Abstract
Using a polymerase chain reaction strategy aimed at detecting recombinant feline leukemia virus (FeLV) genomes with 5' env sequences originating from an endogenous source and 3' env sequences resulting from FeLV subgroup A (FeLV-A), we detected recombinant proviruses in approximately three-fourths of naturally occurring thymic and alimentary feline lymphosarcomas (LSAs) and one-third of the multicentric LSAs from cats determined to be FeLV capsid antigen positive by immunofluorescence assay. In contrast, only 1 of 22 naturally arising FeLV-negative feline LSAs contained recombinant proviruses, and no recombinant env gene was detected in seven samples from normal tissues or tissues from FeLV-positive animals that died from other diseases. Four preferred structural motifs were identified in the recombinants; one is FeLV-B like (recognizing that FeLV-B itself is a product of recombination between FeLV-A and endogenous env genes), and three contain variable amounts of endogenous-like env gene before crossing over to FeLV-A-related sequences: (i) a combination of full-length and deleted env genes with recombination at sites in the middle of the surface glycoprotein (SU), (ii) the entire SU encoded by endogenous-like sequences, and (iii) the entire SU and approximately half of the transmembrane protein encoded by endogenous-like sequences. Additionally, three of the thymic tumors contained recombinant proviruses with mutations in the vicinity of the major neutralizing determinant for the SU protein. These molecular genetic analyses of the LSA DNAs correspond to our previous results in vitro and support the occurrence and association of viral recombinants and mutants in vivo in FeLV-induced leukemogenesis.
Full text
PDF

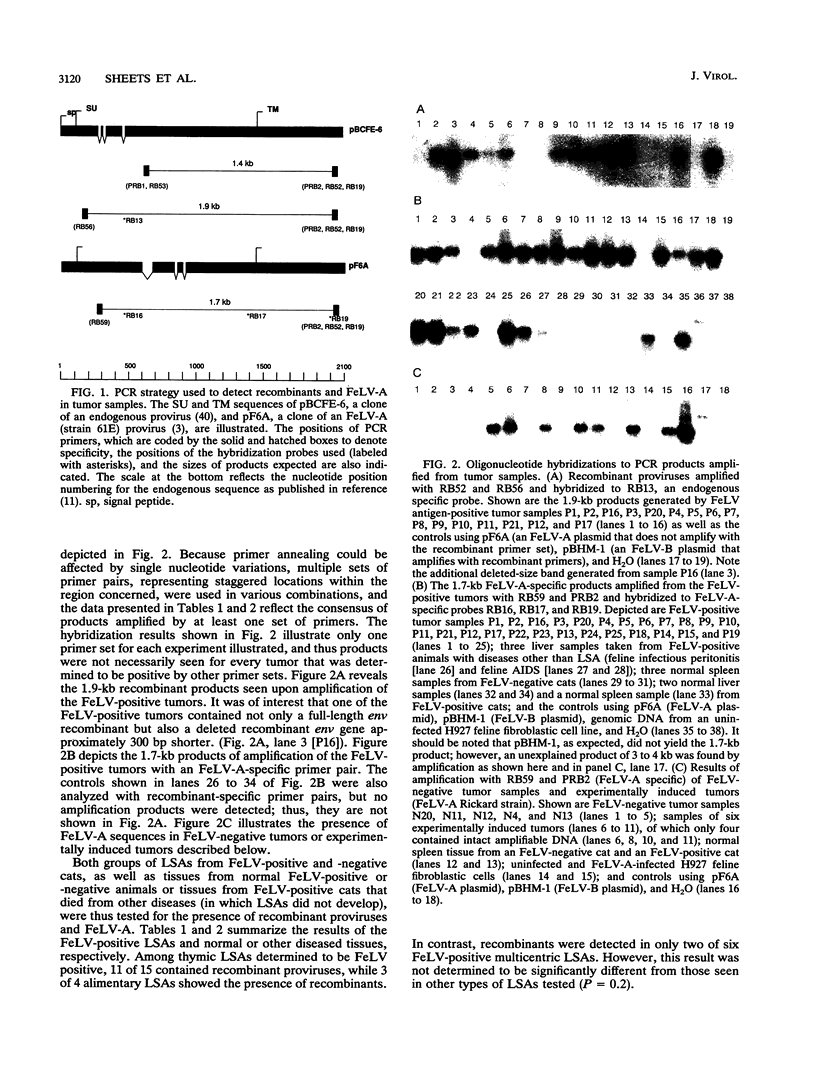
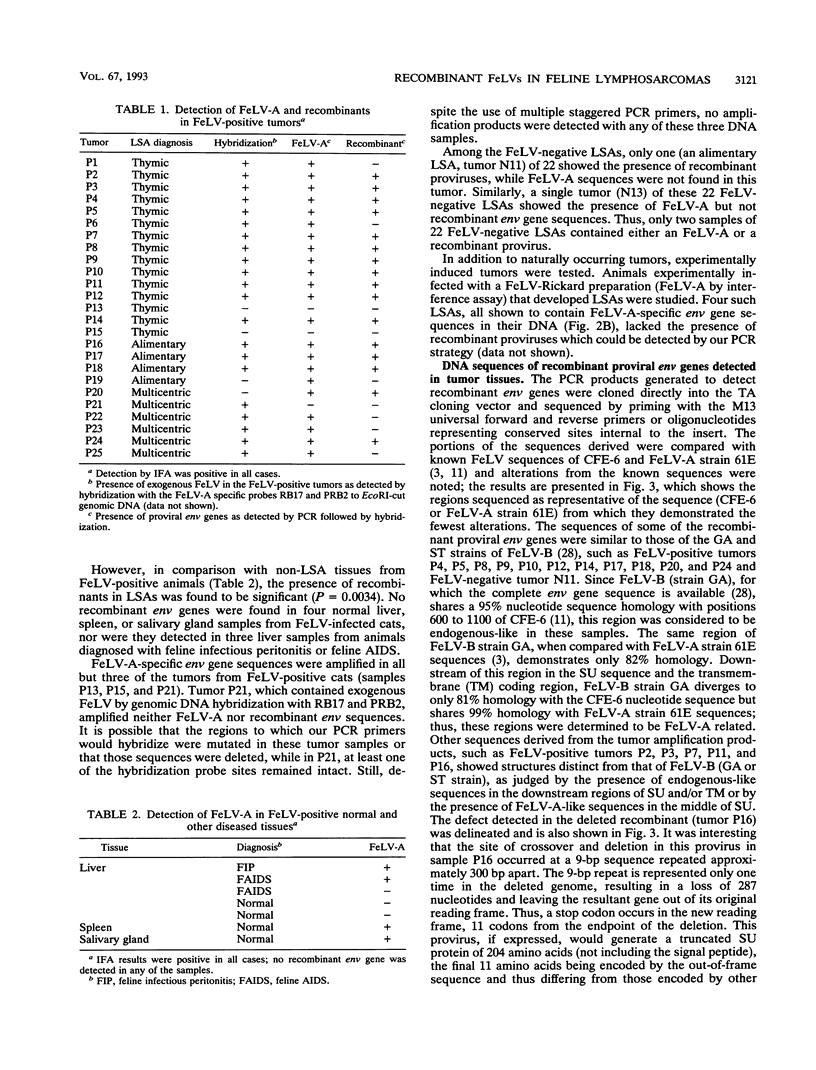




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berry B. T., Ghosh A. K., Kumar D. V., Spodick D. A., Roy-Burman P. Structure and function of endogenous feline leukemia virus long terminal repeats and adjoining regions. J Virol. 1988 Oct;62(10):3631–3641. doi: 10.1128/jvi.62.10.3631-3641.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen E. Y., Seeburg P. H. Supercoil sequencing: a fast and simple method for sequencing plasmid DNA. DNA. 1985 Apr;4(2):165–170. doi: 10.1089/dna.1985.4.165. [DOI] [PubMed] [Google Scholar]
- Donahue P. R., Hoover E. A., Beltz G. A., Riedel N., Hirsch V. M., Overbaugh J., Mullins J. I. Strong sequence conservation among horizontally transmissible, minimally pathogenic feline leukemia viruses. J Virol. 1988 Mar;62(3):722–731. doi: 10.1128/jvi.62.3.722-731.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elder J. H., Gautsch J. W., Jensen F. C., Lerner R. A., Hartley J. W., Rowe W. P. Biochemical evidence that MCF murine leukemia viruses are envelope (env) gene recombinants. Proc Natl Acad Sci U S A. 1977 Oct;74(10):4676–4680. doi: 10.1073/pnas.74.10.4676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elder J. H., McGee J. S., Munson M., Houghten R. A., Kloetzer W., Bittle J. L., Grant C. K. Localization of neutralizing regions of the envelope gene of feline leukemia virus by using anti-synthetic peptide antibodies. J Virol. 1987 Jan;61(1):8–15. doi: 10.1128/jvi.61.1.8-15.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forrest D., Onions D., Lees G., Neil J. C. Altered structure and expression of c-myc in feline T-cell tumours. Virology. 1987 May;158(1):194–205. doi: 10.1016/0042-6822(87)90253-4. [DOI] [PubMed] [Google Scholar]
- Hartley J. W., Wolford N. K., Old L. J., Rowe W. P. A new class of murine leukemia virus associated with development of spontaneous lymphomas. Proc Natl Acad Sci U S A. 1977 Feb;74(2):789–792. doi: 10.1073/pnas.74.2.789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jarrett O., Hardy W. D., Jr, Golder M. C., Hay D. The frequency of occurrence of feline leukaemia virus subgroups in cats. Int J Cancer. 1978 Mar 15;21(3):334–337. doi: 10.1002/ijc.2910210314. [DOI] [PubMed] [Google Scholar]
- Jarrett O., Laird H. M., Hay D. Determinants of the host range of feline leukaemia viruses. J Gen Virol. 1973 Aug;20(2):169–175. doi: 10.1099/0022-1317-20-2-169. [DOI] [PubMed] [Google Scholar]
- Kumar D. V., Berry B. T., Roy-Burman P. Nucleotide sequence and distinctive characteristics of the env gene of endogenous feline leukemia provirus. J Virol. 1989 May;63(5):2379–2384. doi: 10.1128/jvi.63.5.2379-2384.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levesque K. S., Bonham L., Levy L. S. flvi-1, a common integration domain of feline leukemia virus in naturally occurring lymphomas of a particular type. J Virol. 1990 Jul;64(7):3455–3462. doi: 10.1128/jvi.64.7.3455-3462.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levy L. S., Gardner M. B., Casey J. W. Isolation of a feline leukaemia provirus containing the oncogene myc from a feline lymphosarcoma. 1984 Apr 26-May 2Nature. 308(5962):853–856. doi: 10.1038/308853a0. [DOI] [PubMed] [Google Scholar]
- Levy L. S., Lobelle-Rich P. A. Insertional mutagenesis of flvi-2 in tumors induced by infection with LC-FeLV, a myc-containing strain of feline leukemia virus. J Virol. 1992 May;66(5):2885–2892. doi: 10.1128/jvi.66.5.2885-2892.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mead D. A., Pey N. K., Herrnstadt C., Marcil R. A., Smith L. M. A universal method for the direct cloning of PCR amplified nucleic acid. Biotechnology (N Y) 1991 Jul;9(7):657–663. doi: 10.1038/nbt0791-657. [DOI] [PubMed] [Google Scholar]
- Miura T., Tsujimoto H., Fukasawa M., Kodama T., Shibuya M., Hasegawa A., Hayami M. Structural abnormality and over-expression of the myc gene in feline leukemias. Int J Cancer. 1987 Oct 15;40(4):564–569. doi: 10.1002/ijc.2910400422. [DOI] [PubMed] [Google Scholar]
- Miyada C. G., Wallace R. B. Oligonucleotide hybridization techniques. Methods Enzymol. 1987;154:94–107. doi: 10.1016/0076-6879(87)54072-1. [DOI] [PubMed] [Google Scholar]
- Mullins J. I., Brody D. S., Binari R. C., Jr, Cotter S. M. Viral transduction of c-myc gene in naturally occurring feline leukaemias. 1984 Apr 26-May 2Nature. 308(5962):856–858. doi: 10.1038/308856a0. [DOI] [PubMed] [Google Scholar]
- Neil J. C., Forrest D., Doggett D. L., Mullins J. I. The role of feline leukaemia virus in naturally occurring leukaemias. Cancer Surv. 1987;6(1):117–137. [PubMed] [Google Scholar]
- Neil J. C., Fulton R., Rigby M., Stewart M. Feline leukaemia virus: generation of pathogenic and oncogenic variants. Curr Top Microbiol Immunol. 1991;171:67–93. doi: 10.1007/978-3-642-76524-7_4. [DOI] [PubMed] [Google Scholar]
- Neil J. C., Hughes D., McFarlane R., Wilkie N. M., Onions D. E., Lees G., Jarrett O. Transduction and rearrangement of the myc gene by feline leukaemia virus in naturally occurring T-cell leukaemias. 1984 Apr 26-May 2Nature. 308(5962):814–820. doi: 10.1038/308814a0. [DOI] [PubMed] [Google Scholar]
- Nicolaisen-Strouss K., Kumar H. P., Fitting T., Grant C. K., Elder J. H. Natural feline leukemia virus variant escapes neutralization by a monoclonal antibody via an amino acid change outside the antibody-binding epitope. J Virol. 1987 Nov;61(11):3410–3415. doi: 10.1128/jvi.61.11.3410-3415.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niman H. L., Akhavi M., Gardner M. B., Stephenson J. R., Roy-Burman P. Differential expression of two distinct endogenous retrovisus genomes in developing tissues of the domestic cat. J Natl Cancer Inst. 1980 Mar;64(3):587–594. [PubMed] [Google Scholar]
- Niman H. L., Gardner M. B., Stephenson J. R., Roy-Burman P. Endogenous RD-114 virus genome expression in malignant tissues of domestic cats. J Virol. 1977 Sep;23(3):578–586. doi: 10.1128/jvi.23.3.578-586.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niman H. L., Stephenson J. R., Gardner M. B., Roy-Burman P. RD-114 and feline leukaemia virus genome expression in natural lymphomas of domestic cats. Nature. 1977 Mar 24;266(5600):357–360. doi: 10.1038/266357a0. [DOI] [PubMed] [Google Scholar]
- Nunberg J. H., Rodgers G., Gilbert J. H., Snead R. M. Method to map antigenic determinants recognized by monoclonal antibodies: localization of a determinant of virus neutralization on the feline leukemia virus envelope protein gp70. Proc Natl Acad Sci U S A. 1984 Jun;81(12):3675–3679. doi: 10.1073/pnas.81.12.3675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nunberg J. H., Williams M. E., Innis M. A. Nucleotide sequences of the envelope genes of two isolates of feline leukemia virus subgroup B. J Virol. 1984 Feb;49(2):629–632. doi: 10.1128/jvi.49.2.629-632.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Overbaugh J., Riedel N., Hoover E. A., Mullins J. I. Transduction of endogenous envelope genes by feline leukaemia virus in vitro. Nature. 1988 Apr 21;332(6166):731–734. doi: 10.1038/332731a0. [DOI] [PubMed] [Google Scholar]
- Pandey R., Ghosh A. K., Kumar D. V., Bachman B. A., Shibata D., Roy-Burman P. Recombination between feline leukemia virus subgroup B or C and endogenous env elements alters the in vitro biological activities of the viruses. J Virol. 1991 Dec;65(12):6495–6508. doi: 10.1128/jvi.65.12.6495-6508.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rein A., Schultz A. Different recombinant murine leukemia viruses use different cell surface receptors. Virology. 1984 Jul 15;136(1):144–152. doi: 10.1016/0042-6822(84)90255-1. [DOI] [PubMed] [Google Scholar]
- Rickard C. G., Post J. E., Noronha F., Barr L. M. A transmissible virus-induced lymphocytic leukemia of the cat. J Natl Cancer Inst. 1969 Jun;42(6):987–1014. [PubMed] [Google Scholar]
- Sanger F., Coulson A. R., Barrell B. G., Smith A. J., Roe B. A. Cloning in single-stranded bacteriophage as an aid to rapid DNA sequencing. J Mol Biol. 1980 Oct 25;143(2):161–178. doi: 10.1016/0022-2836(80)90196-5. [DOI] [PubMed] [Google Scholar]
- Sarma P. S., Log T., Jain D., Hill P. R., Huebner R. J. Differential host range of viruses of feline leukemia-sarcoma complex. Virology. 1975 Apr;64(2):438–446. doi: 10.1016/0042-6822(75)90121-x. [DOI] [PubMed] [Google Scholar]
- Sarma P. S., Log T. Subgroup classification of feline leukemia and sarcoma viruses by viral interference and neutralization tests. Virology. 1973 Jul;54(1):160–169. doi: 10.1016/0042-6822(73)90125-6. [DOI] [PubMed] [Google Scholar]
- Sheets R. L., Pandey R., Klement V., Grant C. K., Roy-Burman P. Biologically selected recombinants between feline leukemia virus (FeLV) subgroup A and an endogenous FeLV element. Virology. 1992 Oct;190(2):849–855. doi: 10.1016/0042-6822(92)90924-e. [DOI] [PubMed] [Google Scholar]
- Soe L. H., Devi B. G., Mullins J. I., Roy-Burman P. Molecular cloning and characterization of endogenous feline leukemia virus sequences from a cat genomic library. J Virol. 1983 Jun;46(3):829–840. doi: 10.1128/jvi.46.3.829-840.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soe L. H., Ghosh A. K., Maxson R. E., Hoover E. A., Hardy W. D., Jr, Roy-Burman P. Nucleotide sequence of the 1.2-kb 3'-region and genotype distribution of two common c-myc alleles of the domestic cat. Gene. 1986;47(2-3):185–192. doi: 10.1016/0378-1119(86)90062-4. [DOI] [PubMed] [Google Scholar]
- Soe L. H., Shimizu R. W., Landolph J. R., Roy-Burman P. Molecular analysis of several classes of endogenous feline leukemia virus elements. J Virol. 1985 Dec;56(3):701–710. doi: 10.1128/jvi.56.3.701-710.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stoye J. P., Moroni C., Coffin J. M. Virological events leading to spontaneous AKR thymomas. J Virol. 1991 Mar;65(3):1273–1285. doi: 10.1128/jvi.65.3.1273-1285.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]