Abstract
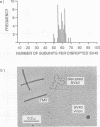
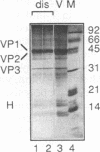
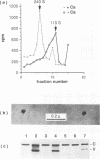
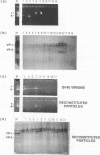
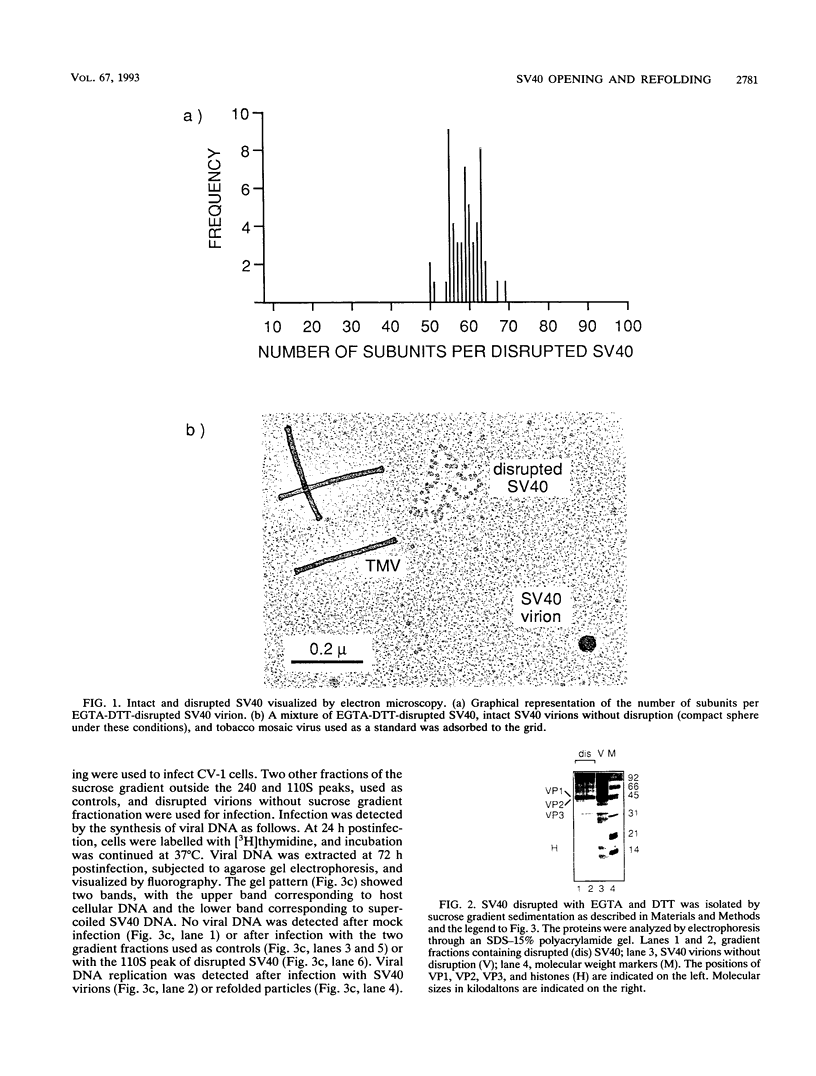
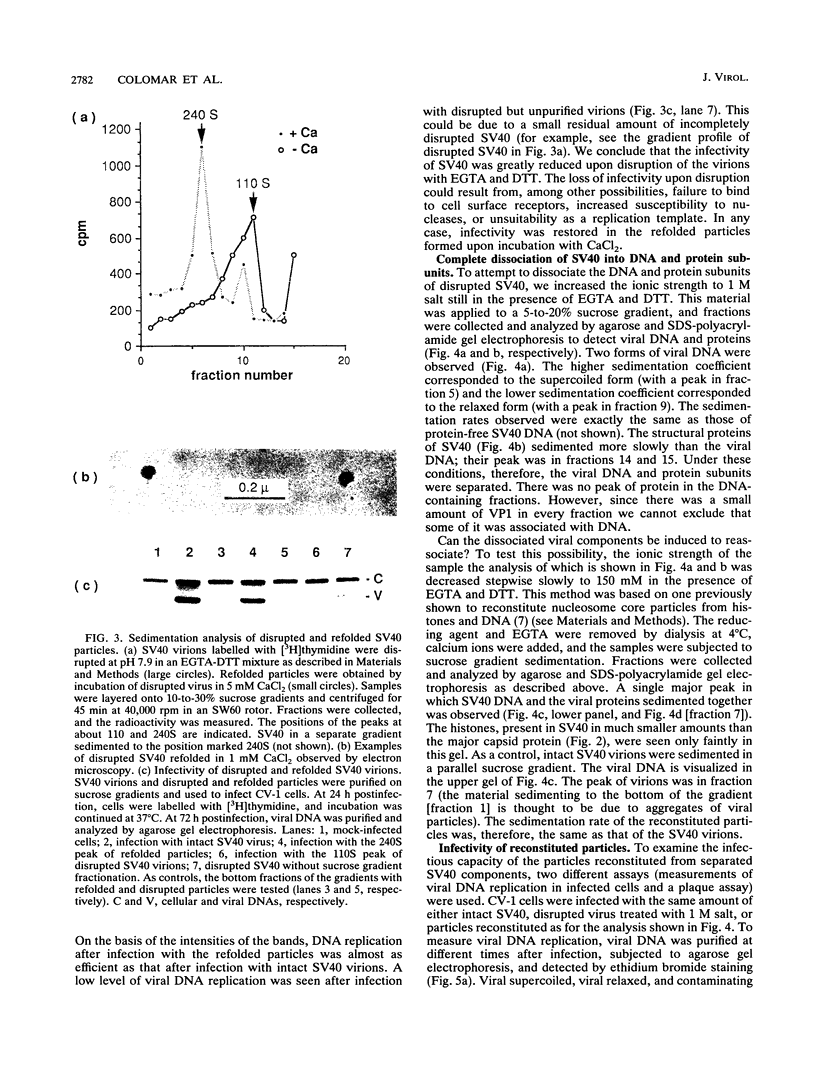
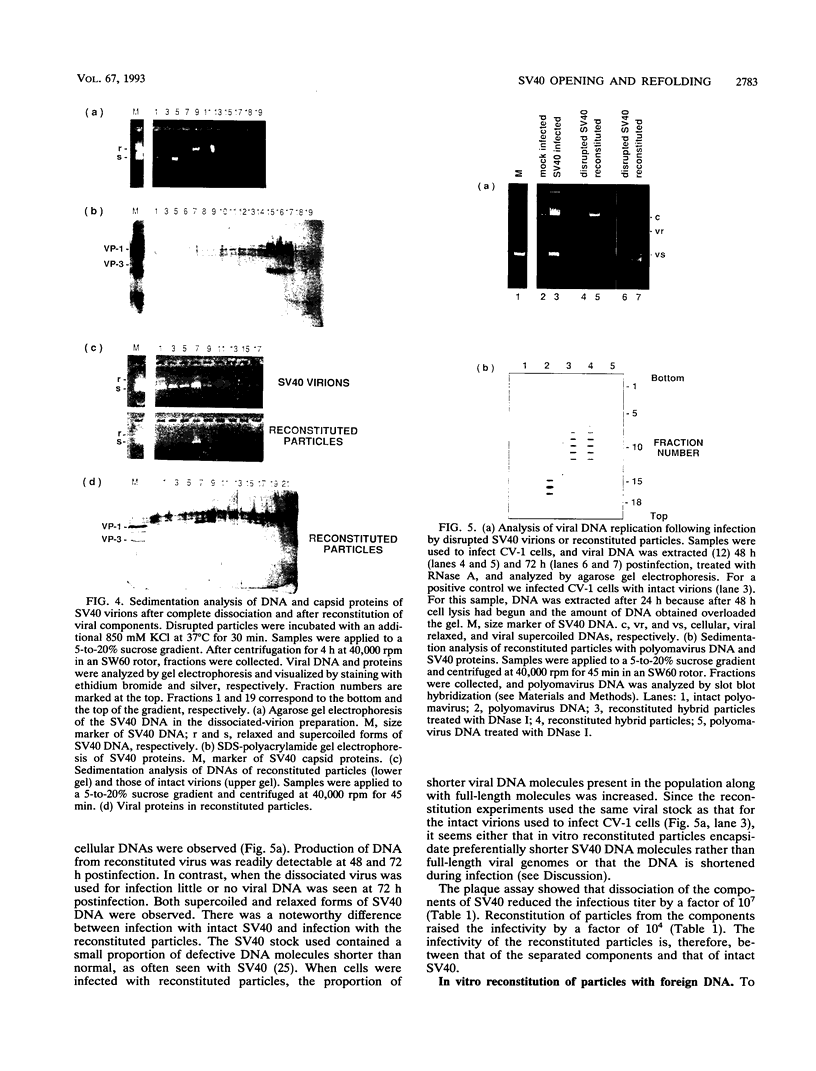
Simian virus 40 (SV40) can be disassembled under mild conditions by reducing disulfide bonds in the capsid and removing calcium ions. The nucleoprotein complexes formed, analyzed by electron microscopy, were circular and made up of 59 +/- 4 subunits, each with a diameter of about 10 nm. The complexes contained the viral DNA, histones, and the viral capsid proteins. The complexes had much-reduced infectivities compared with intact SV40. Addition of calcium ions to the disrupted virus caused the nucleoprotein complexes to refold into virus-like structures which sedimented at the same rate as intact SV40 and regained infectivity. Treatment of the disrupted SV40 with a high concentration of salt dissociated the viral proteins from the DNA. Lowering stepwise the salt concentration, removing the reducing agent, and adding calcium ions allowed structures to be reformed, and these structures sedimented, like SV40, at 240S and were infectious. The plaque-forming ability of the reconstituted particles was between that of the dissociated components and that of intact SV40. The addition of purified DNA of polyomavirus to the dissociated SV40 before the lowering of the salt concentration showed that virus-like structures could be formed from SV40 proteins and a foreign DNA.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brady J. N., Lavialle C., Salzman N. P. Efficient transcription of a compact nucleoprotein complex isolated from purified simian virus 40 virions. J Virol. 1980 Aug;35(2):371–381. doi: 10.1128/jvi.35.2.371-381.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brady J. N., Winston V. D., Consigli R. A. Dissociation of polyoma virus by the chelation of calcium ions found associated with purified virions. J Virol. 1977 Sep;23(3):717–724. doi: 10.1128/jvi.23.3.717-724.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christensen M., Rachmeler M. Studies on the in vitro formation of infectious DNA-proteins aggregates from SV40 components. Virology. 1976 Dec;75(2):433–441. doi: 10.1016/0042-6822(76)90041-6. [DOI] [PubMed] [Google Scholar]
- Christiansen G., Landers T., Griffith J., Berg P. Characterization of components released by alkali disruption of simian virus 40. J Virol. 1977 Mar;21(3):1079–1084. doi: 10.1128/jvi.21.3.1079-1084.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cremisi C., Pignatti P. F., Croissant O., Yaniv M. Chromatin-like structures in polyoma virus and simian virus 10 lytic cycle. J Virol. 1975 Jan;17(1):204–211. doi: 10.1128/jvi.17.1.204-211.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drew H. R., Travers A. A. DNA bending and its relation to nucleosome positioning. J Mol Biol. 1985 Dec 20;186(4):773–790. doi: 10.1016/0022-2836(85)90396-1. [DOI] [PubMed] [Google Scholar]
- Dubochet J., Ducommun M., Zollinger M., Kellenberger E. A new preparation method for dark-field electron microscopy of biomacromolecules. J Ultrastruct Res. 1971 Apr;35(1):147–167. doi: 10.1016/s0022-5320(71)80148-x. [DOI] [PubMed] [Google Scholar]
- Engel A., Baumeister W., Saxton W. O. Mass mapping of a protein complex with the scanning transmission electron microscope. Proc Natl Acad Sci U S A. 1982 Jul;79(13):4050–4054. doi: 10.1073/pnas.79.13.4050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiers W., Contreras R., Haegemann G., Rogiers R., Van de Voorde A., Van Heuverswyn H., Van Herreweghe J., Volckaert G., Ysebaert M. Complete nucleotide sequence of SV40 DNA. Nature. 1978 May 11;273(5658):113–120. doi: 10.1038/273113a0. [DOI] [PubMed] [Google Scholar]
- Hirt B. Selective extraction of polyoma DNA from infected mouse cell cultures. J Mol Biol. 1967 Jun 14;26(2):365–369. doi: 10.1016/0022-2836(67)90307-5. [DOI] [PubMed] [Google Scholar]
- Liddington R. C., Yan Y., Moulai J., Sahli R., Benjamin T. L., Harrison S. C. Structure of simian virus 40 at 3.8-A resolution. Nature. 1991 Nov 28;354(6351):278–284. doi: 10.1038/354278a0. [DOI] [PubMed] [Google Scholar]
- Menck C. F., James M., Benoit A., Sarasin A. Constraints in simian virus 40 (SV40) encapsidation, as determined by SV40-based shuttle viruses. J Gen Virol. 1990 Jan;71(Pt 1):143–150. doi: 10.1099/0022-1317-71-1-143. [DOI] [PubMed] [Google Scholar]
- Michel M. R., Hirt B., Weil R. Mouse cellular DNA enclosed in polyoma viral capsids (pseudovirions). Proc Natl Acad Sci U S A. 1967 Oct;58(4):1381–1388. doi: 10.1073/pnas.58.4.1381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moyne G., Harper F., Saragosti S., Yaniv M. Absence of nucleosomes in a histone-containing nucleoprotein complex obtained by dissociation of purified SV40 virions. Cell. 1982 Aug;30(1):123–130. doi: 10.1016/0092-8674(82)90018-6. [DOI] [PubMed] [Google Scholar]
- Oppenheim A., Sandalon Z., Peleg A., Shaul O., Nicolis S., Ottolenghi S. A cis-acting DNA signal for encapsidation of simian virus 40. J Virol. 1992 Sep;66(9):5320–5328. doi: 10.1128/jvi.66.9.5320-5328.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rayment I., Baker T. S., Caspar D. L., Murakami W. T. Polyoma virus capsid structure at 22.5 A resolution. Nature. 1982 Jan 14;295(5845):110–115. doi: 10.1038/295110a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reddy V. B., Thimmappaya B., Dhar R., Subramanian K. N., Zain B. S., Pan J., Ghosh P. K., Celma M. L., Weissman S. M. The genome of simian virus 40. Science. 1978 May 5;200(4341):494–502. doi: 10.1126/science.205947. [DOI] [PubMed] [Google Scholar]
- Subramani S., Southern P. J. Analysis of gene expression using simian virus 40 vectors. Anal Biochem. 1983 Nov;135(1):1–15. doi: 10.1016/0003-2697(83)90723-6. [DOI] [PubMed] [Google Scholar]
- Switzer R. C., 3rd, Merril C. R., Shifrin S. A highly sensitive silver stain for detecting proteins and peptides in polyacrylamide gels. Anal Biochem. 1979 Sep 15;98(1):231–237. doi: 10.1016/0003-2697(79)90732-2. [DOI] [PubMed] [Google Scholar]
- Tack L. C., Beard P. Both trans-acting factors and chromatin structure are involved in the regulation of transcription from the early and late promoters in simian virus 40 chromosomes. J Virol. 1985 Apr;54(1):207–218. doi: 10.1128/jvi.54.1.207-218.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tai H. T., Smith C. A., Sharp P. A., Vinograd J. Sequence heterogeneity in closed simian virus 40 deoxyribonucleic acid. J Virol. 1972 Feb;9(2):317–325. doi: 10.1128/jvi.9.2.317-325.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trilling D. M., Axelrod D. Encapsidation of free host DNA by simian virus 40: a simian virus 40 pseudovirus. Science. 1970 Apr 10;168(3928):268–271. doi: 10.1126/science.168.3928.268. [DOI] [PubMed] [Google Scholar]
- Varshavsky A. J., Nedospasov S. A., Schmatchenko V. V., Bakayev V. V., Chumackov P. M., Georgiev G. P. Compact form of SV40 viral minichromosome is resistant to nuclease: possible implications for chromatin structure. Nucleic Acids Res. 1977 Oct;4(10):3303–3325. doi: 10.1093/nar/4.10.3303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walter G., Deppert W. Intermolecular disulfide bonds: an important structural feature of the polyoma virus capsid. Cold Spring Harb Symp Quant Biol. 1975;39(Pt 1):255–257. doi: 10.1101/sqb.1974.039.01.033. [DOI] [PubMed] [Google Scholar]