Abstract
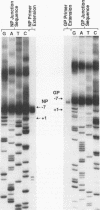
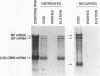
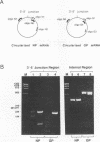
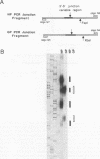
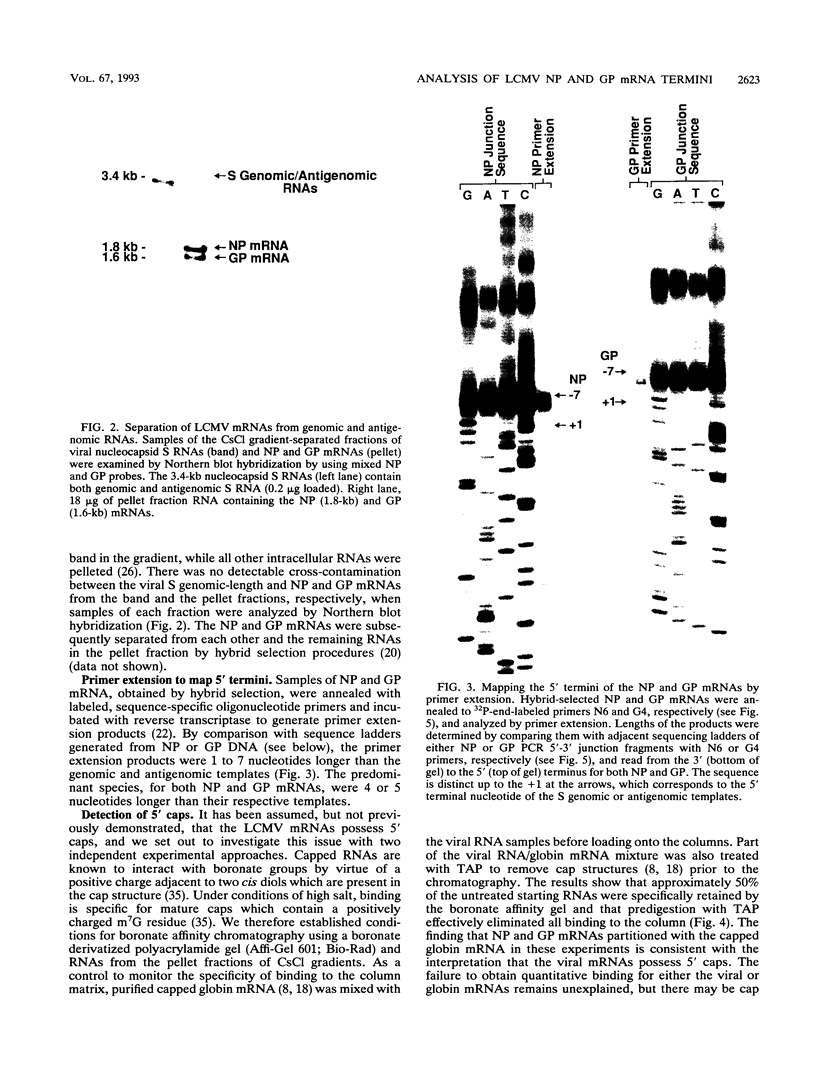
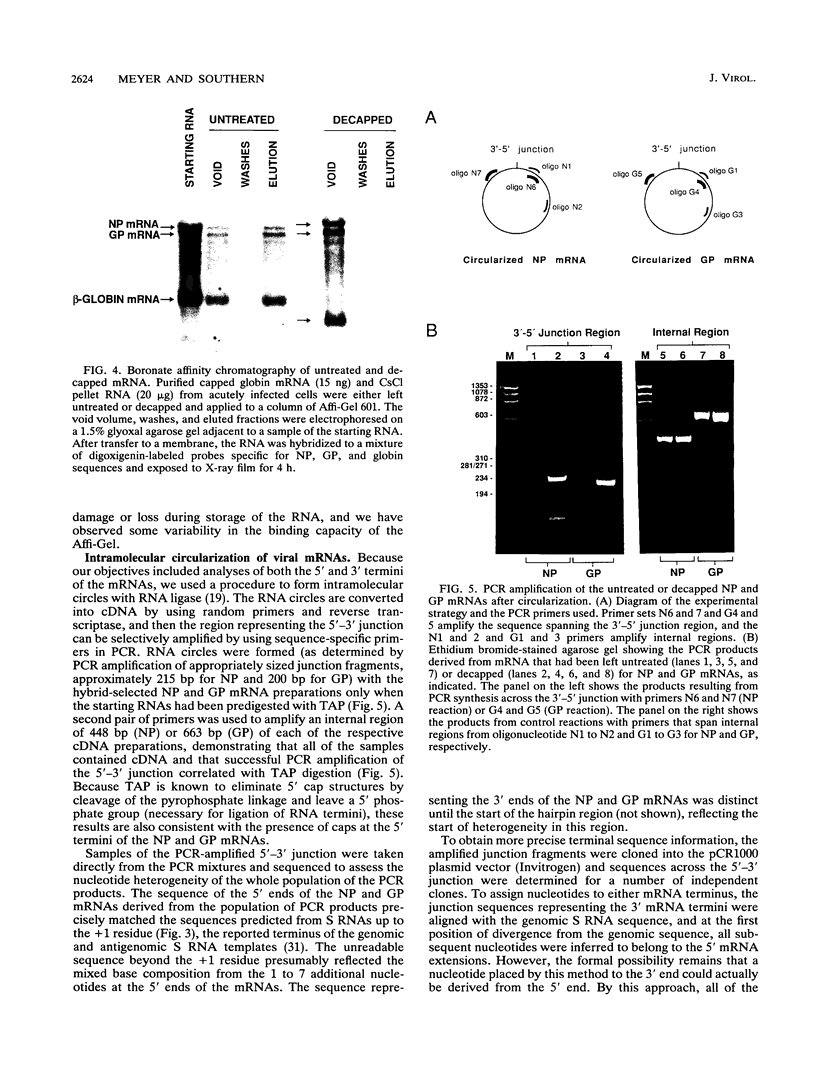
We have used a technique of RNA circularization coupled with polymerase chain reaction amplification for simultaneous analysis of the 5' and 3' termini of subgenomic mRNAs derived from the S RNA of lymphocytic choriomeningitis virus during an acute infection of BHK cells. These mRNAs possess 1 to 7 nontemplated nucleotides of apparently random sequence at their 5' ends. The predominant mRNA species have 4 or 5 nontemplated nucleotides. The 5' termini of the mRNAs also have properties consistent with the presence of a 5' cap structure. The 3' termini of the mRNAs lack poly(A) tails, and we have shown that transcription termination occurs at heterogeneous positions within the intergenic region of the S RNA. The identification of several distinct termini in the vicinity of a putative stem-loop structure in the RNA templates suggests that transcription termination may be mediated by a structural signal rather than a precise sequence signal.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Auperin D. D., Romanowski V., Galinski M., Bishop D. H. Sequencing studies of pichinde arenavirus S RNA indicate a novel coding strategy, an ambisense viral S RNA. J Virol. 1984 Dec;52(3):897–904. doi: 10.1128/jvi.52.3.897-904.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bishop D. H., Gay M. E., Matsuoko Y. Nonviral heterogeneous sequences are present at the 5' ends of one species of snowshoe hare bunyavirus S complementary RNA. Nucleic Acids Res. 1983 Sep 24;11(18):6409–6418. doi: 10.1093/nar/11.18.6409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouloy M., Plotch S. J., Krug R. M. Globin mRNAs are primers for the transcription of influenza viral RNA in vitro. Proc Natl Acad Sci U S A. 1978 Oct;75(10):4886–4890. doi: 10.1073/pnas.75.10.4886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchmeier M. J., Parekh B. S. Protein structure and expression among arenaviruses. Curr Top Microbiol Immunol. 1987;133:41–57. doi: 10.1007/978-3-642-71683-6_4. [DOI] [PubMed] [Google Scholar]
- Caton A. J., Robertson J. S. Structure of the host-derived sequences present at the 5' ends of influenza virus mRNA. Nucleic Acids Res. 1980 Jun 25;8(12):2591–2603. doi: 10.1093/nar/8.12.2591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collett M. S. Messenger RNA of the M segment RNA of Rift Valley fever virus. Virology. 1986 May;151(1):151–156. doi: 10.1016/0042-6822(86)90114-5. [DOI] [PubMed] [Google Scholar]
- Dutko F. J., Oldstone M. B. Genomic and biological variation among commonly used lymphocytic choriomeningitis virus strains. J Gen Virol. 1983 Aug;64(Pt 8):1689–1698. doi: 10.1099/0022-1317-64-8-1689. [DOI] [PubMed] [Google Scholar]
- Efstratiadis A., Vournakis J. N., Donis-Keller H., Chaconas G., Dougall D. K., Kafatos F. C. End labeling of enzymatically decapped mRNA. Nucleic Acids Res. 1977 Dec;4(12):4165–4174. doi: 10.1093/nar/4.12.4165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Francis S. J., Southern P. J. Deleted viral RNAs and lymphocytic choriomeningitis virus persistence in vitro. J Gen Virol. 1988 Aug;69(Pt 8):1893–1902. doi: 10.1099/0022-1317-69-8-1893. [DOI] [PubMed] [Google Scholar]
- Francis S. J., Southern P. J. Molecular analysis of viral RNAs in mice persistently infected with lymphocytic choriomeningitis virus. J Virol. 1988 Apr;62(4):1251–1257. doi: 10.1128/jvi.62.4.1251-1257.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franze-Fernández M. T., Zetina C., Iapalucci S., Lucero M. A., Bouissou C., López R., Rey O., Daheli M., Cohen G. N., Zakin M. M. Molecular structure and early events in the replication of Tacaribe arenavirus S RNA. Virus Res. 1987 Jun;7(4):309–324. doi: 10.1016/0168-1702(87)90045-1. [DOI] [PubMed] [Google Scholar]
- Fuller-Pace F. V., Southern P. J. Detection of virus-specific RNA-dependent RNA polymerase activity in extracts from cells infected with lymphocytic choriomeningitis virus: in vitro synthesis of full-length viral RNA species. J Virol. 1989 May;63(5):1938–1944. doi: 10.1128/jvi.63.5.1938-1944.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuller-Pace F. V., Southern P. J. Temporal analysis of transcription and replication during acute infection with lymphocytic choriomeningitis virus. Virology. 1988 Jan;162(1):260–263. doi: 10.1016/0042-6822(88)90419-9. [DOI] [PubMed] [Google Scholar]
- Garcin D., Kolakofsky D. A novel mechanism for the initiation of Tacaribe arenavirus genome replication. J Virol. 1990 Dec;64(12):6196–6203. doi: 10.1128/jvi.64.12.6196-6203.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcin D., Kolakofsky D. Tacaribe arenavirus RNA synthesis in vitro is primer dependent and suggests an unusual model for the initiation of genome replication. J Virol. 1992 Mar;66(3):1370–1376. doi: 10.1128/jvi.66.3.1370-1376.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iapalucci S., López N., Franze-Fernández M. T. The 3' end termini of the Tacaribe arenavirus subgenomic RNAs. Virology. 1991 May;182(1):269–278. doi: 10.1016/0042-6822(91)90670-7. [DOI] [PubMed] [Google Scholar]
- Lockard R. E., Rieser L., Vournakis J. N. Labeling of eukaryotic messenger RNA 5' terminus with phosphorus -32: use of tobacco acid pyrophosphatase for removal of cap structures. Gene Amplif Anal. 1981;2:229–251. [PubMed] [Google Scholar]
- Mandl C. W., Heinz F. X., Puchhammer-Stöckl E., Kunz C. Sequencing the termini of capped viral RNA by 5'-3' ligation and PCR. Biotechniques. 1991 Apr;10(4):484–486. [PubMed] [Google Scholar]
- McMaster G. K., Carmichael G. G. Analysis of single- and double-stranded nucleic acids on polyacrylamide and agarose gels by using glyoxal and acridine orange. Proc Natl Acad Sci U S A. 1977 Nov;74(11):4835–4838. doi: 10.1073/pnas.74.11.4835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer B. J., Schottel J. L. Characterization of cat messenger RNA decay suggests that turnover occurs by endonucleolytic cleavage in a 3' to 5' direction. Mol Microbiol. 1992 May;6(9):1095–1104. doi: 10.1111/j.1365-2958.1992.tb01547.x. [DOI] [PubMed] [Google Scholar]
- Patterson J. L., Holloway B., Kolakofsky D. La Crosse virions contain a primer-stimulated RNA polymerase and a methylated cap-dependent endonuclease. J Virol. 1984 Oct;52(1):215–222. doi: 10.1128/jvi.52.1.215-222.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Platt T. Transcription termination and the regulation of gene expression. Annu Rev Biochem. 1986;55:339–372. doi: 10.1146/annurev.bi.55.070186.002011. [DOI] [PubMed] [Google Scholar]
- Plotch S. J., Bouloy M., Ulmanen I., Krug R. M. A unique cap(m7GpppXm)-dependent influenza virion endonuclease cleaves capped RNAs to generate the primers that initiate viral RNA transcription. Cell. 1981 Mar;23(3):847–858. doi: 10.1016/0092-8674(81)90449-9. [DOI] [PubMed] [Google Scholar]
- Raju R., Raju L., Hacker D., Garcin D., Compans R., Kolakofsky D. Nontemplated bases at the 5' ends of Tacaribe virus mRNAs. Virology. 1990 Jan;174(1):53–59. doi: 10.1016/0042-6822(90)90053-t. [DOI] [PubMed] [Google Scholar]
- Reynolds R., Bermúdez-Cruz R. M., Chamberlin M. J. Parameters affecting transcription termination by Escherichia coli RNA polymerase. I. Analysis of 13 rho-independent terminators. J Mol Biol. 1992 Mar 5;224(1):31–51. doi: 10.1016/0022-2836(92)90574-4. [DOI] [PubMed] [Google Scholar]
- Reynolds R., Chamberlin M. J. Parameters affecting transcription termination by Escherichia coli RNA. II. Construction and analysis of hybrid terminators. J Mol Biol. 1992 Mar 5;224(1):53–63. doi: 10.1016/0022-2836(92)90575-5. [DOI] [PubMed] [Google Scholar]
- Romanowski V., Matsuura Y., Bishop D. H. Complete sequence of the S RNA of lymphocytic choriomeningitis virus (WE strain) compared to that of Pichinde arenavirus. Virus Res. 1985 Sep;3(2):101–114. doi: 10.1016/0168-1702(85)90001-2. [DOI] [PubMed] [Google Scholar]
- Salvato M. S., Shimomaye E. M. The completed sequence of lymphocytic choriomeningitis virus reveals a unique RNA structure and a gene for a zinc finger protein. Virology. 1989 Nov;173(1):1–10. doi: 10.1016/0042-6822(89)90216-x. [DOI] [PubMed] [Google Scholar]
- Salvato M., Shimomaye E., Southern P., Oldstone M. B. Virus-lymphocyte interactions. IV. Molecular characterization of LCMV Armstrong (CTL+) small genomic segment and that of its variant, Clone 13 (CTL-). Virology. 1988 Jun;164(2):517–522. doi: 10.1016/0042-6822(88)90566-1. [DOI] [PubMed] [Google Scholar]
- Singh M. K., Fuller-Pace F. V., Buchmeier M. J., Southern P. J. Analysis of the genomic L RNA segment from lymphocytic choriomeningitis virus. Virology. 1987 Dec;161(2):448–456. doi: 10.1016/0042-6822(87)90138-3. [DOI] [PubMed] [Google Scholar]
- Southern P. J., Singh M. K., Riviere Y., Jacoby D. R., Buchmeier M. J., Oldstone M. B. Molecular characterization of the genomic S RNA segment from lymphocytic choriomeningitis virus. Virology. 1987 Mar;157(1):145–155. doi: 10.1016/0042-6822(87)90323-0. [DOI] [PubMed] [Google Scholar]
- Wilk H. E., Kecskemethy N., Schäfer K. P. m-Aminophenylboronate agarose specifically binds capped snRNA and mRNA. Nucleic Acids Res. 1982 Dec 11;10(23):7621–7633. doi: 10.1093/nar/10.23.7621. [DOI] [PMC free article] [PubMed] [Google Scholar]