Abstract
Objectives
Posttraumatic hypotension is believed to increase morbidity and mortality in traumatically brain-injured patients. Using a clinically relevant model of combined traumatic brain injury with superimposed hemorrhagic hypotension in rats, the present study evaluated whether a reduction in mean arterial blood pressure aggravates regional brain edema formation, regional cell death, and neurologic motor/cognitive deficits associated with traumatic brain injury.
Design
Experimental prospective, randomized study in rodents.
Setting
Experimental laboratory at a university hospital.
Subjects
One hundred nineteen male Sprague-Dawley rats weighing 350-385 g.
Interventions
Experimental traumatic brain injury of mild to moderate severity was induced using the lateral fluid percussion brain injury model in anesthetized rats (n = 89). Following traumatic brain injury, in surviving animals one group of animals was subjected to pressure-controlled hemorrhagic hypotension, maintaining the mean arterial blood pressure at 50-60 mm Hg for 30 mins (n = 47). The animals were subsequently either resuscitated with lactated Ringer’s solution (three times shed blood volume, n = 18) or left uncompensated (n = 29). Other groups of animals included those with isolated traumatic brain injury (n = 34), those with isolated hemorrhagic hypotension (n = 8), and sham-injured control animals receiving anesthesia and surgery alone (n = 22).
Measurements and Main Results
The withdrawal of 6-7 mL of arterial blood significantly reduced mean arterial blood pressure by 50% without decreasing arterial oxygen saturation or Pao2. Brain injury induced significant cerebral edema (p < .001) in vulnerable brain regions and cortical tissue loss (p < .01) compared with sham-injured animals. Neither regional brain edema formation at 24 hrs postinjury nor the extent of cortical tissue loss assessed at 7 days postinjury was significantly aggravated by superimposed hemorrhagic hypotension. Brain injury-induced neurologic deficits persisted up to 20 wks after injury and were also not aggravated by the hemorrhagic hypotension. Cognitive dysfunction persisted for up to 16 wks postinjury. The superimposition of hemorrhagic hypotension significantly delayed the time course of cognitive recovery.
Conclusions
A single, acute hypotensive event lasting 30 mins did not aggravate the short- and long-term structural and motor deficits but delayed the speed of recovery of cognitive function associated with experimental traumatic brain injury.
Keywords: head injury, secondary brain damage, Morris water maze, hemorrhagic shock, fluid resuscitation
Approximately 50% of severely head-injured patients suffer additional potentially life-threatening extracranial injuries such as multiple fractures, lung contusion, traumatic pneumothorax, and injury to intra-abdominal organs. Despite the high prevalence of multiple trauma in brain-injured patients, only a few studies have investigated the effect of extracranial injuries on the clinical course and outcome of patients with traumatic brain injury (TBI) (1, 2). Nevertheless, the prognosis and outcome have been shown to be worsened when multiple extracranial injuries are involved (1, 3). This exacerbation of injury is believed to be a consequence of secondary insults to the brain resulting from hypoxemia, hypotension, elevated intracranial pressure, insufficient cerebral perfusion pressure, hypercapnia, hyperthermia, hypo/hyperglycemia, hyponatremia, and sepsis (4-6) that can be caused by extracranial traumatic injuries. Although the uninjured brain can normally withstand these secondary insults by activating compensatory mechanisms, the brain is rendered vulnerable to secondary insults due to TBI-induced structural and metabolic deficits. Blood loss and subsequent systemic hypotension are major causes of secondary insult to the brain following TBI and occur more frequently in multiply injured patients (4, 7). Moreover, hypotension in the traumatically injured patient has been shown to be predictive for higher mortality and morbidity rates (8).
Under laboratory conditions, experimentally induced hemorrhagic hypotension (HH) when superimposed on a traumatically injured brain has been shown to impair cerebral perfusion (9, 10) and reduce the amount of oxygen carriers contributing to the already reduced brain tissue oxygenation (11, 12). Additionally, secondary hypotension, with and without concomitant hypoxia, following TBI has been reported to impair systemic pressure autoregulation (13-15), impair cerebral autoregulation (15, 16), and aggravate TBI-induced structural damage as reflected by a significant increase in contusion volume (9, 17-19). Secondary hypoxia has also been reported to exacerbate acute sensorimotor and cognitive deficits following moderate fluid percussion (FP) brain injury in rats (20). To date, most preclinical pathophysiologic and pharmacologic studies have been conducted primarily in animals subjected to isolated TBI without HH, and a strong need exists to develop combined injury models to elucidate optimal treatment and management strategies for multiply-injured patients (see the excellent review in Ref. 21). Although several rodent models of combined injury (TBI with hypoxia, with and without HH) have been established (9, 15, 17, 20, 22, 23), experimental studies investigating the impact of superimposed HH have focused primarily on the pharmacologic restitution of mean arterial blood pressure (MABP) and/or cerebral perfusion pressure by infusing catecholamines (15, 24) and the evaluation of various resuscitation fluids (13, 25, 26). To date, the impact of hemorrhagic hypotension on neurologic and cognitive outcome following TBI has not been investigated.
The present study was designed to reproduce the clinical scenario in which a TBI patient experiences an acute, transient (≤30 mins) arterial hemorrhage immediately following TBI. In one subgroup of brain-injured animals, lactated Ringer’s solution was infused to reverse the arterial hypotension whereas another subgroup was not fluid resuscitated. The impact of superimposed compensated or uncompensated HH on the acute pathologic response to TBI (brain edema formation at 24 hrs postinjury), long-term (5 month) neurobehavioral outcome variables, and hemispheric tissue loss after TBI was also evaluated in the current study.
MATERIALS AND METHODS
Animal Groups
A total of 119 male Sprague Dawley rats (392 ± 22.9 g) were included in the present investigation. All animal procedures followed the National Institutes of Health Guide for the Care and Use of Laboratory Animals (27).
Anesthesia
All rats were anesthetized with sodium pentobarbital (Nembutal®, Abbott Laboratories, North Chicago, IL; 60 mg/kg intraperitoneally). To maintain anesthesia, supplemental pentobarbital was injected at 60 mins after FP injury/HH at a lower dose (15 mg/kg intraperitoneally) to avoid cardiorespiratory depression related to systemic pentobarbital accumulation. For the additional measurements of MABP and arterial blood gases at 24 or 48 hrs, rats were anesthetized with a lower dose (40-50 mg/kg intraperitoneally of sodium pentobarbital). Before kill and brain removal, rats were anesthetized with pentobarbital (60 mg/kg intraperitoneally).
Body Temperature, Mean Arterial Blood Pressure, and Arterial Blood Gases
Within 10 mins following the initial pentobarbital injection, all animals were positioned on a homeothermic heating pad, and body temperature was maintained between 36.5 and 37.5°C during the entire study period. Using a surgical microscope, the left groin was incised and the left femoral artery and vein were catheterized with commercially available polyethylene tubing (PE 50, inner diameter 0.58 mm, outer diameter 0.965 mm; Becton Dickinson, Sparks, MD). The catheters were advanced to a depth of 2 cm. To reduce the incidence of muscle ischemia (observed during our pilot studies in the cannulated leg of FP brain-injured rats subjected to superimposed HH), the arterial catheter was modified by inserting a PE 10 tubing (inner diameter 0.28 mm, outer diameter 0.61 mm) into the tip of the PE 50 tubing. MABP was continuously monitored with the Cardiomax II/85 (Columbus Instruments, Columbus, OH) before TBI-HH, during and after HH, and during subsequent fluid resuscitation. Anesthetized rats were recannulated at 24 hrs, 48 hrs, and 5 wks via the left (24 and 48 hrs) or the right femoral artery (5 wks) to asses the prolonged effect of superimposed HH on blood pressure and blood gases.
Arterial blood gases were determined before TBI-HH, directly after HH, following fluid resuscitation, and again at 24 hrs, 48 hrs, and 1 wk after TBI-HH using the precalibrated cartridges for the bedside with an i-Stat blood gas analyzer (Abbott Laboratories, North Chicago, IL). Based on the hemoglobin, arterial oxygen saturation (Sao2), and Pao2 values, the arterial oxygen content was calculated according to the following equation: cao2 = ([1.36 × Hb × Sao2] + [0.003 × Pao2]) where (1.36 × Hb × Sao2) reflects the amount of bound oxygen and (0.003 × Pao2) represents the free, soluble oxygen transported in blood.
Induction of Traumatic Brain Injury
Following the initial cannulation of femoral artery and vein, rats were positioned in a stereotactic frame, and a craniectomy was performed over the left parietal cortex within the anatomical boundaries outlined by the sagittal, anterior, and posterior cranial sutures. A hollow female Luer lock was positioned inside the craniectomy and held in place with glue and dental cement. Filling of the Luer lock with physiologic saline solution (0.9%) prevented drying and damaging of the dura and underlying cortex. At 90 mins after pentobarbital injection, the rats (89 of 119) were connected to the FP device via the Luer lock, and a brain injury of mild to moderate severity (2.6-2.8 atm) was induced by rapidly injecting a pressure pulse of saline solution onto the exposed left parietotemporal sensory cortex as originally described (28). After the brain injury, the Luer lock and dental cement were removed and the brain was visually inspected for signs of dural breaches using the surgical microscope. Any animal with a compromised dura was excluded, as the extent of injury was artifactually lowered due to the breach, and the animal was immediately killed. Within 2 mins after injury the scalp was sutured, and by 4 mins rats were reconnected to the pressure transducer and pressure-controlled HH was initiated. Sham-injured animals (n = 22) received anesthesia and surgery without brain injury.
Superimposed Hemorrhagic Hypotension and Fluid Resuscitation
After reconnecting the animal to the Cardiomax monitor, one subgroup of brain-injured animals (n = 47) were subjected to pressure-controlled HH beginning 5 mins after TBI. A second group of uninjured animals (n = 8) were subjected to isolated HH. The MABP was maintained between 50 and 60 mm Hg for 30 mins. To this end, an estimated 30% of the total blood volume based on the calculations of Wang and colleagues (28a), equaling 6% of the body weight, was withdrawn via the arterial catheter, resulting in a blood loss of 6-7 mL/rat. Rats assigned to the compensated HH group (n = 18) received lactated Ringer’s solution, which was infused over 30 mins at three times the shed blood volume starting 10 mins after the end of the HH period. The uncompensated HH rats (n = 29) did not receive any fluid resuscitation (Table 1 shows group allocation).
Table 1.
Overview of study group allocation and kill time
| Combined Injury (FP and HH) |
|||||||
|---|---|---|---|---|---|---|---|
| Group | Kill Time | Sham | HH | Isolated FP | Total | Compensated | Uncompensated |
| Initial | 22 | 8 | 34 (+8 initial mortality) | 47 | 18 | 29 | |
| Edema | 24 hrs | 8 | - | 9 | 15 | 7 | 8 |
| Lesion area | 7 days | 7 | 8 | 9 | 13 | - | 13 |
| Behavior | 8/20 wks | 7 | - | 16 | 19 | 11 | 8 |
| Total | 22 | 8 | 34 (+8) | 47 | 18 | 29 | |
| n | 111 (+8) = 119 | ||||||
FP, fluid percussion; HH, hemorrhagic hypotension.
Analysis of Brain Edema Formation
At 24 hrs after isolated FP brain injury (n = 9) or FP brain injury with uncompensated (n = 8) or compensated superimposed HH (n = 7), brain-injured and sham-injured control rats (n = 8) were re-anesthetized and recannulated. Following measurement of MABP and blood withdrawal for arterial blood gases, rats were exsanguinated by withdrawing 8-11 mL of blood within 1 min to reduce an artificial increase in cerebral water content due to the remaining blood within the cerebral vessels and venous sinus. Thereafter, the rats were decapitated and the brains removed quickly. Brains were dissected at approximately -4 mm from bregma (3 mm thick slice) to evaluate regional edema formation in the injured and adjacent cortex, the corresponding contralateral cortical regions, and both hippocampal and thalamic structures as previously described (29). The brain sections were weighed to determine wet weight, dried at 104°C, and then reweighed after 24 hrs to assess dry weight. Based on the dry and wet weight values, the regional water content expressed in percent was calculated according to the following equation
| [1] |
Changes in cerebral water content were determined by an investigator blinded to the injury/treatment status of the rat.
Neurologic Deficit
Neurologic performance was evaluated as early as 24 hrs after injury in those rats used to assess edema formation. Persistence of the neurologic deficit was determined through neurologic evaluation at 1, 3, 5, 7, 9, 12, 16, and 20 wks postinjury. Motor function was assessed using the composite neuroscore test that has been previously described (30). In brief, extension and flexion of the fore- and hindlimbs as well as resistance to lateral pulsion were scored according to a standard protocol comparing the right vs. left side in each rat between the different groups of rats. Neurologic motor deficits were evaluated by two investigators blinded to the status of these rats.
Cognitive Deficits
Beginning at 4 wks following FP brain injury with or without HH (compensated or uncompensated), injured and sham-injured rats were subjected to a consecutive 3-day learning paradigm in a 1.8-m Morris water maze with the objective of localizing a submerged platform within 60 secs using visuospatial cues posted on the walls surrounding the water-filled tank as previously described (31). Once the rats reached the platform, they were allowed to remain on the platform for 15 secs. If the rats did not reach the platform within 60 secs, they were manually guided to the platform. All animals completed a total of eight trials per day (four in the morning, four in the afternoon) for 3 days. At 8, 12, and 16 wks postinjury, the same test was repeated to evaluate long-term memory formation. The established laboratory criterion of ≤12 secs (mean) was used to determine the percentage of animals at weeks 4, 8, 12, and 16 that successfully completed the learning/memory task. The actual tracking patterns of swim distance were continuously recorded, to allow for analysis of swimming behavior and searching pattern and to calculate the individual swim speed. The Morris water maze experiments were conducted by an investigator blinded to the injury status of the animals.
Assessment of Cortical Tissue Loss
In a subset of injured animals at 1 wk postinjury (sham, n = 7; FP injury only, n = 7; FP injury and HH, n = 9), the volume of cortical tissue loss was determined using image analysis software (MCID, Imaging Research, St. Catharines, Ontario, Canada). Sections from bregma -0.3 to bregma -6.3 (32) were stained with hematoxylin and eosin, and the area of the contralateral and ipsilateral cortexes was then measured using a light microscope and image analysis system, as described previously (33). The cortexes of both hemispheres were outlined by hand by an evaluator blinded to injury and shock status, and the cortical areas were computed by the calibrated image analysis program. The lesion area (mm2) was then calculated with the following formula: contralateral cortex - ipsilateral cortex. The total lesion volume (mm3) was calculated by integrating the cortical lesion area over the 6-mm rostro-caudal distance using the following formula:
Statistical Analysis
All results were entered into a statistical software package (SigmaStat 3.0; SPSS, Chicago, IL) for analysis. Graphs and figures were created using SigmaPlot 8.02 (SPSS). Distribution of data were evaluated using the Shapiro-Wilks normality test. Kruskal-Wallis analysis of variance (ANOVA) was used to evaluate the group differences in the components of the neuroscore at each time point, followed by individual Mann-Whitney U post hoc tests. ANOVA was used to evaluate differences in brain water content; the volume of cortical tissue lost at 1 wk postinjury and among sham-injured, brain-injured, and brain-injured plus HH groups; and hematocrit and arterial oxygen content followed by the Student Newman-Keuls’ post hoc test when appropriate. Repeated-measures ANOVA was used to evaluate differences in MABP among the groups over time followed by the Student Newman-Keuls’ post hoc test when appropriate. Morris water maze latencies were not normally distributed; therefore, the ability of animals to perform the learning/memory test was evaluated using Kruskal-Wallis ANOVA on each trial block followed by Mann-Whitney U post hoc tests with Bonferonni correction. As fluid resuscitation following FP-HH had no effect on motor or cognitive function, these groups were combined for analysis and ease of data presentation. The range of the arithmetical mean is presented using sd.
RESULTS
Physiology and Mortality
The overall mortality rate caused by the FP brain injury was 9% (eight of 89 FP brain-injured animals) during the first 24 hrs, consistent with previous reports modeling mild TBI (28). Following the first 24 hrs, five brain-injured animals had to be killed over the 5-month study period due to excessive weight loss; their data are included where available and appropriate.
MABP and Arterial Blood Gases
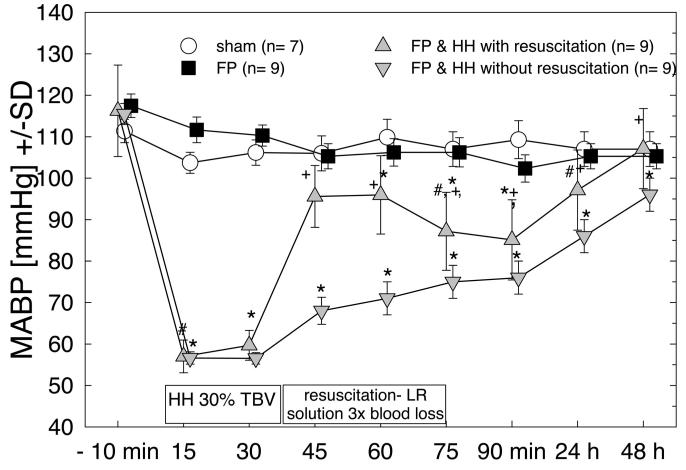
Whereas MABP remained unchanged in the uninjured controls and rats subjected to isolated TBI, MABP was significantly reduced from 115 ± 2 to 58 ± 2 mm Hg (p < .001) during the 30-min phase of arterial HH (Fig. 1). During infusion of lactated Ringer’s solution, MABP significantly increased to 96 ± 8 mm Hg (p < .001) without reaching preinjury and prehemorrhage levels (Fig. 1). Within the first 30 mins after the cessation of fluid resuscitation, MABP again decreased to 85 ± 10 mm Hg (p = .008) and increased slowly thereafter to reach baseline values within the following 24-48 hrs (Fig. 1). Unresuscitated animals exhibited a slow but continuous increase of MAPB over time following the arterial HH period. However, the MABP of unresuscitated animals remained significantly below that of the resuscitated group at all time points and had almost reached baseline levels by 1-2 days after the injury (96 ± 11 mm Hg, Fig. 1).
Figure 1.
Mean arterial blood pressure (MABP) change during the first 48 hrs following fluid percussion (FP) or FP-hemorrhagic hypotension (HH). MABP was significantly reduced in the FP-HH group compared with the isolated fluid percussion injured rats and the noninjured controls. Fluid resuscitation with lactated Ringer’s (LR) solution significantly increased MABP only transiently in brain-injured rats subjected to hemorrhagic hypotension, which remained significantly decreased compared with the control levels. Following fluid resuscitation, MABP continued to increase over time, reaching control levels at 48 hrs. In brain-injured rats subjected to hemorrhagic hypotension without fluid resuscitation, MABP slowly increased over time but remained significantly lower compared with the fluid resuscitated rats. Repeated-measures analysis of variance contrasts: *p < .01 compared with sham or FP alone; +p < .01 compared with nonresuscitated FP and HH counterparts; #p < .05 compared with previous time point. TBV, total blood volume
The acute withdrawal of 6-7 mL of blood also resulted in a significant reduction of arterial hematocrit and arterial oxygen content (p < .005). Fluid resuscitation with lactated Ringer’s solution did not ameliorate these changes at 30 mins and 24 hrs after hemorrhagic hypotension (data not shown). Arterial Pao2 and Sao2 values were similar in all rats before, during, and after hemorrhagic hypotension, irrespective of fluid resuscitation (data not shown).
Edema Formation
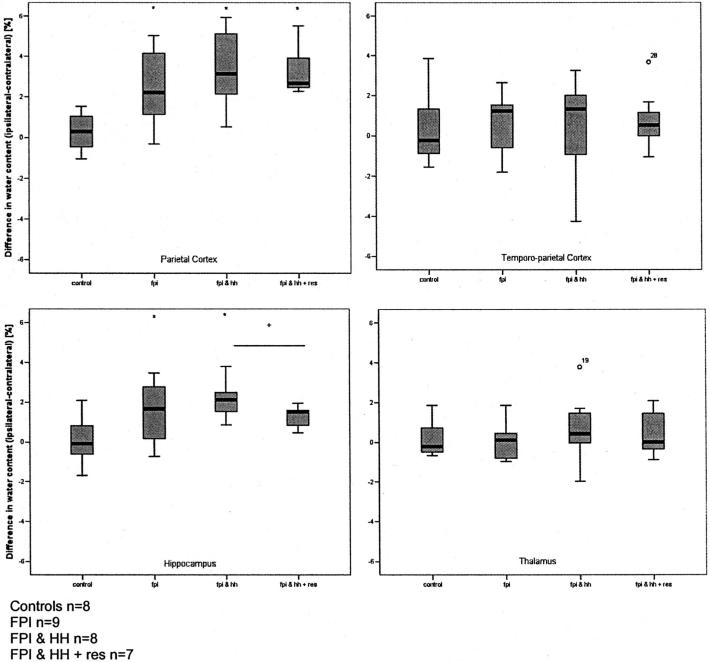
Compared with the uninjured control rats, FP brain injury significantly increased edema formation in the parietal cortex (injury area) and ipsilateral hippocampus (p < .0005) but not in the temporoparietal cortex (adjacent cortex) or ipsilateral thalamus at 24 hrs postinjury (Fig. 2). Superimposed HH did not aggravate the underlying edema formation. Surprisingly, fluid resuscitation significantly reduced edema formation (p = .04) in the hippocampus of the injured hemisphere (Fig. 2).
Figure 2.
Regional brain water content (cerebral edema) at 24 hrs following fluid percussion injury (FPI) or FPI-hemorrhagic hypotension (HH) in rats. FPI significantly increased cerebral water content in the ipsilateral parietal cortex and hippocampus as reflected by the calculated difference between the left (injured) and right hemisphere. Superimposed hemorrhagic hypotension had no effect on underlying edema formation. *p < .005; +p = .004.
Neurologic Impairment
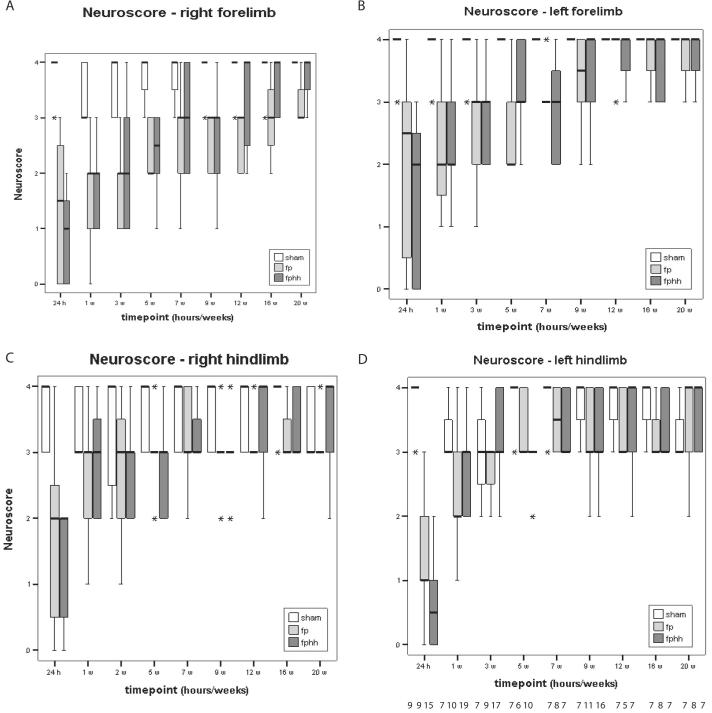
Compared with sham-injured controls, FP brain injury significantly impaired bilateral fore- and hindlimb reflexes (Fig. 3) and resistance to lateral pulsion (p < .001). For the right forelimb, the deficit persisted up to 20 wks, whereas reflex behavior returned in the left forelimb by 9 wks (Fig. 3, A and B). Function of the hindlimbs was significantly reduced bilaterally at 24 hrs and on the left side for up to 1 wk (Fig. 3, C and D). At 24 hrs, the impairment of hindlimb function was more severe in the cannulated left leg compared with the right hindlimb in animals subjected to HH alone and FP and HH combined. Resistance to lateral pulsion following FP brain injury was significantly reduced (p < .001) for 5 wks (FP, 3.1 ± 0.7; FP-HH, 3.0 ± 0.6) on the left side (ipsilateral) when compared with sham-injured animals (3.7 ± 0.5). On the contralateral side, this deficit persisted for 3 wks (p < .001) in injured animals (FP, 2.7 ± 0.8; FP-HH, 2.9 ± 0.7; sham, 3.6 ± 0.8). Superimposed HH with or without fluid resuscitation in the acute postinjury period did not aggravate the brain injury induced neurologic deficits (data not shown).
Figure 3.
Neurologic motor function following fluid percussion (fp) and FP-hemorrhagic hypotension (fphh) over a 20-wk postinjury period. Temporal profile of the scored neurologic deficit determined for the right (A) and left (B) forelimbs, right (C) and left (D) hindlimbs in control rats (white bars), isolated brain injured rats (light gray bars), and hemorrhaged brain-injured rats with and without fluid resuscitation (dark gray bars). The bilateral deficit was most pronounced in the contralateral forelimb and persisted as long as 20 wks, without being aggravated by the superimposed hemorrhagic hypotension. Cannulation of the left femoral artery impaired left leg function in the brain-injured and hemorrhaged rats. Fluid resuscitation had no influence on neurologic function (data not shown). *p < .001.
Learning, Memory Formation, and Information Retrieval
Sixty-four percent of sham-injured rats learned the task of finding the submerged platform (Table 2) in the 1.8-m water maze at 4 wks postinjury. By 8 wks postinjury, all shaminjured rats had met criteria for learning (Table 2). FP brain injury significantly impaired learning latencies up to 8 wks postinjury (mean = 19 ± 15 secs for FP injury vs. 8 ± 1 secs for sham-injured controls, Fig. 4, Table 2). Hemorrhagic hypotension with or without fluid resuscitation in the acute postinjury period did not aggravate the magnitude of TBI-induced impaired learning deficits (Fig. 4). However, the learning latencies of animals with combined injury remained significantly increased compared with sham-injured rats up to 12 wks postinjury (57% of animals did not meet criteria, p < .05, Table 2). At week 16, day 3, mean learning latencies in FP-HH animals remained significantly longer compared with sham injured animals (Fig. 4). Moreover, the differences in learning latencies between isolated FP-brain injured animals and sham-injured controls were not significant when evaluated at 12 and 16 wks after surgery. The learning latencies of combined FP-HH animals were approximately 1 month delayed when compared with animals subjected to mild to moderate TBI alone (Table 2). These data indicate that the time course of recovery of cognitive function was significantly delayed by the superimposition of hemorrhagic hypotension after TBI.
Table 2.
Percentage of animals able to successfully complete the learning/memory task
FP, fluid percussion; HH, hemorrhagic hypotension.
p < .05 in comparison to sham. Values are percentages. There were significant injury related deficits in both the isolated FP and the FP and HH groups up to 8 weeks postinjury, but only the FP and HH group had a significantly lower percentage of animals able to complete the task at 12 weeks postinjury.
Figure 4.

Cognitive (learning/memory) function in animals subjected to fluid percussion injury (FPI) and FPI-hemorrhagic hypotension (HH). FPI significantly impaired learning ability from 1 day through 8 wks postinjury whereupon animals recovered their learning ability (*p < .025 in comparison to sham). The superimposition of HH caused a significantly longer duration of cognitive dysfunction up to 16 wks postinjury (+p < .025 compared with sham-injured controls).
Histopathologic Damage
Experimental FP brain injury resulted in a statistically significant loss of cortical tissue (corresponding to the area of edema) compared with sham-injured animals (Fig. 5). The superimposition of HH did not exacerbate cortical cell death following FP brain injury.
Figure 5.

Volumetric analysis of cortical tissue loss following fluid percussion (FP) or FP-hemorrhagic hypotension (HH) in rats. FP injury significantly increased the volume of cortical tissue lost over sham (n = 7) regardless of group (*p = .001) 1 wk postinjury. Superimposed HH (n = 9) had no effect on the amount of cell death when compared with FP alone (n = 7, p = .25).
DISCUSSION
In the present study, acute regional brain edema formation and hemispheric tissue loss as well as chronic neurologic motor deficits induced by isolated FP brain injury of mild to moderate severity were not aggravated by a superimposed, acute, and transient HH insult of 30 mins duration. However, the addition of HH significantly delayed the recovery of cognitive (learning/memory) function that was observed to occur at 12 and 16 wks following isolated TBI. Fluid resuscitation following the hypotensive period transiently improved arterial blood pressure and lessened brain edema formation in the ipsilateral hippocampus but had no effect on behavioral outcome.
Questions regarding the effects of HH on outcome after TBI have been raised by clinical investigations in multiple trauma patients (4, 12, 34). In a retrospective multiple-center study of 49,143 patients from 95 trauma centers, Gennarelli et al. (34) reported that a) the overall mortality rate of trauma patients was three times higher if a head injury occurred; b) mortality rate was primarily influenced by severe coexisting extracranial injuries; and c) head injury was the most important contributor to trauma center deaths. Chesnut and colleagues (4) analyzed the data from the National Traumatic Coma Data Bank to assess the impact of early and late hypotension on outcome and observed that early hypotension, present in 34.6% of the patients, was associated with a doubling in mortality rate (55% vs. 27%). Delayed hypotension, which occurs in 32% of all patients, was also associated with death or vegetative state, even with only one documented hypotensive episode. It remains unclear whether the hypotension observed in these studies was associated with more severe TBI or if its occurrence was unrelated to the severity of TBI and whether the worsening of outcome might have been related to factors that are directly or indirectly related to the hypotension, such as the inflammatory response.
In more recent prospective studies, the outcome of patients suffering severe TBI with and without extracranial injuries was reported to be comparable (2). This is likely due to the improved management of TBI patients and the introduction of clinical guidelines (5, 35). Sarrafzadeh et al. (2) concluded that multimodal monitoring and targeted management, including intracranial pressure/cerebral perfusion pressureguided therapy as well as delayed surgery for extracranial lesions, markedly reduced secondary insults and the occurrence of unfavorable outcome. These recent reports raise the question whether an isolated event of hemorrhagic hypotension is related to outcome in severely injured TBI patients. Virtually nothing is known, to date, regarding the effect of acute HH on outcome following mild to moderate TBI.
Development of a Clinically Relevant Model of HH and TBI
As outlined by Statler and colleagues (21), the clinically predictive value of current experimental models of TBI is limited by the fact that the majority of experimental approaches are often restricted to isolated TBI. In an initial attempt to increase the clinical relevance of well-established model of lateral FP brain injury, we subjected brain-injured rats to an additional superimposed HH by withdrawing 30% of the calculated total blood volume, thereby reducing the MABP by approximately 50% to 50-60 mm Hg for 30 mins. The underlying rationale for the present study was related, in part, to the observation that a similar transient decrease in MABP followed by autotransfusion of the withdrawn or pooled blood increased cortical contusion volume at 24 hrs and 7 days in more severely brain-injured rats (9, 18). As the present results reveal, the superimposed transient HH in the acute posttraumatic period following mild to moderate TBI was not sufficient to worsen the underlying progressive structural damage and long-term (4 months) motor impairment induced by FP brain injury alone. However, the recovery of cognitive function that occurred in brain-injured animals by 12 wks was significantly delayed (up to 16 wks) in the HH-TBI group. It is possible that the instruments used in this study are not sensitive enough to detect small differences in motor outcome. Future studies should include a more rigorous neuromotor test such as the beam or gridwalk test. It might be necessary to increase the injury severity or add additional clinically relevant factors to mimic the clinical situation more closely. Holtzer and colleagues (15) reported that the addition of hypoxia to the underlying HH was necessary to impair catecholamine-mediated vasoconstriction and thus attenuate pressure regulation following TBI in rats. Since we determined here that HH alone did not compromise arterial oxygenation, it is also possible that the further addition of hypoxia may result in more severe and readily detectable exacerbation of TBI-induced motor deficits and histopathologic damage.
Interestingly, multiple trauma patients without TBI have increased serum levels of proinflammatory cytokines, and the addition of TBI to multiple trauma seems to significantly augment the systemic inflammatory response (36). Since the occurrence of HH in severely brain-injured human patients is typically associated with multiple trauma, it is possible that the reported worsening of outcome is associated with a systemic inflammatory response to injury rather than to the HH alone.
Pilot studies, performed in our laboratory, showed that increasing the severity of the injury or duration of the HH resulted in a significant increase in muscle infarction of the cannulated hindlimb (from approximately 10% to >75%) and an unacceptable increase in mortality rate. The pilot studies revealed a linear relationship between brain injury severity and the percentage of animals developing leg infarctions, supporting the concept of a systemic inflammatory response following injury. Therefore, in our study we chose to subject the rats to FP brain injury of mild to moderate severity followed by a moderate HH (30% of total blood volume), a nonlethal combination that resulted in a low incidence of limb infarcts (<10%). Any functional deficits potentially associated with hindleg ischemia were reversible within 10 days and did not interfere with the rat’s swimming abilities at 4, 8, 12, and 16 wks as evidenced by the neurologic evaluation of the hindlimbs and swim speed analysis (data not shown). Our model of mild to moderate brain injury, moderate HH, and femoral catheterization allowed animals to survive without resuscitation, but in additional pilot studies, further increases in either the level of hemorrhage or brain injury resulted in limb ischemia or high levels of mortality (data not shown). Future work is being directed at adapting this model using tail artery catheterization to allow for further manipulation in both level of brain injury and hemorrhage without the same adverse events.
Histopathologic Damage
Our results are partially in contrast to recent studies reporting a significant increase in focal tissue injury or sustained neurologic deficit related to the superimposed HH in more severe models of weight-drop brain injury in rats (37). Interestingly, using a mild FP brain injury model, Matushita et al. (9) reported an exacerbation of tissue damage with the superimposed HH (MABP 50-60 mm Hg for 30 mins). It is possible, therefore, that the greater extent of tissue damage subsequent to the higher injury severity in our study might have obscured the effect of superimposed shock on cortical cell loss. Additionally, as per animal welfare guidelines, animals were anesthetized preinjury with pentobarbital, known to have neuroprotective properties (38), which could also have diminished the effects of injury on cortical cell loss; however, all anesthetic agents used in FP injury provide some degree of neuroprotection but have not been studied extensively following injury (39). Moreover, in our study, injured animals either were fluid resuscitated with lactated Ringer’s solution or remained unresuscitated. In the previous reports, the autotransfusion of heparinized, shed blood might have had a deleterious effect on the injured brain, thereby contributing to the increase in contusion volume. The storage of heparinized blood at room temperature appears to result in a gradual increase in plasma glutamate when intracellular glutamate penetrates the damaged erythrocytic cell membrane (JF Stover, unpublished data, 2001) either following its concentration gradient (40) or in an effort to counterregulate evolving edema. Increased plasma glutamate concentrations, which, in combination with generated free radicals and liberated hemoglobin, are known to bind nitric oxide and inhibit nitric oxide-mediated vasodilation (41), might therefore have aggravated the evolving secondary tissue damage (17, 18, 20, 24, 42).
Our results show an increase in brain edema formation in the parietal cortex, hippocampus, and thalamus following TBI that was not exacerbated by superimposed hemorrhagic hypotension (Fig. 2). We were surprised to find that fluid resuscitation significantly reduced edema in the hippocampus, suggesting that fluid resuscitation can limit tissue damage and swelling in this selectively vulnerable region, most likely by improving circulation. Previous reports have suggested that the administration of large volumes of iso-osmolar fluid does not interfere with brain edema in models of head injury (43). As crystalloid solutions do not remain in the vascular space for an extended period of time, this improvement in circulation may be transient, and future studies should evaluate the utility of combined fluid resuscitation. An evaluation of ultrastructural damage using electron microscopy or other techniques may also prove to be a useful addition to future combined models studies to discern the mechanisms of edema formation and cell loss.
Neurologic and Cognitive Function
In the present study, the bilateral impairment of forelimb extension and flexion with bilateral spasticity suggests that the lateral FP brain injury induces widespread damage to the motor pathways and white matter. Over time, the observed neurologic deficit shows a slight improvement, with the contralateral forelimb showing the most prolonged injury-induced deficit, up to 12 wks postinjury, similar to previously published results (30). With the exception of contralateral (right) forelimb flexion test, all indexes of motor function recovered to sham-injured control values within the 4-month evaluation period following mild to moderate TBI. The superimposition of HH did not exacerbate motor dysfunction nor did it inhibit or retard neuromotor recovery. Our results confirm those of Eilig et al. (44), who found no effect of different resuscitation fluids on neurologic status following weight-drop closed head injury and uncontrolled hemorrhage.
The complex cortico-cortical, hippocampo-cortical, and cortico-hippocampal projection circuits are integral and important components of the system necessary to generate, store, and retrieve encoded information (45). Functional and mechanical injury to both the hippocampus and different cortical areas can significantly impair spatial navigation and memory formation (46, 47). Experimentally induced FP brain injury or controlled cortical impact brain injury results in learning and memory deficits by disrupting cortico-hippocampal connections and inducing characteristic hippocampal neuronal cell loss in the CA3 and hilar region (47, 48). Prolonged memory impairment has also been described in the absence of hippocampal and cortical cell death following FP (49) and controlled cortical impact injury in rats (50). The superimposition of HH significantly reduced the rate of long-term cognitive recovery observed in animals subjected to TBI alone. Animals subjected to combined injury were 1 month delayed in reaching learning criteria compared with those subjected to mild to moderate TBI alone.
CONCLUSION
Overall, the superimposition of acute, transient uncompensated or compensated hemorrhagic hypotension did not aggravate the long-term neurologic motor deficits but did significantly retard long-term cognitive recovery associated with lateral FP brain injury of mild to moderate severity. Further studies should be designed to mimic the complex situation of multiple trauma more closely by including and controlling for key events such as peripheral fractures, inflammatory response, hypothermia, and/or fluid resuscitation. Increasing the severity of TBI might render the brain more vulnerable to secondary injury and/or induce relevant key events of multiple trauma.
Overall, the superimposition of acute, transient uncompensated or compensated hemorrhagic hypotension did not aggravate the long-term neurologic motor deficits but did significantly retard long-term cognitive recovery associated with lateral fluid percussion brain injury of mild to moderate severity.
Acknowledgments
Supported, in part, by grant NIGMS GM RO1-34690 from the National Institutes of Health; grants NS P50-08803 (University of Pennsylvania, Philadelphia) and NINR T32-NR07106 (University of Washington, Seattle, HJT) from NINDS; a Veterans Administration Merit Review grant (Veteran’s Administration Medical Center, Philadelphia); and the Endowed Chair, Department of Anesthesia and Critical Care Medicine, at the Children’s Hospital of Philadelphia.
The authors have no financial interests to disclose.
Footnotes
Christian Schütz and John F. Stover contributed equally to this project.
REFERENCES
- 1.Gobiet W. Effect of multiple trauma on rehabilitation of patients with craniocerebral injuries [in German] Zentralbl Chir. 1995;120:544–550. [PubMed] [Google Scholar]
- 2.Sarrafzadeh AS, Peltonen EE, Kaisers U, et al. Secondary insults in severe head injury—Do multiply injured patients do worse? Crit Care Med. 2001;29:1116–1123. doi: 10.1097/00003246-200106000-00004. [DOI] [PubMed] [Google Scholar]
- 3.Welkerling H, Wening JV, Langendorff HU, et al. Computer-assisted data analysis of injuries of the skeletal system in polytrauma patients [in German] Zentralbl Chir. 1991;116:1263–1272. [PubMed] [Google Scholar]
- 4.Chestnut RM, Marshall LF, Kauber MR, et al. The role of secondary brain injury in determining outcome from severe head injury. J Trauma. 1993;34:216–222. doi: 10.1097/00005373-199302000-00006. [DOI] [PubMed] [Google Scholar]
- 5.Maas AI, Dearden M, Teasdale GM, et al. EBIC-guidelines for management of severe head injury in adults. European Brain Injury Consortium. Acta Neurochir. 1997;139:286–294. doi: 10.1007/BF01808823. [DOI] [PubMed] [Google Scholar]
- 6.Vincent J, Berre J. Primer on medical management of severe brain injury. Crit Care Med. 2005;33:1392–1399. doi: 10.1097/01.ccm.0000166890.93559.2d. [DOI] [PubMed] [Google Scholar]
- 7.Siegel JH. The effect of associated injuries, blood loss, and oxygen debt on death and disability in blunt traumatic brain injury: The need for early physiologic predictors of severity. J Neurotrauma. 1995;12:579–590. doi: 10.1089/neu.1995.12.579. [DOI] [PubMed] [Google Scholar]
- 8.Heckbert SR, Vedder NB, Hoffman W, et al. Outcome after hemorrhagic shock in trauma patients. J Trauma. 1998;45:545–549. doi: 10.1097/00005373-199809000-00022. [DOI] [PubMed] [Google Scholar]
- 9.Matsushita Y, Bramlett HM, Kuluz JW, et al. Delayed hemorrhagic hypotension exacerbates the hemodynamic and histopathologic consequences of traumatic brain injury in rats. J Cereb Blood Flow Metab. 2001;21:847–856. doi: 10.1097/00004647-200107000-00010. [DOI] [PubMed] [Google Scholar]
- 10.DeWitt DS, Prough DS, Taylor CL, et al. Reduced cerebral blood flow, oxygen delivery and electroencephalographic activity after traumatic brain injury and mild hemorrhage in cats. J Neurosurg. 1992;76:812–821. doi: 10.3171/jns.1992.76.5.0812. [DOI] [PubMed] [Google Scholar]
- 11.Levasseur JE, Alessandri B, Reinert M, et al. Fluid percussion injury transiently increases then decreases brain oxygen consumption in the rat. J Neurotrauma. 2000;17:101–112. doi: 10.1089/neu.2000.17.101. [DOI] [PubMed] [Google Scholar]
- 12.Manley GT, Hemphill JC, Morabito D, et al. Cerebral oxygenation during hemorrhagic shock: Perils of hyperventilation and the therapeutic potential of hypoventilation. J Trauma. 2000;48:1025–1032. doi: 10.1097/00005373-200006000-00005. [DOI] [PubMed] [Google Scholar]
- 13.Yuan X-Q, Wade CE, Clifford CB. Suppression by traumatic brain injury of spontaneous hemodynamic recovery from hemorrhagic shock. J Neurosurg. 1991;75:408–414. doi: 10.3171/jns.1991.75.3.0408. [DOI] [PubMed] [Google Scholar]
- 14.Law MM, Hovda D, Cryer HG. Fluidpercussion brain injury adversely affects control of vascular tone during hemorrhagic shock. Shock. 1996;6:213–217. [PubMed] [Google Scholar]
- 15.Holtzer S, Vigue B, Ract C, et al. Hypoxiahypotension decreases pressor responsiveness to exogenous catecholamines after severe traumatic brain injury in rats. Crit Care Med. 2001;29:1609–1614. doi: 10.1097/00003246-200108000-00018. [DOI] [PubMed] [Google Scholar]
- 16.DeWitt DS, Prough DS, Taylor CL, et al. Regional cerebrovascular responses to progressive hypotension after traumatic brain injury in cats. Am J Physiol. 1992;263:H1276–H1284. doi: 10.1152/ajpheart.1992.263.4.H1276. [DOI] [PubMed] [Google Scholar]
- 17.Bramlett HM, Green EJ, Dietrich WD. Exacerbation of cortical and hippocampal CA1 damage due to posttraumatic hypoxia following moderate fluid-percussion brain injury in rats. J Neurosurg. 1999;91:653–659. doi: 10.3171/jns.1999.91.4.0653. [DOI] [PubMed] [Google Scholar]
- 18.Kroppenstedt S-N, Kern M, Thomale UW, et al. Effect of cerebral perfusion on contusion volume following impact injury. J Neurosurg. 1999;90:520–526. doi: 10.3171/jns.1999.90.3.0520. [DOI] [PubMed] [Google Scholar]
- 19.Matsushita Y, Bramlett HM, Alonso O, et al. Posttraumatic hypothermia is neuroprotective in a model of traumatic brain injury complicated by a secondary hypoxic insult. Crit Care Med. 2001;29:2060–2066. doi: 10.1097/00003246-200111000-00004. [DOI] [PubMed] [Google Scholar]
- 20.Bramlett HM, Dietrich WD, Green EJ. Secondary hypoxia following moderate fluid percussion brain injury in rats exacerbates sensorimotor and cognitive deficits. J Neurotrauma. 1999;16:1035–1047. doi: 10.1089/neu.1999.16.1035. [DOI] [PubMed] [Google Scholar]
- 21.Statler KD, Jenkins LW, Dixon CE, et al. The simple model versus the super model: Translating experimental traumatic brain injury research to the bedside. J Neurotrauma. 2001;18:1195–1206. doi: 10.1089/089771501317095232. [DOI] [PubMed] [Google Scholar]
- 22.Ishige N, Pitts LH, Berry I, et al. The effect of hypoxia on traumatic brain injury in rats: Alterations in neurologic function, brain edema, and cerebral blood flow. J Cereb Blood Flow Metab. 1987;7:759–767. doi: 10.1038/jcbfm.1987.131. [DOI] [PubMed] [Google Scholar]
- 23.Jenkins LW, Moszynski K, Lyeth BG, et al. Increased vulnerability of the mildly traumatized rat brain to cerebral ischemia: The use of controlled secondary ischemia as a research tool to identify common or different mechanisms contributing to mechanical and ischemic brain injury. Brain Res. 1989;477:211–224. doi: 10.1016/0006-8993(89)91409-1. [DOI] [PubMed] [Google Scholar]
- 24.Kroppenstedt SN, Sakowitz OW, Thomale UW, et al. Influence of norepinephrine and dopamine on cortical perfusion, EEG activity, extracellular glutamate, and brain edema in rats after controlled cortical impact injury. J Neurotrauma. 2002;19:1421–1432. doi: 10.1089/089771502320914651. [DOI] [PubMed] [Google Scholar]
- 25.Hartl R, Schurer L, Goetz C, et al. The effect of hypertonic fluid resuscitation on brain edema in rabbits subjected to brain injury and hemorrhagic shock. Shock. 1995;3:274–279. doi: 10.1097/00024382-199504000-00005. [DOI] [PubMed] [Google Scholar]
- 26.Schmoker JD, Zhuang J, Shackford SR. Hypertonic fluid resuscitation improves cerebral oxygen delivery and reduces intracranial pressure after hemorrhagic shock. J Trauma. 1991;31:1607–1613. doi: 10.1097/00005373-199112000-00007. [DOI] [PubMed] [Google Scholar]
- 27.National Institutes of Health . Guide for the Care and Use of Laboratory Animals (National Institutes of Health Publication 86-23) National Institutes of Health; Bethesda, MD: 1985. [Google Scholar]
- 28.McIntosh TK, Vink R, Noble L, et al. Traumatic brain injury in the rat: Characterization of a lateral fluid percussion model. Neuroscience. 1989;28:233–244. doi: 10.1016/0306-4522(89)90247-9. [DOI] [PubMed] [Google Scholar]
- 28a.Wang P, Ba ZF, Lu MC, et al. Measurement of circulating blood volume in vivo after trauma-hemorrhage and hemodilution. Am J Physiol. 1994;266(2 Pt 2):R368–R374. doi: 10.1152/ajpregu.1994.266.2.R368. [DOI] [PubMed] [Google Scholar]
- 29.Soares HD, Thomas M, Cloherty K, et al. Development of prolonged focal cerebral edema and regional cation change following experimental brain injury in the rat. J Neurochem. 1992;58:1845–1852. doi: 10.1111/j.1471-4159.1992.tb10061.x. [DOI] [PubMed] [Google Scholar]
- 30.Pierce JES, Smith DH, Trojanowski JQ, et al. Enduring cognitive, neurobehavioral, and histopathological changes persist for up to one year following severe experimental brain injury in rats. Neuroscience. 1998;87:359–369. doi: 10.1016/s0306-4522(98)00142-0. [DOI] [PubMed] [Google Scholar]
- 31.Smith DH, Okiyama K, Thomas MJ, et al. Evaluation of memory dysfunction following experimental brain injury using the Morris water maze. J Neurotrauma. 1991;8:259–269. doi: 10.1089/neu.1991.8.259. [DOI] [PubMed] [Google Scholar]
- 32.Paxinos G, Watson S. A Stereotactic Atlas of the Rat Brain. Academic Press; New York: 1990. [Google Scholar]
- 33.Zhang C, Raghupathi R, Saatman KE, et al. Riluzole attenuates cortical lesion size, but not hippocampal neuronal loss, following traumatic brain injury in the rat. J Neurosci Res. 1998;52:342–349. doi: 10.1002/(SICI)1097-4547(19980501)52:3<342::AID-JNR10>3.0.CO;2-8. [DOI] [PubMed] [Google Scholar]
- 34.Gennarelli TA, Champion HR, Sacco WJ, et al. Mortality of patients with head injury and extracranial injury treated in trauma centers. J Trauma. 1989;29:1193–1201. doi: 10.1097/00005373-198909000-00002. [DOI] [PubMed] [Google Scholar]
- 35.Bullock R, Chestnut RM, Clifton G, et al. Guidelines for the management of severe head injury. J Neurotrauma. 1996;14:639–734. [Google Scholar]
- 36.Morganti-Kossmann MC, Rancan M, Stahel PF, et al. Inflammatory response in acute traumatic brain injury: A double-edged sword. Curr Opin Crit Care. 2002;8:101–105. doi: 10.1097/00075198-200204000-00002. [DOI] [PubMed] [Google Scholar]
- 37.Engelborghs K, Haseldonckx M, van Reempts J, et al. Impaired autoregulation of cerebral blood flow in an experimental model of traumatic brain injury. J Neurotrauma. 2000;17:667–677. doi: 10.1089/089771500415418. [DOI] [PubMed] [Google Scholar]
- 38.Warner DS, Takoaka S, Wu B, et al. Electroencephalographic burst suppression is not required to elicit maximal neuroprotection from pentobarbital in a rat model of focal cerebral ischemia. Anesthesiology. 1996;84:1475–1484. doi: 10.1097/00000542-199606000-00024. [DOI] [PubMed] [Google Scholar]
- 39.Thompson HJ, Lifshitz J, Marklund N, et al. Lateral fluid percussion in the rat: A 15-year review and evaluation. J Neurotrauma. 2005;22:42–75. doi: 10.1089/neu.2005.22.42. [DOI] [PubMed] [Google Scholar]
- 40.Watford M. Net interorgan transport of L-glutamate in rats occurs via the plasma, not via erythrocytes. J Nutr. 2002;132:952–956. doi: 10.1093/jn/132.5.952. [DOI] [PubMed] [Google Scholar]
- 41.Hobbs AJ, Gladwin MT, Patel RP, et al. Haemoglobin: NO transporter, NO inactivator or NOne of the above? Trends Pharmacol Sci. 2002;23:406–411. doi: 10.1016/s0165-6147(02)02067-9. [DOI] [PubMed] [Google Scholar]
- 42.Clark RS, Kochanek PM, Dixon CE, et al. Early neuropathologic effects of mild or moderate hypoxemia after controlled cortical impact injury in rats. J Neurotrauma. 1997;14:179–189. doi: 10.1089/neu.1997.14.179. [DOI] [PubMed] [Google Scholar]
- 43.Wisner D, Busche F, Sturm J, et al. Traumatic shock and head injury: Effects of fluid resuscitation on the brain. J Surg Res. 1989;46:49–59. doi: 10.1016/0022-4804(89)90181-9. [DOI] [PubMed] [Google Scholar]
- 44.Eilig I, Rachinsky M, Artru AA, et al. The effect of treatment with albumin, hetastarch, or hypertonic saline on neurological status and brain edema in a rat model of closed head trauma combined with uncontrolled hemorrhage and concurrent resuscitation in rats. Anesth Analg. 2001;92:669–675. doi: 10.1097/00000539-200103000-00023. [DOI] [PubMed] [Google Scholar]
- 45.Save E, Poucet B. Hippocampal-parietal cortical interactions in spatial cognition. Hippocampus. 2000;10:491–499. doi: 10.1002/1098-1063(2000)10:4<491::AID-HIPO16>3.0.CO;2-0. [DOI] [PubMed] [Google Scholar]
- 46.Riedel G, Micheau J, Lam AG, et al. Reversible neural inactivation reveals hippocampal participation in several memory processes. Nat Neurosci. 1999;2:898–905. doi: 10.1038/13202. [DOI] [PubMed] [Google Scholar]
- 47.Skelton RW. Modelling recovery of cognitive function after traumatic brain injury: Spatial navigation in the Morris water maze after complete or partial transections of the perforant path in rats. Behav Br Res. 1998;96:13–35. doi: 10.1016/s0166-4328(97)00199-x. [DOI] [PubMed] [Google Scholar]
- 48.Hicks RR, Soares HD, Smith DH, et al. Temporal and spatial characterization of neuronal injury following lateral fluidpercussion brain injury in the rat. Acta Neuropathol. 1996;91:236–246. doi: 10.1007/s004010050421. [DOI] [PubMed] [Google Scholar]
- 49.Lyeth BG, Jenkins LW, Hamm RJ, et al. A fluid percussion model of experimental brain injury in the rat: Neurological, physiological, and histopathological characteristics. J Neurosurg. 1987;67:110–119. doi: 10.3171/jns.1987.67.1.0110. [DOI] [PubMed] [Google Scholar]
- 50.Dixon CE, Kochanek PM, Yan HQ, et al. One-year study of spatial memory performance, brain morphology and cholinergic markers after moderate controlled cortical impact in rats. J Neurotrauma. 1999;16:109–122. doi: 10.1089/neu.1999.16.109. [DOI] [PubMed] [Google Scholar]