle distance athlete.
compared to non-heat stressed conditions (Figs 4 and 5; González-Alonso & Calbet, 2003; González-Alonso et al. 2004). It therefore seems that heat stress more quickly pushes the cardiovascular system to its regulatory limit, where cardiac output and subsequently perfusion pressure cannot be maintained resulting in reduced muscle blood flow. Importantly, fatigue during maximal exercise does not appear to be associated with an impaired brain aerobic metabolism, even though brain blood flow and O2 delivery decline. Rather, brain aerobic metabolism is enhanced because the increase in O2 extraction outstrips the decline in blood flow (Fig. 4) (González-Alonso et al. 2004). Hence, the reductions in O2 delivery to and aerobic metabolism in primary locomotor muscles could also be a crucial component in the development of fatigue for the middle distance athlete.
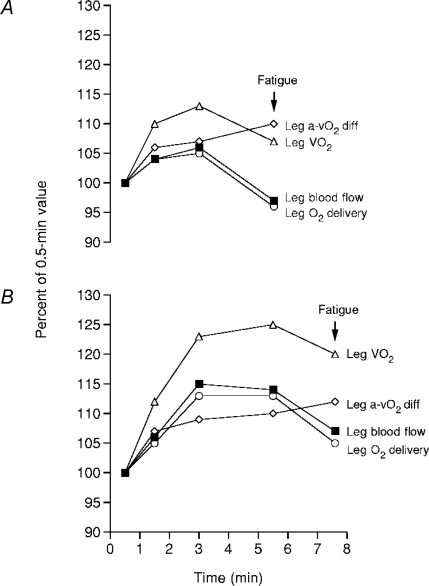
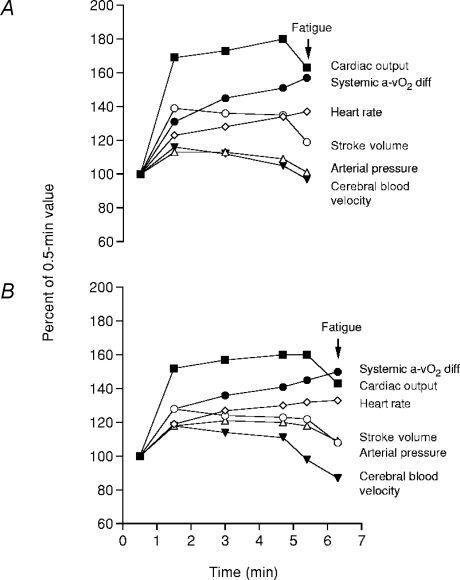
Figure 4. Effects of heat stress (A) on leg haemodynamics during maximal cycling compared to control (B) conditions.
Time to fatigue while cycling at 356 ± 14 W was diminished in heat stress compared to control (5.5 ± 0.2 versus 7.6 ± 0.2 min, respectively). Note that fatigue in both conditions was preceded by reductions in leg blood flow, O2 delivery and 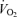 . Heat stress also reduced
. Heat stress also reduced 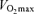 from 4.7 ± 0.2 to 4.3 ± 0.2 l min−1 despite equal maximal heart rates. Redrawn from González-Alonso & Calbet (2003).
from 4.7 ± 0.2 to 4.3 ± 0.2 l min−1 despite equal maximal heart rates. Redrawn from González-Alonso & Calbet (2003).
Figure 5. Effects of heat stress (A) on systemic haemodynamics and cerebral circulation during maximal exercise compared to control (B) conditions.
Time to fatigue while cycling at 360 ± 10 W was diminished in heat stress compared to control (5.8 ± 0.2 versus 7.5 ± 0.4 min, respectively). Note that fatigue in both conditions was preceded by reductions in cardiac output, stroke volume, arterial blood pressure and middle cerebral arterial blood velocity. Redrawn from González-Alonso et al. (2004).
An intriguing phenomenon in sprinting (supramaximal) events is the progressive decline in speed during competition. The universal fact is that the winner is the athlete who slows down least, not the one who starts the fastest. Knowledge of the mechanisms underlying this phenomenon is essentially nonexistent, in spite of the fact that many of the Olympic sports involve exercise in this intensity domain. Thermal stress is normally not a problem during supramaximal exercise, unless the sprinting athlete starts competition extremely dehydrated and hyperthermic. In this exercise domain, hyperthermia (∼39–40°C in core and muscle temperature) might impair repeated sprinting performance, but not a single 15 s supramaximal bout (Drust et al. 2005). This suggests that the effect of hyperthermia on fatigue is indirect, possibly via its effect on cardiovascular function. In this regard, repeated sprint performance in the hyperthermic state was associated with a lower whole-body 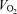 after repeated bouts, suggesting that locomotor muscle perfusion and O2 supply are blunted when sprinting exercise is repeated (Drust et al. 2005).
after repeated bouts, suggesting that locomotor muscle perfusion and O2 supply are blunted when sprinting exercise is repeated (Drust et al. 2005).
Summary and future directions
High level dynamic exercise, by itself, engages a complex collection of local metabolic and reflex adjustments to enable the active muscle to meet its energetic requirements. In short-term, high intensity sprint exercise, such adjustments are essentially limited to meeting those energetic needs, although the understanding of the cardiovascular dynamics in that setting is far from complete. As dynamic exercise and heat production continue, thermoregulatory demands add to the problem of integrative control, such that the demands of elevated body temperature for increasing skin blood flow are in competition with the metabolic demands to increase blood flow to active muscle. At the Olympic level, or any highly competitive level, that combination typically outstrips the pumping capacity of the heart. In the relative short-term, before there has been much fluid loss, blood flow to locomotor muscles is unimpaired, while there are clear limits on the ability to increase skin blood flow to the levels consistent with the thermoregulatory demands. Although the precise mechanisms of that limitation are not currently clear, the result of impaired temperature regulation is clear. In this scenario, the increased heat production, coupled with the limits to heat loss, can promote marked elevations in internal temperature, depending on the environmental conditions. This increased internal temperature, by its primary effects on the central nervous system, limits the ability to continue exercise; i.e. fatigue is not the result of limited perfusion of muscle or of falling blood pressure, but of high brain temperature. However, if exercise is of longer duration and if fluids lost through sweating are not replaced, the ensuing dehydration brings into play further problems of cardiovascular control. Again, the precise mechanisms of the effects of dehydration are the subject of continuing investigation, but the consequence is that the perfusion of active muscle, as well as skin, becomes limited. In this case, fatigue is a more complex issue of the combination of high body temperatures and altered distribution and osmolar makeup of body fluid volumes.
Acknowledgments
The authors' work has been supported by the Marie Curie Research Training programme, Gatorade Sports Science Institute, Team Denmark, Danish National Research Foundation (J.G.-A.), and National Institutes of Health (HL61388 and HL84072 (C.G.C.), and HL59166 (J. M. J.)).
References
- Andersen P, Saltin B. Maximal perfusion of skeletal muscle in man. J Physiol. 1985;366:233–249. doi: 10.1113/jphysiol.1985.sp015794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bangsbo J. Quantification of anaerobic energy production during intense exercise. Med Sci Sports Exerc. 1998;30:47–52. doi: 10.1097/00005768-199801000-00007. [DOI] [PubMed] [Google Scholar]
- Brengelmann GL. Circulatory adjustments to exercise and heat stress. Annu Rev Physiol. 1983;45:191–212. doi: 10.1146/annurev.ph.45.030183.001203. [DOI] [PubMed] [Google Scholar]
- Brengelmann GL, Johnson JM, Hermansen L, Rowell LB. Altered control of skin blood flow during exercise at high internal temperatures. J Appl Physiol. 1977;43:790–794. doi: 10.1152/jappl.1977.43.5.790. [DOI] [PubMed] [Google Scholar]
- Drust B, Rasmussen P, Mohr M, Nielsen B, Nybo L. Elevations in core and muscle temperature impair repeated sprint performance. Acta Physiol Scand. 2005;183:181–190. doi: 10.1111/j.1365-201X.2004.01390.x. [DOI] [PubMed] [Google Scholar]
- Ekelund LG. Circulatory and respiratory adaptation during prolonged exercise. Acta Physiol Scand. 1967;70(Suppl. 68):5–38. [PubMed] [Google Scholar]
- González-Alonso J. Hyperthermia impairs brain, heart and muscle function in exercising humans. Sports Med. 2007;37:371–373. doi: 10.2165/00007256-200737040-00025. [DOI] [PubMed] [Google Scholar]
- González-Alonso J, Calbet JA. Reductions in systemic and skeletal muscle blood flow and oxygen delivery limit maximal aerobic capacity in humans. Circulation. 2003;107:824–830. doi: 10.1161/01.cir.0000049746.29175.3f. [DOI] [PubMed] [Google Scholar]
- González-Alonso J, Calbet JAL, Nielsen B. Muscle blood flow is reduced with dehydration during prolonged exercise in humans. J Physiol. 1998;513:895–905. doi: 10.1111/j.1469-7793.1998.895ba.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- González-Alonso J, Calbet JAL, Nielsen B. Metabolic and thermodynamic responses to dehydration-induced reductions in muscle blood flow in exercising humans. J Physiol. 1999b;520:577–589. doi: 10.1111/j.1469-7793.1999.00577.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- González-Alonso J, Dalsgaard MK, Osada T, Volianitis S, Dawson EA, Yoshiga CC, Secher NH. Brain and central haemodynamics and oxygenation during maximal exercise in humans. J Physiol. 2004;557:331–342. doi: 10.1113/jphysiol.2004.060574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- González-Alonso J, Mora-Rodríguez R, Below PR, Coyle EF. Dehydration reduces cardiac output and increases systemic and cutaneous vascular resistance during exercise. J Appl Physiol. 1995;79:1487–1496. doi: 10.1152/jappl.1995.79.5.1487. [DOI] [PubMed] [Google Scholar]
- González-Alonso J, Mora-Rodríguez R, Coyle EF. Dehydration markedly impairs cardiovascular function in hyperthermic endurance athletes during exercise. J Appl Physiol. 1997;82:1229–1236. doi: 10.1152/jappl.1997.82.4.1229. [DOI] [PubMed] [Google Scholar]
- González-Alonso J, Mora-Rodríguez R, Coyle EF. Supine exercise restores arterial blood pressure and skin blood flow despite dehydration and hyperthermia. Am J Physiol Heart Circ Physiol. 1999c;277:H576–H583. doi: 10.1152/ajpheart.1999.277.2.H576. [DOI] [PubMed] [Google Scholar]
- González-Alonso J, Mora-Rodríguez R, Coyle EF. Stroke volume during exercise: interaction of environment and hydration. Am J Physiol Heart Circ Physiol. 2000;278:H321–H330. doi: 10.1152/ajpheart.2000.278.2.H321. [DOI] [PubMed] [Google Scholar]
- González-Alonso J, Teller C, Andersen SL, Jensen FB, Hyldeg T, Nielsen B. Influence of body temperature on the development of fatigue during prolonged exercise in the heat. J Appl Physiol. 1999a;86:1032–1039. doi: 10.1152/jappl.1999.86.3.1032. [DOI] [PubMed] [Google Scholar]
- Hargreaves M, Dillo P, Angus D, Howlett K, Conus NM, Febbraio M. Effect of fluid ingestion on muscle metabolism during prolonged exercise. J Appl Physiol. 1996;81:1594–1597. doi: 10.1152/jappl.1996.80.1.363. [DOI] [PubMed] [Google Scholar]
- Johnson JM. Exercise and the cutaneous circulation. Exerc Sports Sci Rev. 1992;20:59–97. [PubMed] [Google Scholar]
- Johnson JM, Proppe DW. Cardiovascular adjustments to heat stress. In: Fregly MJ, Blatteis CM, editors. Handbook of Physiology, section 4, Environmental Physiology. New York: Oxford University Press; 1996. pp. 215–243. chapt 11. [Google Scholar]
- Johnson JM, Rowell LB. Forearm skin and muscle vascular responses to prolonged leg exercise in man. J Appl Physiol. 1975;39:920–924. doi: 10.1152/jappl.1975.39.6.920. [DOI] [PubMed] [Google Scholar]
- Keller DM, Cui J, Davis SL, Low DA, Crandall CG. Heat stress enhances arterial baroreflex control of muscle sympathetic nerve activity via increased sensitivity of burst gating, not burst area, in humans. J Physiol. 2006;573:445–451. doi: 10.1113/jphysiol.2006.108662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kellogg DL, Jr, Johnson JM, Kenny WL, Pérgola PE, Kosiba WA. Mechanisms of control of skin blood flow during prolonged exercise in humans. Am J Physiol Heart Circ Physiol. 1993;265:H562–H568. doi: 10.1152/ajpheart.1993.265.2.H562. [DOI] [PubMed] [Google Scholar]
- Kellogg DL, Jr, Johnson JM, Kosiba WA. Elevation of the internal temperature threshold for active cutaneous vasodilation by dynamic exercise. J Appl Physiol. 1991;71:2476–2482. doi: 10.1152/jappl.1991.71.6.2476. [DOI] [PubMed] [Google Scholar]
- Kellogg DL, Jr, Pérgola PE, Kosiba WA, Grossmann M, Johnson JM. Cutaneous active vasodilation in humans is mediated by cholinergic nerve co-transmission. Circ Res. 1995;77:1222–1228. doi: 10.1161/01.res.77.6.1222. [DOI] [PubMed] [Google Scholar]
- Kenney WL, Johnson JM. Control of skin blood flow during exercise. Med Sci Sports Exerc. 1992;24:303–312. [PubMed] [Google Scholar]
- Krediet CTP, Wilde AAM, Wieling W, Halliwill JR. Exericse related syncope, when it's not the heart. Clin Auton Res. 2004;14(Suppl. 1):1/25-1/21/36. doi: 10.1007/s10286-004-1005-1. [DOI] [PubMed] [Google Scholar]
- Laughlin MH, Armstrong RB. Rat muscle blood flows as a function of time during prolonged slow treadmill exercise. Am J Physiol Heart Circ Physiol. 1983;244:H814–H824. doi: 10.1152/ajpheart.1983.244.6.H814. [DOI] [PubMed] [Google Scholar]
- Montain SJ, Coyle EF. Influences of graded dehydration on hyperthermia and cardiovascular drift during exercise. J Appl Physiol. 1992;73:1340–1350. doi: 10.1152/jappl.1992.73.4.1340. [DOI] [PubMed] [Google Scholar]
- Mortensen SP, Dawson EA, Yoshiga CC, Dalsgaard MK, Damsgaard R, Secher NH, González-Alonso J. Limitations to systemic and locomotor limb muscle oxygen delivery and uptake during maximal exercise in humans. J Physiol. 2005;566:273–285. doi: 10.1113/jphysiol.2005.086025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen B, Hales JRS, Strange S, Christiensen NJ, Warberg J, Saltin B. Human circulatory and thermoregulatory adaptations with heat acclimation and exercise in a hot, dry environment. J Physiol. 1993;460:467–485. doi: 10.1113/jphysiol.1993.sp019482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen B, Hyldig T, Bidstrup F, González-Alonso J, Christofferson GRJ. Brain activity and fatigue during prolonged exercise in the heat. Pflugers Arch. 2001;442:41–48. doi: 10.1007/s004240100515. [DOI] [PubMed] [Google Scholar]
- Nielsen B, Nybo L. Cerebral changes during exercise in the heat. Sports Med. 2003;33:1–11. doi: 10.2165/00007256-200333010-00001. [DOI] [PubMed] [Google Scholar]
- Nielsen B, Savard G, Richter EA, Hargreaves M, Saltin B. Muscle blood flow and metabolism during exercise and heat stress. J Appl Physiol. 1990;69:1040–1046. doi: 10.1152/jappl.1990.69.3.1040. [DOI] [PubMed] [Google Scholar]
- Niimi Y, Matsukawa T, Sugiyama Y, Shamsuzzaman AS, Ito H, Sobue G, Mano T. Effect of heat stress on muscle sympathetic nerve activity in humans. J Auton Nerv Sys. 1997;63:61–67. doi: 10.1016/s0165-1838(96)00134-8. [DOI] [PubMed] [Google Scholar]
- Nybo L, Jensen T, Nielsen B, González-Alonso J. Effects of marked hyperthermia with and without dehydration on VO2 kinetics during intense exercise. J Appl Physiol. 2001;90:1057–1064. doi: 10.1152/jappl.2001.90.3.1057. [DOI] [PubMed] [Google Scholar]
- Nybo L, Nielsen B. Middle cerebral artery blood velocity is reduced with hyperthermia during prolonged exercise in humans. J Physiol. 2001;534:279–286. doi: 10.1111/j.1469-7793.2001.t01-1-00279.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puvi-Rajasingham S, Smith GD, Akinola A, Mathias CJ. Abnormal regional blood flow responses during and after exercise in human sympathetic denervation. J Physiol. 1997;505:841–849. doi: 10.1111/j.1469-7793.1997.841ba.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ray CA, Gracey KH. Augmentation of exercise-induced muscle sympathetic nerve activity during muscle heating. J Appl Physiol. 1997;82:1719–1725. doi: 10.1152/jappl.1997.82.6.1719. [DOI] [PubMed] [Google Scholar]
- Rowell LB. Human cardiovascular adjustments to exercise and thermal stress. Physiol Rev. 1974;54:75–159. doi: 10.1152/physrev.1974.54.1.75. [DOI] [PubMed] [Google Scholar]
- Rowell LB, O'Leary DS, Kellogg DL., Jr . Integration of cardiovascular control systems in dynamic exercise. In: Rowell LB, Shepherd JT, editors. Handbook of Physiology, section 12, Exercise: Regulation and Integration of Multiple Systems. New York: Oxford University Press; 1996. pp. 770–838. chapt 17. [Google Scholar]
- Rowell LB, Saltin B, Kiens B, Christensen NJ. Is peak quadriceps blood flow even higher during exercise with hypoxemia? Am J Physiol Heart Circ Physiol. 1986;251:H1038–H1044. doi: 10.1152/ajpheart.1986.251.5.H1038. [DOI] [PubMed] [Google Scholar]
- Savard GK, Nielsen B, Laszcynska J, Larsen BE, Saltin B. Muscle blood flow is not reduced in humans during moderate exercise and heat stress. J Appl Physiol. 1988;64:649–657. doi: 10.1152/jappl.1988.64.2.649. [DOI] [PubMed] [Google Scholar]
- Sawka MN, Knowlton RG, Critz JB. Thermal and circulatory responses to repeated bouts of prolonged running. Med Sci Sports Exerc. 1979;11:177–180. [PubMed] [Google Scholar]
- Shibasaki M, Secher NH, Johnson JM, Crandall CG. Central command and the cutaneous vascular response to isometric exercise in heated human. J Physiol. 2005;565:667–673. doi: 10.1113/jphysiol.2005.083535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smolander J, Saalo J, Korhonen O. Effect of work load on cutaneous vascular response to exercise. J Appl Physiol. 1991;71:1614–1619. doi: 10.1152/jappl.1991.71.4.1614. [DOI] [PubMed] [Google Scholar]
- Thauer R. Circulatory adjustments to climatic requirements. In: Hamilton WF, editor. Handbook of Physiology, section 2, Circulation. III. Washington, DC: American Physiological Society; 1965. pp. 1921–1966. chapt 55. [Google Scholar]
- Weyand PG, Bundle MW. Energetics of high-speed running: integrating classical theory and contemporary observations. Am J Physiol Regul Integr Comp Physiol. 2005;288:R956–R965. doi: 10.1152/ajpregu.00628.2004. [DOI] [PubMed] [Google Scholar]