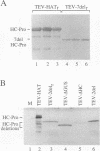
Abstract
The RNA genome of tobacco etch potyvirus (TEV) was engineered to express bacterial beta-glucuronidase (GUS) fused to the virus helper component proteinase (HC-Pro). It was shown previously that prolonged periods (approximately 1 month) of TEV-GUS propagation in plants resulted in the appearance of spontaneous deletion variants. Nine deletion mutants were identified by nucleotide sequence analysis of 40 cDNA clones obtained after polymerase chain reaction amplification. The mutants were missing between 1,741 and 2,074 nucleotides from TEV-GUS, including the sequences coding for most of GUS and the N-terminal region of HC-Pro. This region of HC-Pro contains determinants involved in helper component activity during aphid transmission, as well as a highly conserved series of cysteine residues. The deletion variants were shown to replicate and move systemically without the aid of a helper virus. Infectious viruses harboring the two largest HC-Pro deletions (termed TEV-2del and TEV-7del) were reconstructed by subcloning the corresponding mutated regions into full-length DNA copies of the TEV genome. Characterization of these and additional variants derived by site-directed mutagenesis demonstrated that deletion of sequences coding for the HC-Pro N-terminal domain had a negative effect on accumulation of viral RNA and coat protein. The TEV-2del variant possessed an aphid-nontransmissible phenotype that could be rescued partially by prefeeding of aphids on active HC-Pro from another potyvirus. These data suggest that the N-terminal domain of HC-Pro or its coding sequence enhances virus replication or genome expression but does not provide an activity essential for these processes. The function of this domain, as well as a proposed deletion mechanism involving nonhomologous recombination, is discussed.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Atreya C. D., Atreya P. L., Thornbury D. W., Pirone T. P. Site-directed mutations in the potyvirus HC-Pro gene affect helper component activity, virus accumulation, and symptom expression in infected tobacco plants. Virology. 1992 Nov;191(1):106–111. doi: 10.1016/0042-6822(92)90171-k. [DOI] [PubMed] [Google Scholar]
- Atreya P. L., Atreya C. D., Pirone T. P. Amino acid substitutions in the coat protein result in loss of insect transmissibility of a plant virus. Proc Natl Acad Sci U S A. 1991 Sep 1;88(17):7887–7891. doi: 10.1073/pnas.88.17.7887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banner L. R., Lai M. M. Random nature of coronavirus RNA recombination in the absence of selection pressure. Virology. 1991 Nov;185(1):441–445. doi: 10.1016/0042-6822(91)90795-D. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berger P. H., Hunt A. G., Domier L. L., Hellmann G. M., Stram Y., Thornbury D. W., Pirone T. P. Expression in transgenic plants of a viral gene product that mediates insect transmission of potyviruses. Proc Natl Acad Sci U S A. 1989 Nov;86(21):8402–8406. doi: 10.1073/pnas.86.21.8402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Cary S. M., Parks T. D., Dougherty W. G. A second proteinase encoded by a plant potyvirus genome. EMBO J. 1989 Feb;8(2):365–370. doi: 10.1002/j.1460-2075.1989.tb03386.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Dougherty W. G. Small nuclear inclusion protein encoded by a plant potyvirus genome is a protease. J Virol. 1987 Aug;61(8):2540–2548. doi: 10.1128/jvi.61.8.2540-2548.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Freed D. D. Cap-independent enhancement of translation by a plant potyvirus 5' nontranslated region. J Virol. 1990 Apr;64(4):1590–1597. doi: 10.1128/jvi.64.4.1590-1597.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Freed D. D., Sanders T. C. Autocatalytic processing of the potyvirus helper component proteinase in Escherichia coli and in vitro. J Virol. 1989 Oct;63(10):4459–4463. doi: 10.1128/jvi.63.10.4459-4463.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapman S., Kavanagh T., Baulcombe D. Potato virus X as a vector for gene expression in plants. Plant J. 1992 Jul;2(4):549–557. doi: 10.1046/j.1365-313x.1992.t01-24-00999.x. [DOI] [PubMed] [Google Scholar]
- Choi G. H., Pawlyk D. M., Nuss D. L. The autocatalytic protease p29 encoded by a hypovirulence-associated virus of the chestnut blight fungus resembles the potyvirus-encoded protease HC-Pro. Virology. 1991 Aug;183(2):747–752. doi: 10.1016/0042-6822(91)91004-z. [DOI] [PubMed] [Google Scholar]
- Davidson A. D., Pröls M., Schell J., Steinbiss H. H. The nucleotide sequence of RNA 2 of barley yellow mosaic virus. J Gen Virol. 1991 Apr;72(Pt 4):989–993. doi: 10.1099/0022-1317-72-4-989. [DOI] [PubMed] [Google Scholar]
- Dolja V. V., McBride H. J., Carrington J. C. Tagging of plant potyvirus replication and movement by insertion of beta-glucuronidase into the viral polyprotein. Proc Natl Acad Sci U S A. 1992 Nov 1;89(21):10208–10212. doi: 10.1073/pnas.89.21.10208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donson J., Kearney C. M., Hilf M. E., Dawson W. O. Systemic expression of a bacterial gene by a tobacco mosaic virus-based vector. Proc Natl Acad Sci U S A. 1991 Aug 15;88(16):7204–7208. doi: 10.1073/pnas.88.16.7204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guilford P. J., Ziegler-Graff V., Baulcombe D. C. Mutation and replacement of the 16-kDa protein gene in RNA-1 of tobacco rattle virus. Virology. 1991 Jun;182(2):607–614. doi: 10.1016/0042-6822(91)90601-7. [DOI] [PubMed] [Google Scholar]
- Jarvis T. C., Kirkegaard K. The polymerase in its labyrinth: mechanisms and implications of RNA recombination. Trends Genet. 1991 Jun;7(6):186–191. doi: 10.1016/0168-9525(91)90434-R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kashiwazaki S., Minobe Y., Hibino H. Nucleotide sequence of barley yellow mosaic virus RNA 2. J Gen Virol. 1991 Apr;72(Pt 4):995–999. doi: 10.1099/0022-1317-72-4-995. [DOI] [PubMed] [Google Scholar]
- Kirkegaard K., Baltimore D. The mechanism of RNA recombination in poliovirus. Cell. 1986 Nov 7;47(3):433–443. doi: 10.1016/0092-8674(86)90600-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koonin E. V., Boyko V. P., Dolja V. V. Small cysteine-rich proteins of different groups of plant RNA viruses are related to different families of nucleic acid-binding proteins. Virology. 1991 Mar;181(1):395–398. doi: 10.1016/0042-6822(91)90512-a. [DOI] [PubMed] [Google Scholar]
- Koonin E. V., Choi G. H., Nuss D. L., Shapira R., Carrington J. C. Evidence for common ancestry of a chestnut blight hypovirulence-associated double-stranded RNA and a group of positive-strand RNA plant viruses. Proc Natl Acad Sci U S A. 1991 Dec 1;88(23):10647–10651. doi: 10.1073/pnas.88.23.10647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kunkel T. A., Roberts J. D., Zakour R. A. Rapid and efficient site-specific mutagenesis without phenotypic selection. Methods Enzymol. 1987;154:367–382. doi: 10.1016/0076-6879(87)54085-x. [DOI] [PubMed] [Google Scholar]
- Lai M. M. RNA recombination in animal and plant viruses. Microbiol Rev. 1992 Mar;56(1):61–79. doi: 10.1128/mr.56.1.61-79.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mavankal G., Rhoads R. E. In vitro cleavage at or near the N-terminus of the helper component protein in the tobacco vein mottling virus polyprotein. Virology. 1991 Dec;185(2):721–731. doi: 10.1016/0042-6822(91)90543-k. [DOI] [PubMed] [Google Scholar]
- Meyers G., Tautz N., Dubovi E. J., Thiel H. J. Viral cytopathogenicity correlated with integration of ubiquitin-coding sequences. Virology. 1991 Feb;180(2):602–616. doi: 10.1016/0042-6822(91)90074-L. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyers G., Tautz N., Stark R., Brownlie J., Dubovi E. J., Collett M. S., Thiel H. J. Rearrangement of viral sequences in cytopathogenic pestiviruses. Virology. 1992 Nov;191(1):368–386. doi: 10.1016/0042-6822(92)90199-Y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niesbach-Klösgen U., Guilley H., Jonard G., Richards K. Immunodetection in vivo of beet necrotic yellow vein virus-encoded proteins. Virology. 1990 Sep;178(1):52–61. doi: 10.1016/0042-6822(90)90378-5. [DOI] [PubMed] [Google Scholar]
- Nuss D. L. Biological control of chestnut blight: an example of virus-mediated attenuation of fungal pathogenesis. Microbiol Rev. 1992 Dec;56(4):561–576. doi: 10.1128/mr.56.4.561-576.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oh C. S., Carrington J. C. Identification of essential residues in potyvirus proteinase HC-Pro by site-directed mutagenesis. Virology. 1989 Dec;173(2):692–699. doi: 10.1016/0042-6822(89)90582-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petty I. T., French R., Jones R. W., Jackson A. O. Identification of barley stripe mosaic virus genes involved in viral RNA replication and systemic movement. EMBO J. 1990 Nov;9(11):3453–3457. doi: 10.1002/j.1460-2075.1990.tb07553.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Restrepo M. A., Freed D. D., Carrington J. C. Nuclear transport of plant potyviral proteins. Plant Cell. 1990 Oct;2(10):987–998. doi: 10.1105/tpc.2.10.987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riechmann J. L., Laín S., García J. A. Highlights and prospects of potyvirus molecular biology. J Gen Virol. 1992 Jan;73(Pt 1):1–16. doi: 10.1099/0022-1317-73-1-1. [DOI] [PubMed] [Google Scholar]
- Thornbury D. W., Patterson C. A., Dessens J. T., Pirone T. P. Comparative sequence of the helper component (HC) region of potato virus Y and a HC-defective strain, potato virus C. Virology. 1990 Oct;178(2):573–578. doi: 10.1016/0042-6822(90)90356-v. [DOI] [PubMed] [Google Scholar]
- Verchot J., Herndon K. L., Carrington J. C. Mutational analysis of the tobacco etch potyviral 35-kDa proteinase: identification of essential residues and requirements for autoproteolysis. Virology. 1992 Sep;190(1):298–306. doi: 10.1016/0042-6822(92)91216-h. [DOI] [PubMed] [Google Scholar]
- Verchot J., Koonin E. V., Carrington J. C. The 35-kDa protein from the N-terminus of the potyviral polyprotein functions as a third virus-encoded proteinase. Virology. 1991 Dec;185(2):527–535. doi: 10.1016/0042-6822(91)90522-d. [DOI] [PubMed] [Google Scholar]