Abstract
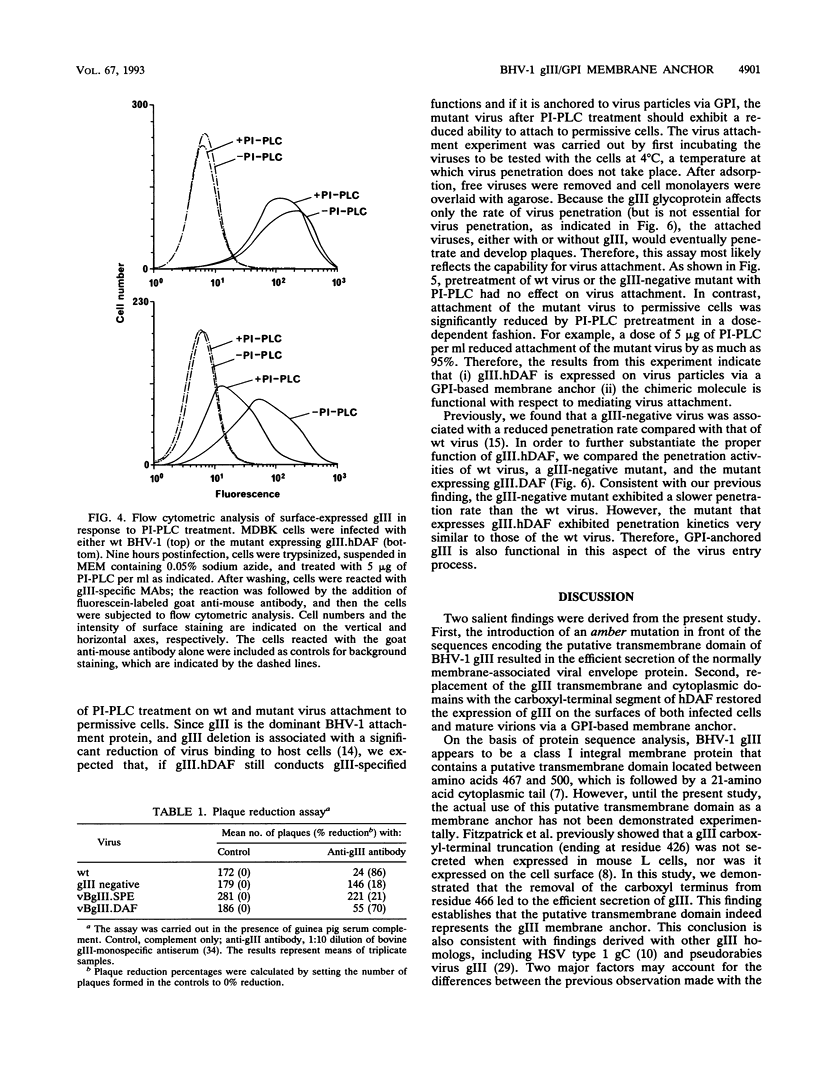
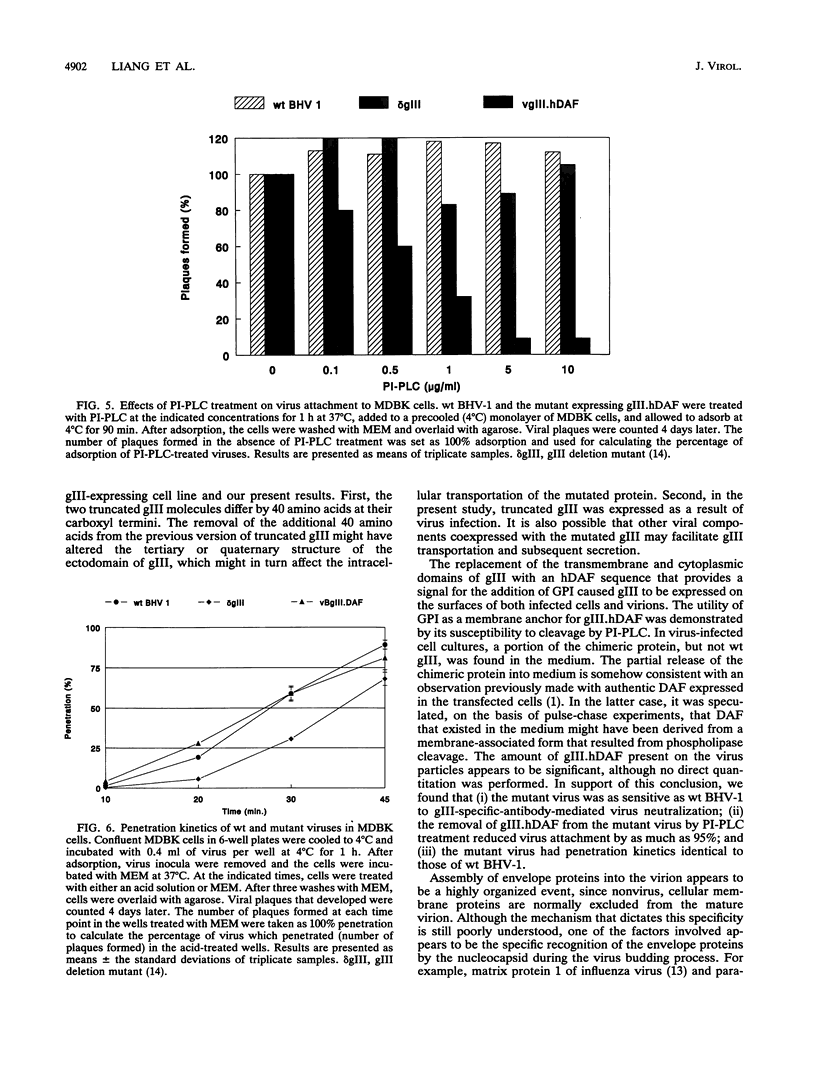
Mutants of bovine herpesvirus 1 that express a truncated envelope glycoprotein gIII or a gIII-human decay-accelerating factor (hDAF) chimeric protein (gIII.hDAF) were employed to evaluate the function of the transmembrane and cytoplasmic domains of the gIII molecule. Truncated gIII (i.e., lacking the transmembrane and cytoplasmic region) was readily released from infected cells and was not detected on mature virus particles. In contrast, replacement of the transmembrane and cytoplasmic domains with the carboxyl-terminal portion of hDAF restored the expression of gIII on the membranes of infected cells as well as on virion surfaces. The presence of the gIII.hDAF chimera on virus particles was also associated with normal gIII function, i.e., the mediation of virus attachment and penetration. The gIII-hDAF chimera, which is present on both infected cell surfaces and virions, could be cleaved by a phosphatidylinositol-specific phospholipase C, indicating that it was anchored in the membrane via glycosyl phosphatidylinositol. Our results from this study suggest that the transmembrane and cytoplasmic regions of the gIII molecule serve as a general membrane anchor, but they do not contain structural signals required for the specific assembly of envelope proteins into mature virions.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Caras I. W., Weddell G. N., Davitz M. A., Nussenzweig V., Martin D. W., Jr Signal for attachment of a phospholipid membrane anchor in decay accelerating factor. Science. 1987 Nov 27;238(4831):1280–1283. doi: 10.1126/science.2446389. [DOI] [PubMed] [Google Scholar]
- Caras I. W., Weddell G. N. Signal peptide for protein secretion directing glycophospholipid membrane anchor attachment. Science. 1989 Mar 3;243(4895):1196–1198. doi: 10.1126/science.2466338. [DOI] [PubMed] [Google Scholar]
- Chu G., Hayakawa H., Berg P. Electroporation for the efficient transfection of mammalian cells with DNA. Nucleic Acids Res. 1987 Feb 11;15(3):1311–1326. doi: 10.1093/nar/15.3.1311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cross G. A. Eukaryotic protein modification and membrane attachment via phosphatidylinositol. Cell. 1987 Jan 30;48(2):179–181. doi: 10.1016/0092-8674(87)90419-3. [DOI] [PubMed] [Google Scholar]
- Deeg M. A., Humphrey D. R., Yang S. H., Ferguson T. R., Reinhold V. N., Rosenberry T. L. Glycan components in the glycoinositol phospholipid anchor of human erythrocyte acetylcholinesterase. Novel fragments produced by trifluoroacetic acid. J Biol Chem. 1992 Sep 15;267(26):18573–18580. [PubMed] [Google Scholar]
- Fitzpatrick D. R., Babiuk L. A., Zamb T. J. Nucleotide sequence of bovine herpesvirus type 1 glycoprotein gIII, a structural model for gIII as a new member of the immunoglobulin superfamily, and implications for the homologous glycoproteins of other herpesviruses. Virology. 1989 Nov;173(1):46–57. doi: 10.1016/0042-6822(89)90220-1. [DOI] [PubMed] [Google Scholar]
- Fitzpatrick D. R., Redmond M. J., Attah-Poku S. K., van Drunen Littel-van den Hurk S., Babiuk L. A., Zamb T. J. Mapping of 10 epitopes on bovine herpesvirus type 1 glycoproteins gI and gIII. Virology. 1990 May;176(1):145–157. doi: 10.1016/0042-6822(90)90239-n. [DOI] [PubMed] [Google Scholar]
- Fitzpatrick D. R., Zamb T. J., Babiuk L. A. Expression of bovine herpesvirus type 1 glycoprotein gI in transfected bovine cells induces spontaneous cell fusion. J Gen Virol. 1990 May;71(Pt 5):1215–1219. doi: 10.1099/0022-1317-71-5-1215. [DOI] [PubMed] [Google Scholar]
- Holland T. C., Homa F. L., Marlin S. D., Levine M., Glorioso J. Herpes simplex virus type 1 glycoprotein C-negative mutants exhibit multiple phenotypes, including secretion of truncated glycoproteins. J Virol. 1984 Nov;52(2):566–574. doi: 10.1128/jvi.52.2.566-574.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hughes G., Babiuk L. A., van Drunen Littel-van den Hurk S. Functional and topographical analyses of epitopes on bovine herpesvirus type 1 glycoprotein IV. Arch Virol. 1988;103(1-2):47–60. doi: 10.1007/BF01319808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang X. P., Babiuk L. A., Zamb T. J. Pseudorabies virus gIII and bovine herpesvirus 1 gIII share complementary functions. J Virol. 1991 Oct;65(10):5553–5557. doi: 10.1128/jvi.65.10.5553-5557.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang X. P., Babiuk L. A., van Drunen Littel-van den Hurk S., Fitzpatrick D. R., Zamb T. J. Bovine herpesvirus 1 attachment to permissive cells is mediated by its major glycoproteins gI, gIII, and gIV. J Virol. 1991 Mar;65(3):1124–1132. doi: 10.1128/jvi.65.3.1124-1132.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang X., Babiuk L. A., Zamb T. J. An in vivo study of a glycoprotein gIII-negative bovine herpesvirus 1 (BHV-1) mutant expressing beta-galactosidase: evaluation of the role of gIII in virus infectivity and its use as a vector for mucosal immunization. Virology. 1992 Aug;189(2):629–639. doi: 10.1016/0042-6822(92)90586-e. [DOI] [PubMed] [Google Scholar]
- Liang X., Babiuk L. A., Zamb T. J. Mapping of heparin-binding structures on bovine herpesvirus 1 and pseudorabies virus gIII glycoproteins. Virology. 1993 May;194(1):233–243. doi: 10.1006/viro.1993.1254. [DOI] [PubMed] [Google Scholar]
- Lublin D. M., Krsek-Staples J., Pangburn M. K., Atkinson J. P. Biosynthesis and glycosylation of the human complement regulatory protein decay-accelerating factor. J Immunol. 1986 Sep 1;137(5):1629–1635. [PubMed] [Google Scholar]
- Magee A. I. Lipid modification of proteins and its relevance to protein targeting. J Cell Sci. 1990 Dec;97(Pt 4):581–584. doi: 10.1242/jcs.97.4.581. [DOI] [PubMed] [Google Scholar]
- Marshall R. L., Rodriguez L. L., Letchworth G. J., 3rd Characterization of envelope proteins of infectious bovine rhinotracheitis virus (bovine herpesvirus 1) by biochemical and immunological methods. J Virol. 1986 Mar;57(3):745–753. doi: 10.1128/jvi.57.3.745-753.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayfield J. E., Good P. J., VanOort H. J., Campbell A. R., Reed D. E. Cloning and cleavage site mapping of DNA from bovine herpesvirus 1 (Cooper strain). J Virol. 1983 Jul;47(1):259–264. doi: 10.1128/jvi.47.1.259-264.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medof M. E., Lublin D. M., Holers V. M., Ayers D. J., Getty R. R., Leykam J. F., Atkinson J. P., Tykocinski M. L. Cloning and characterization of cDNAs encoding the complete sequence of decay-accelerating factor of human complement. Proc Natl Acad Sci U S A. 1987 Apr;84(7):2007–2011. doi: 10.1073/pnas.84.7.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medof M. E., Walter E. I., Roberts W. L., Haas R., Rosenberry T. L. Decay accelerating factor of complement is anchored to cells by a C-terminal glycolipid. Biochemistry. 1986 Nov 4;25(22):6740–6747. doi: 10.1021/bi00370a003. [DOI] [PubMed] [Google Scholar]
- Misra V., Blumenthal R. M., Babiuk L. A. Proteins Specified by bovine herpesvirus 1 (infectious bovine rhinotracheitis virus). J Virol. 1981 Nov;40(2):367–378. doi: 10.1128/jvi.40.2.367-378.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okazaki K., Honda E., Minetoma T., Kumagai T. Bovine herpesvirus type 1 gp87 mediates both attachment of virions to susceptible cells and hemagglutination. Arch Virol. 1987;97(3-4):297–307. doi: 10.1007/BF01314428. [DOI] [PubMed] [Google Scholar]
- Okazaki K., Matsuzaki T., Sugahara Y., Okada J., Hasebe M., Iwamura Y., Ohnishi M., Kanno T., Shimizu M., Honda E. BHV-1 adsorption is mediated by the interaction of glycoprotein gIII with heparinlike moiety on the cell surface. Virology. 1991 Apr;181(2):666–670. doi: 10.1016/0042-6822(91)90900-v. [DOI] [PubMed] [Google Scholar]
- Schreurs C., Mettenleiter T. C., Zuckermann F., Sugg N., Ben-Porat T. Glycoprotein gIII of pseudorabies virus is multifunctional. J Virol. 1988 Jul;62(7):2251–2257. doi: 10.1128/jvi.62.7.2251-2257.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solomon K. A., Robbins A. K., Enquist L. W. Mutations in the C-terminal hydrophobic domain of pseudorabies virus gIII affect both membrane anchoring and protein export. J Virol. 1991 Nov;65(11):5952–5960. doi: 10.1128/jvi.65.11.5952-5960.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tikoo S. K., Fitzpatrick D. R., Babiuk L. A., Zamb T. J. Molecular cloning, sequencing, and expression of functional bovine herpesvirus 1 glycoprotein gIV in transfected bovine cells. J Virol. 1990 Oct;64(10):5132–5142. doi: 10.1128/jvi.64.10.5132-5142.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Drunen Littel-van den Hurk S., Babiuk L. A. Antigenic and immunogenic characteristics of bovine herpesvirus type-1 glycoproteins GVP 3/9 and GVP 6/11a/16, purified by immunoadsorbent chromatography. Virology. 1985 Jul 15;144(1):204–215. doi: 10.1016/0042-6822(85)90318-6. [DOI] [PubMed] [Google Scholar]
- van Drunen Littel-van den Hurk S., Babiuk L. A. Synthesis and processing of bovine herpesvirus 1 glycoproteins. J Virol. 1986 Aug;59(2):401–410. doi: 10.1128/jvi.59.2.401-410.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Drunen Littel-van den Hurk S., Zamb T., Babiuk L. A. Synthesis, cellular location, and immunogenicity of bovine herpesvirus 1 glycoproteins gI and gIII expressed by recombinant vaccinia virus. J Virol. 1989 May;63(5):2159–2168. doi: 10.1128/jvi.63.5.2159-2168.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Drunen Littel-van den Hurk S., van den Hurk J. V., Gilchrist J. E., Misra V., Babiuk L. A. Interactions of monoclonal antibodies and bovine herpesvirus type 1 (BHV-1) glycoproteins: characterization of their biochemical and immunological properties. Virology. 1984 Jun;135(2):466–479. doi: 10.1016/0042-6822(84)90201-0. [DOI] [PubMed] [Google Scholar]