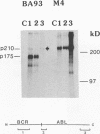
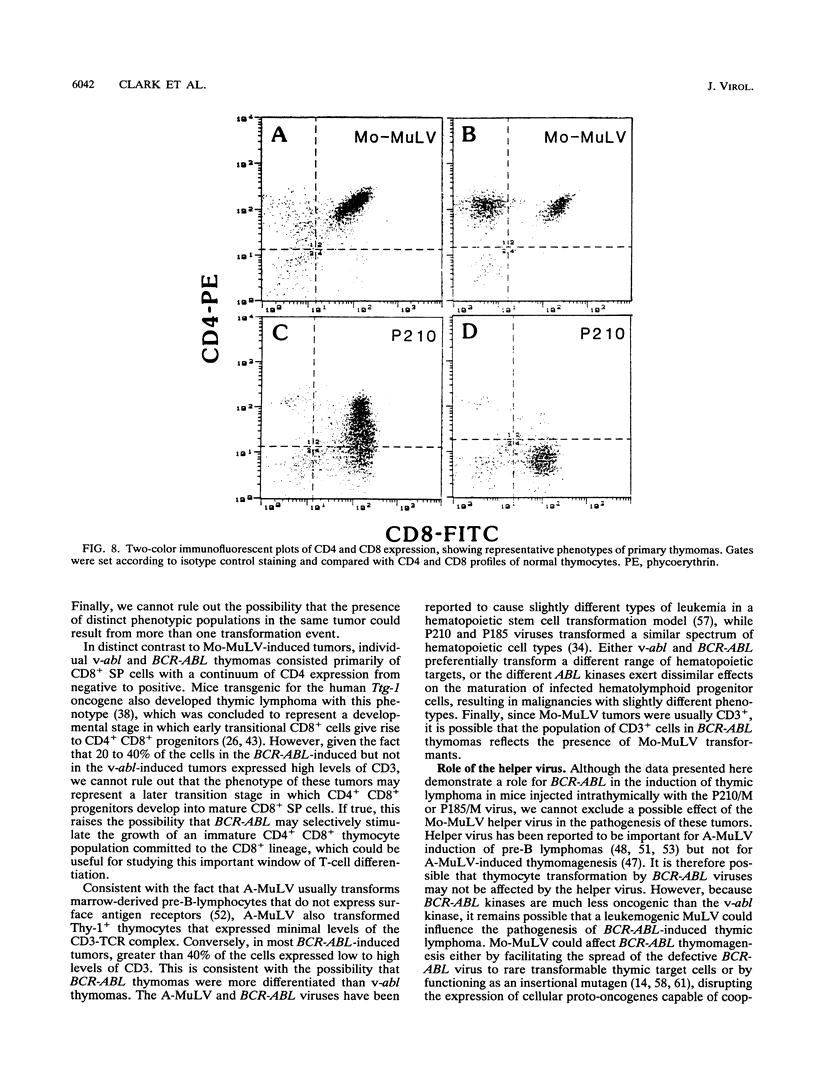
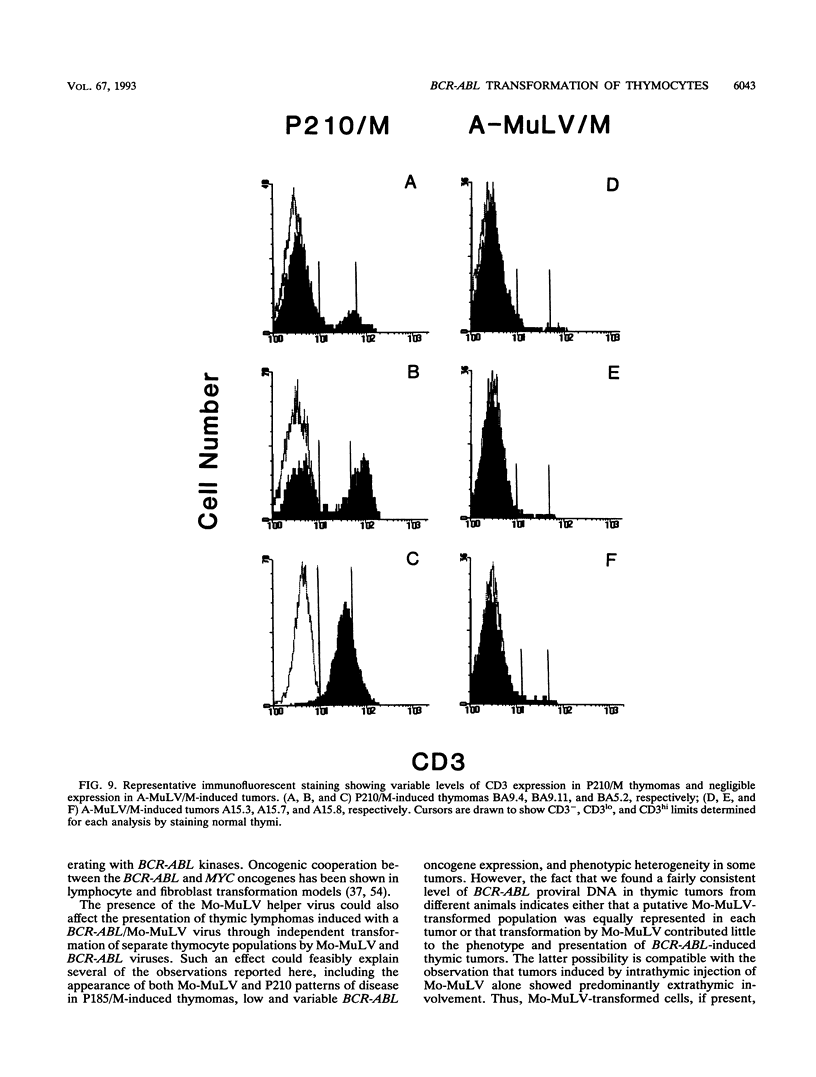
Abstract
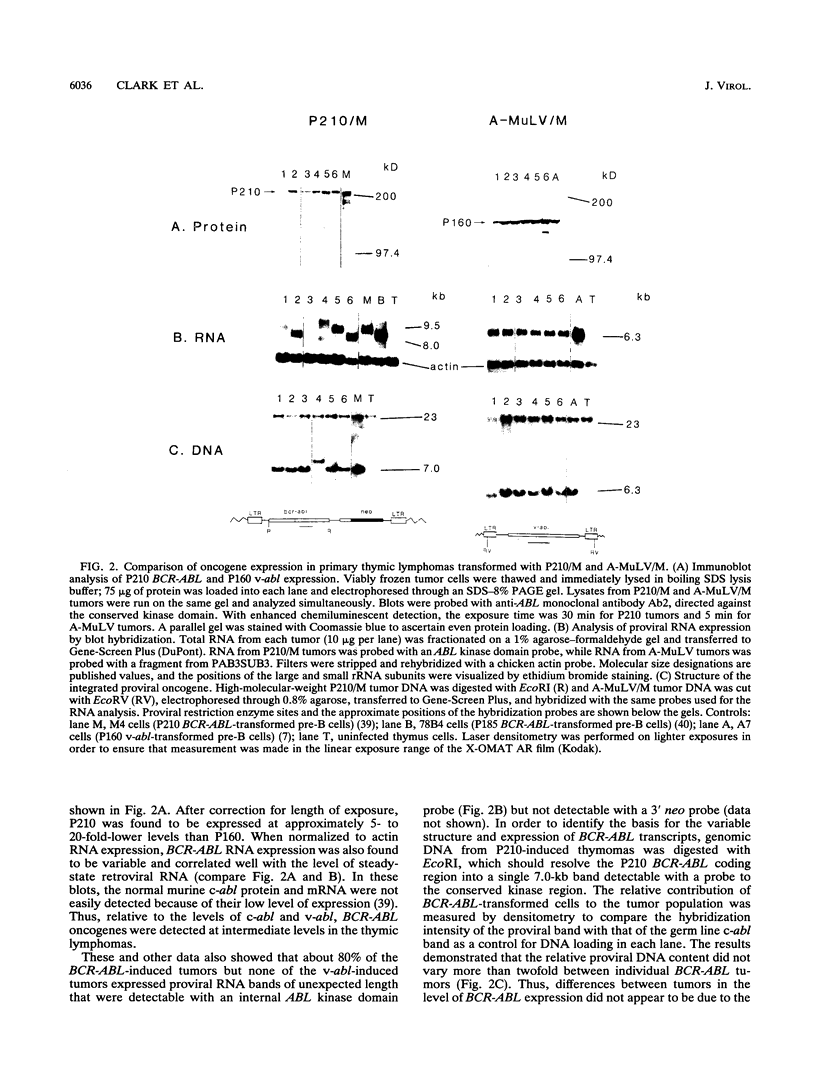
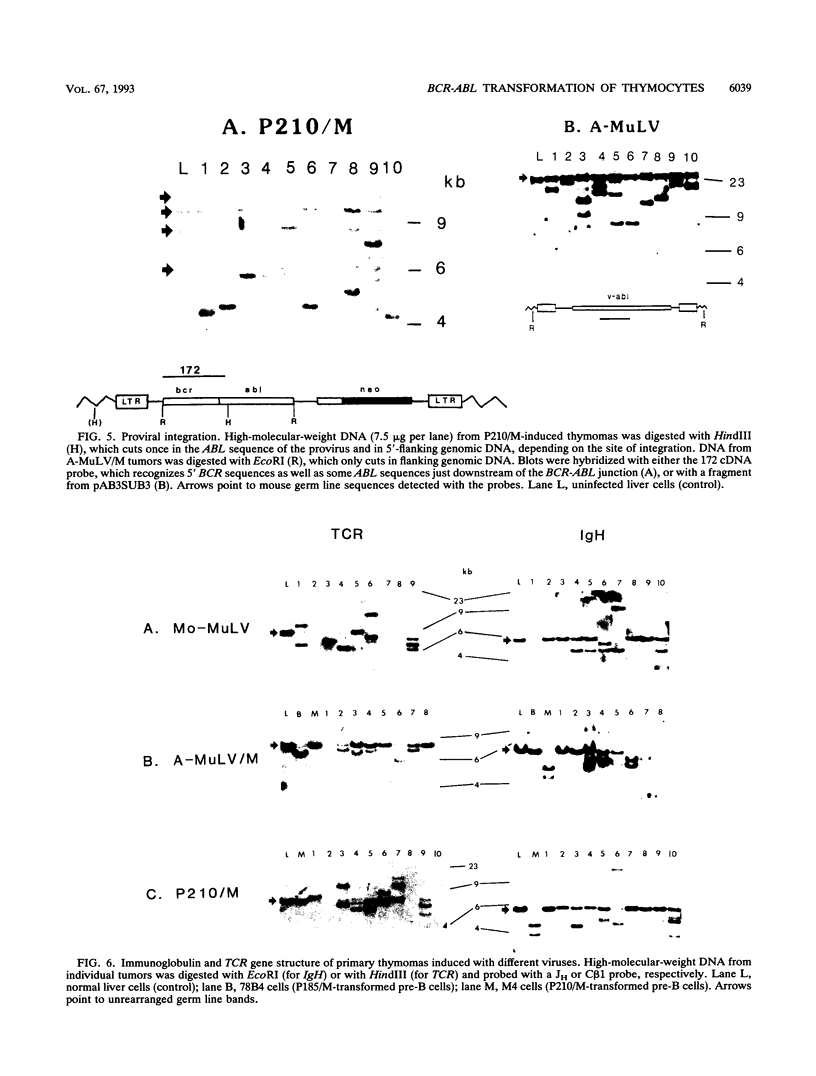
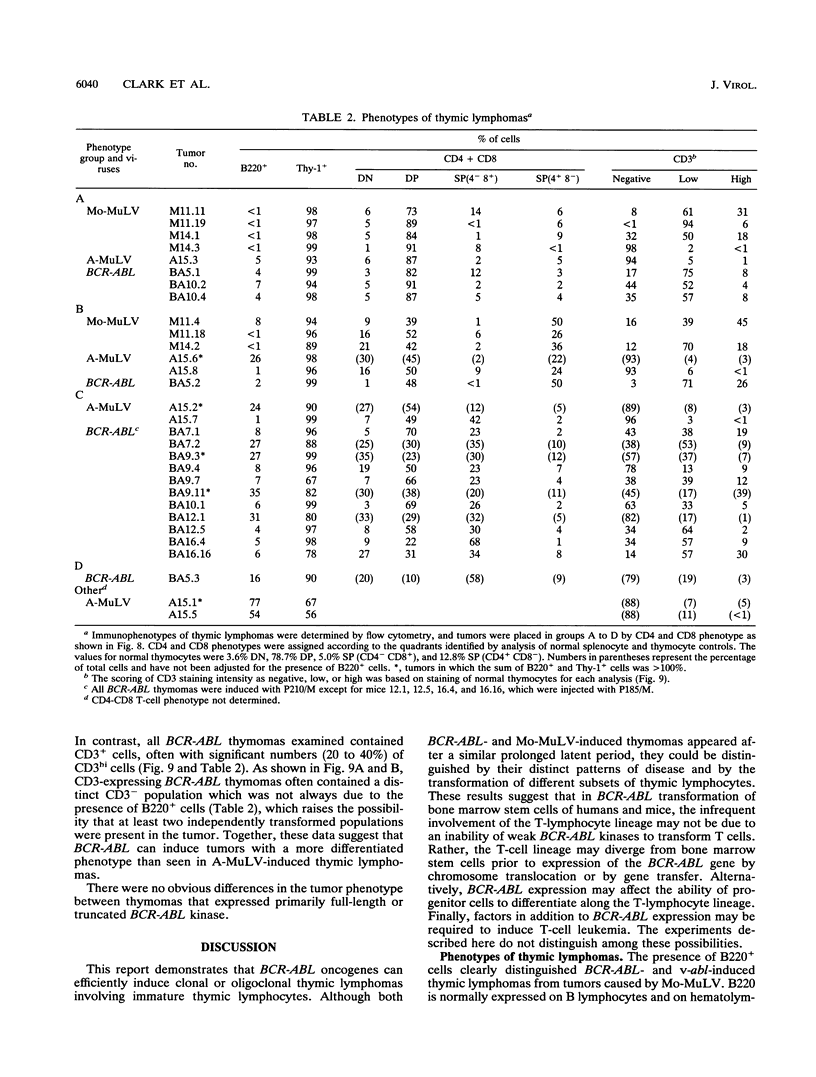
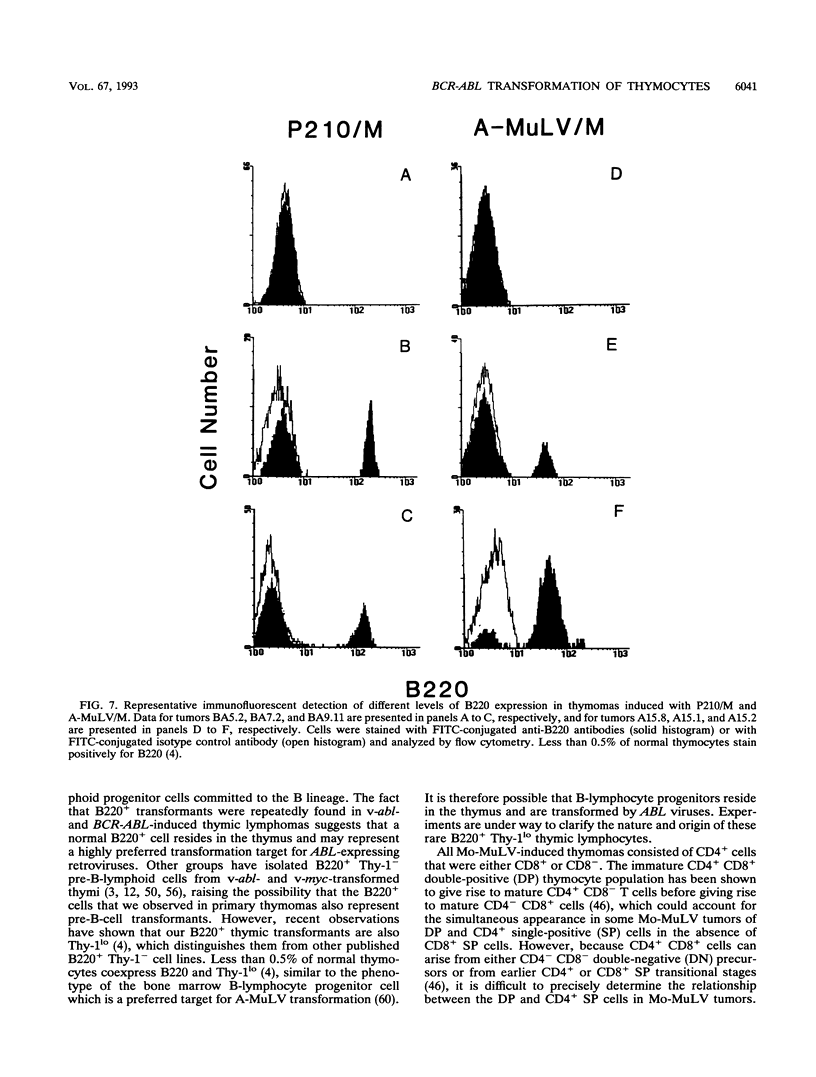
The human BCR-ABL oncogenes encoded by the Philadelphia chromosome (Ph) affect the pathogenesis of diverse types of leukemia and yet are rarely associated with T-lymphoid leukemia. To determine whether BCR-ABL kinases are inefficient in transforming T lymphocytes, BCR-ABL-expressing retroviruses were injected intrathymically into mice. Thymomas that expressed BCR-ABL kinase developed after a relatively long latent period. In most thymomas, deletion of 3' proviral sequences resulted in loss of tk-neo and occasionally caused expression of kinase-active carboxy-terminally truncated BCR-ABL oncoprotein. In contrast, deletion of 3' proviral sequences was not observed in thymomas induced with Abelson murine leukemia virus (A-MuLV). BCR-ABL viruses induced distinct patterns of disease and involved different thymocyte subsets than A-MuLV and Moloney murine leukemia virus (Mo-MuLV). While Mo-MuLV only induced Thy-1+ thymomas, v-abl- and BCR-ABL-induced thymomas often contained mixed populations of B220+ and Thy-1+ lymphocytes in the same tumor. In most v-abl and BCR-ABL tumors, Thy-1+ lymphoid cells expressed CD8 and a continuum of CD4 ranging from negative to positive. Conversely, Mo-MuLV thymomas contained distinct populations of CD4+ cells that were either CD8+ or CD8-. A-MuLV-transformed T-lymphoid cells did not express the CD3/T-cell receptor complex, while BCR-ABL tumors were CD3+. Thus, BCR-ABL viruses preferentially induce somewhat more differentiated T lymphocytes than are transformed by A-MuLV. Furthermore, rare B220+ lymphocytes may represent preferred v-abl and BCR-ABL transformation targets in the thymus.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andrews D. F., 3rd, Collins S. J. Heterogeneity in expression of the bcr-abl fusion transcript in CML blast crisis. Leukemia. 1987 Oct;1(10):718–724. [PubMed] [Google Scholar]
- Born W., Yagüe J., Palmer E., Kappler J., Marrack P. Rearrangement of T-cell receptor beta-chain genes during T-cell development. Proc Natl Acad Sci U S A. 1985 May;82(9):2925–2929. doi: 10.1073/pnas.82.9.2925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brightman B. K., Chandy K. G., Spencer R. H., Gupta S., Pattengale P. K., Fan H. Characterization of lymphoid tumors induced by a recombinant murine retrovirus carrying the avian v-myc oncogene. Identification of novel (B-lymphoid) tumors in the thymus. J Immunol. 1988 Oct 15;141(8):2844–2854. [PubMed] [Google Scholar]
- Clark S. S., Crist W. M., Witte O. N. Molecular pathogenesis of Ph-positive leukemias. Annu Rev Med. 1989;40:113–122. doi: 10.1146/annurev.me.40.020189.000553. [DOI] [PubMed] [Google Scholar]
- Clark S. S., McLaughlin J., Crist W. M., Champlin R., Witte O. N. Unique forms of the abl tyrosine kinase distinguish Ph1-positive CML from Ph1-positive ALL. Science. 1987 Jan 2;235(4784):85–88. doi: 10.1126/science.3541203. [DOI] [PubMed] [Google Scholar]
- Clark S. S., McLaughlin J., Timmons M., Pendergast A. M., Ben-Neriah Y., Dow L. W., Crist W., Rovera G., Smith S. D., Witte O. N. Expression of a distinctive BCR-ABL oncogene in Ph1-positive acute lymphocytic leukemia (ALL). Science. 1988 Feb 12;239(4841 Pt 1):775–777. doi: 10.1126/science.3422516. [DOI] [PubMed] [Google Scholar]
- Cook W. D., Balaton A. M. T-cell receptor and immunoglobulin genes are rearranged together in Abelson virus-transformed pre-B and pre-T cells. Mol Cell Biol. 1987 Jan;7(1):266–272. doi: 10.1128/mcb.7.1.266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook W. D., Metcalf D., Nicola N. A., Burgess A. W., Walker F. Malignant transformation of a growth factor-dependent myeloid cell line by Abelson virus without evidence of an autocrine mechanism. Cell. 1985 Jul;41(3):677–683. doi: 10.1016/s0092-8674(85)80048-9. [DOI] [PubMed] [Google Scholar]
- Cook W. D. Rapid thymomas induced by Abelson murine leukemia virus. Proc Natl Acad Sci U S A. 1982 May;79(9):2917–2921. doi: 10.1073/pnas.79.9.2917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook W. D. Thymocyte subsets transformed by Abelson murine leukemia virus. Mol Cell Biol. 1985 Feb;5(2):390–397. doi: 10.1128/mcb.5.2.390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corcoran L. M., Adams J. M., Dunn A. R., Cory S. Murine T lymphomas in which the cellular myc oncogene has been activated by retroviral insertion. Cell. 1984 May;37(1):113–122. doi: 10.1016/0092-8674(84)90306-4. [DOI] [PubMed] [Google Scholar]
- Daley G. Q., McLaughlin J., Witte O. N., Baltimore D. The CML-specific P210 bcr/abl protein, unlike v-abl, does not transform NIH/3T3 fibroblasts. Science. 1987 Jul 31;237(4814):532–535. doi: 10.1126/science.2440107. [DOI] [PubMed] [Google Scholar]
- Daley G. Q., Van Etten R. A., Baltimore D. Induction of chronic myelogenous leukemia in mice by the P210bcr/abl gene of the Philadelphia chromosome. Science. 1990 Feb 16;247(4944):824–830. doi: 10.1126/science.2406902. [DOI] [PubMed] [Google Scholar]
- Denis K. A., Treiman L. J., St Claire J. I., Witte O. N. Long-term cultures of murine fetal liver retain very early B lymphoid phenotype. J Exp Med. 1984 Oct 1;160(4):1087–1101. doi: 10.1084/jem.160.4.1087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dreazen O., Canaani E., Gale R. P. Molecular biology of chronic myelogenous leukemia. Semin Hematol. 1988 Jan;25(1):35–48. [PubMed] [Google Scholar]
- Elefanty A. G., Cory S. Hematologic disease induced in BALB/c mice by a bcr-abl retrovirus is influenced by the infection conditions. Mol Cell Biol. 1992 Apr;12(4):1755–1763. doi: 10.1128/mcb.12.4.1755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elefanty A. G., Hariharan I. K., Cory S. bcr-abl, the hallmark of chronic myeloid leukaemia in man, induces multiple haemopoietic neoplasms in mice. EMBO J. 1990 Apr;9(4):1069–1078. doi: 10.1002/j.1460-2075.1990.tb08212.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forster A., Huck S., Ghanem N., Lefranc M. P., Rabbitts T. H. New subgroups in the human T cell rearranging V gamma gene locus. EMBO J. 1987 Jul;6(7):1945–1950. doi: 10.1002/j.1460-2075.1987.tb02456.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gishizky M. L., McLaughlin J., Pendergast A. M., Witte O. N. The 5' non-coding region of the BCR/ABL oncogene augments its ability to stimulate the growth of immature lymphoid cells. Oncogene. 1991 Aug;6(8):1299–1306. [PubMed] [Google Scholar]
- Gishizky M. L., Witte O. N. Initiation of deregulated growth of multipotent progenitor cells by bcr-abl in vitro. Science. 1992 May 8;256(5058):836–839. doi: 10.1126/science.1375394. [DOI] [PubMed] [Google Scholar]
- Goff S. P., Tabin C. J., Wang J. Y., Weinberg R., Baltimore D. Transfection of fibroblasts by cloned Abelson murine leukemia virus DNA and recovery of transmissible virus by recombination with helper virus. J Virol. 1982 Jan;41(1):271–285. doi: 10.1128/jvi.41.1.271-285.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guidos C. J., Weissman I. L., Adkins B. Intrathymic maturation of murine T lymphocytes from CD8+ precursors. Proc Natl Acad Sci U S A. 1989 Oct;86(19):7542–7546. doi: 10.1073/pnas.86.19.7542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hariharan I. K., Harris A. W., Crawford M., Abud H., Webb E., Cory S., Adams J. M. A bcr-v-abl oncogene induces lymphomas in transgenic mice. Mol Cell Biol. 1989 Jul;9(7):2798–2805. doi: 10.1128/mcb.9.7.2798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hedrick S. M., Nielsen E. A., Kavaler J., Cohen D. I., Davis M. M. Sequence relationships between putative T-cell receptor polypeptides and immunoglobulins. Nature. 1984 Mar 8;308(5955):153–158. doi: 10.1038/308153a0. [DOI] [PubMed] [Google Scholar]
- Heisterkamp N., Jenster G., ten Hoeve J., Zovich D., Pattengale P. K., Groffen J. Acute leukaemia in bcr/abl transgenic mice. Nature. 1990 Mar 15;344(6263):251–253. doi: 10.1038/344251a0. [DOI] [PubMed] [Google Scholar]
- Henderson A. J., Dorshkind K. In vitro models of B lymphocyte development. Semin Immunol. 1990 May;2(3):181–187. [PubMed] [Google Scholar]
- Holland G. D., Ito K., Kaehler D. A., Tonegawa S., Risser R. Thymic targets for Abelson murine leukemia virus are early gamma/delta T lymphocytes. Proc Natl Acad Sci U S A. 1991 May 1;88(9):3700–3704. doi: 10.1073/pnas.88.9.3700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito K., Bonneville M., Takagaki Y., Nakanishi N., Kanagawa O., Krecko E. G., Tonegawa S. Different gamma delta T-cell receptors are expressed on thymocytes at different stages of development. Proc Natl Acad Sci U S A. 1989 Jan;86(2):631–635. doi: 10.1073/pnas.86.2.631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jonas D., Lübbert M., Kawasaki E. S., Henke M., Bross K. J., Mertelsmann R., Herrmann F. Clonal analysis of bcr-abl rearrangement in T lymphocytes from patients with chronic myelogenous leukemia. Blood. 1992 Feb 15;79(4):1017–1023. [PubMed] [Google Scholar]
- Kelliher M. A., McLaughlin J., Witte O. N., Rosenberg N. Induction of a chronic myelogenous leukemia-like syndrome in mice with v-abl and BCR/ABL. Proc Natl Acad Sci U S A. 1990 Sep;87(17):6649–6653. doi: 10.1073/pnas.87.17.6649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelliher M., Knott A., McLaughlin J., Witte O. N., Rosenberg N. Differences in oncogenic potency but not target cell specificity distinguish the two forms of the BCR/ABL oncogene. Mol Cell Biol. 1991 Sep;11(9):4710–4716. doi: 10.1128/mcb.11.9.4710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konopka J. B., Clark S., McLaughlin J., Nitta M., Kato Y., Strife A., Clarkson B., Witte O. N. Variable expression of the translocated c-abl oncogene in Philadelphia-chromosome-positive B-lymphoid cell lines from chronic myelogenous leukemia patients. Proc Natl Acad Sci U S A. 1986 Jun;83(11):4049–4052. doi: 10.1073/pnas.83.11.4049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lugo T. G., Witte O. N. The BCR-ABL oncogene transforms Rat-1 cells and cooperates with v-myc. Mol Cell Biol. 1989 Mar;9(3):1263–1270. doi: 10.1128/mcb.9.3.1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGuire E. A., Rintoul C. E., Sclar G. M., Korsmeyer S. J. Thymic overexpression of Ttg-1 in transgenic mice results in T-cell acute lymphoblastic leukemia/lymphoma. Mol Cell Biol. 1992 Sep;12(9):4186–4196. doi: 10.1128/mcb.12.9.4186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaughlin J., Chianese E., Witte O. N. Alternative forms of the BCR-ABL oncogene have quantitatively different potencies for stimulation of immature lymphoid cells. Mol Cell Biol. 1989 May;9(5):1866–1874. doi: 10.1128/mcb.9.5.1866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaughlin J., Chianese E., Witte O. N. In vitro transformation of immature hematopoietic cells by the P210 BCR/ABL oncogene product of the Philadelphia chromosome. Proc Natl Acad Sci U S A. 1987 Sep;84(18):6558–6562. doi: 10.1073/pnas.84.18.6558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mes-Masson A. M., McLaughlin J., Daley G. Q., Paskind M., Witte O. N. Overlapping cDNA clones define the complete coding region for the P210c-abl gene product associated with chronic myelogenous leukemia cells containing the Philadelphia chromosome. Proc Natl Acad Sci U S A. 1986 Dec;83(24):9768–9772. doi: 10.1073/pnas.83.24.9768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muller A. J., Young J. C., Pendergast A. M., Pondel M., Landau N. R., Littman D. R., Witte O. N. BCR first exon sequences specifically activate the BCR/ABL tyrosine kinase oncogene of Philadelphia chromosome-positive human leukemias. Mol Cell Biol. 1991 Apr;11(4):1785–1792. doi: 10.1128/mcb.11.4.1785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munroe D. G., Rovinski B., Bernstein A., Benchimol S. Loss of a highly conserved domain on p53 as a result of gene deletion during Friend virus-induced erythroleukemia. Oncogene. 1988 Jun;2(6):621–624. [PubMed] [Google Scholar]
- Nikolić-Zugić J., Bevan M. J. Thymocytes expressing CD8 differentiate into CD4+ cells following intrathymic injection. Proc Natl Acad Sci U S A. 1988 Nov;85(22):8633–8637. doi: 10.1073/pnas.85.22.8633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parmar K., Huebner R. C., Rosenberg N. Carboxyl-terminal determinants of Abelson protein important for lymphoma induction. J Virol. 1991 Dec;65(12):6478–6485. doi: 10.1128/jvi.65.12.6478-6485.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paskind M. P., Weinberg R. A., Baltimore D. Dependence of Moloney murine leukemia virus production on cell growth. Virology. 1975 Sep;67(1):242–248. doi: 10.1016/0042-6822(75)90421-3. [DOI] [PubMed] [Google Scholar]
- Petrie H. T., Hugo P., Scollay R., Shortman K. Lineage relationships and developmental kinetics of immature thymocytes: CD3, CD4, and CD8 acquisition in vivo and in vitro. J Exp Med. 1990 Dec 1;172(6):1583–1588. doi: 10.1084/jem.172.6.1583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poirier Y., Jolicoeur P. Distinct helper virus requirements for Abelson murine leukemia virus-induced pre-B- and T-cell lymphomas. J Virol. 1989 May;63(5):2088–2098. doi: 10.1128/jvi.63.5.2088-2098.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poirier Y., Kozak C., Jolicoeur P. Identification of a common helper provirus integration site in Abelson murine leukemia virus-induced lymphoma DNA. J Virol. 1988 Nov;62(11):3985–3992. doi: 10.1128/jvi.62.11.3985-3992.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prywes R., Foulkes J. G., Rosenberg N., Baltimore D. Sequences of the A-MuLV protein needed for fibroblast and lymphoid cell transformation. Cell. 1983 Sep;34(2):569–579. doi: 10.1016/0092-8674(83)90389-6. [DOI] [PubMed] [Google Scholar]
- Risser R., Kaehler D., Lamph W. W. Different genes control the susceptibility of mice to Moloney or Abelson murine leukemia viruses. J Virol. 1985 Sep;55(3):547–553. doi: 10.1128/jvi.55.3.547-553.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg N., Baltimore D. The effect of helper virus on Abelson virus-induced transformation of lymphoid cells. J Exp Med. 1978 Apr 1;147(4):1126–1141. doi: 10.1084/jem.147.4.1126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg N., Witte O. N. The viral and cellular forms of the Abelson (abl) oncogene. Adv Virus Res. 1988;35:39–81. doi: 10.1016/s0065-3527(08)60708-3. [DOI] [PubMed] [Google Scholar]
- Savard P., DesGroseillers L., Rassart E., Poirier Y., Jolicoeur P. Important role of the long terminal repeat of the helper Moloney murine leukemia virus in Abelson virus-induced lymphoma. J Virol. 1987 Oct;61(10):3266–3275. doi: 10.1128/jvi.61.10.3266-3275.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawyers C. L., Callahan W., Witte O. N. Dominant negative MYC blocks transformation by ABL oncogenes. Cell. 1992 Sep 18;70(6):901–910. doi: 10.1016/0092-8674(92)90241-4. [DOI] [PubMed] [Google Scholar]
- Scherle P. A., Dorshkind K., Witte O. N. Clonal lymphoid progenitor cell lines expressing the BCR/ABL oncogene retain full differentiative function. Proc Natl Acad Sci U S A. 1990 Mar;87(5):1908–1912. doi: 10.1073/pnas.87.5.1908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott M. L., Davis M. M., Feinberg M. B. Transformation of T-lymphoid cells by Abelson murine leukemia virus. J Virol. 1986 Aug;59(2):434–443. doi: 10.1128/jvi.59.2.434-443.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott M. L., Van Etten R. A., Daley G. Q., Baltimore D. v-abl causes hematopoietic disease distinct from that caused by bcr-abl. Proc Natl Acad Sci U S A. 1991 Aug 1;88(15):6506–6510. doi: 10.1073/pnas.88.15.6506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Selten G., Cuypers H. T., Berns A. Proviral activation of the putative oncogene Pim-1 in MuLV induced T-cell lymphomas. EMBO J. 1985 Jul;4(7):1793–1798. doi: 10.1002/j.1460-2075.1985.tb03852.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strife A., Clarkson B. Biology of chronic myelogenous leukemia: is discordant maturation the primary defect? Semin Hematol. 1988 Jan;25(1):1–19. [PubMed] [Google Scholar]
- Tidmarsh G. F., Heimfeld S., Whitlock C. A., Weissman I. L., Müller-Sieburg C. E. Identification of a novel bone marrow-derived B-cell progenitor population that coexpresses B220 and Thy-1 and is highly enriched for Abelson leukemia virus targets. Mol Cell Biol. 1989 Jun;9(6):2665–2671. doi: 10.1128/mcb.9.6.2665. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Watanabe S. M., Witte O. N. Site-directed deletions of Abelson murine leukemia virus define 3' sequences essential for transformation and lethality. J Virol. 1983 Mar;45(3):1028–1036. doi: 10.1128/jvi.45.3.1028-1036.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitlock C. A., Ziegler S. F., Treiman L. J., Stafford J. I., Witte O. N. Differentiation of cloned populations of immature B cells after transformation with Abelson murine leukemia virus. Cell. 1983 Mar;32(3):903–911. doi: 10.1016/0092-8674(83)90075-2. [DOI] [PubMed] [Google Scholar]
- Ziegler S. F., Treiman L. J., Witte O. N. kappa gene diversity among the clonal progeny of pre-B lymphocytes. Proc Natl Acad Sci U S A. 1984 Mar;81(5):1529–1533. doi: 10.1073/pnas.81.5.1529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziegler S. F., Whitlock C. A., Goff S. P., Gifford A., Witte O. N. Lethal effect of the Abelson murine leukemia virus transforming gene product. Cell. 1981 Dec;27(3 Pt 2):477–486. doi: 10.1016/0092-8674(81)90389-5. [DOI] [PubMed] [Google Scholar]
- de Cuevillas F., Lachman H. Transfection of a T-cell line with neo increases dexamethasone cytotoxicity. Leuk Res. 1990;14(7):623–627. doi: 10.1016/0145-2126(90)90017-4. [DOI] [PubMed] [Google Scholar]
- van Lohuizen M., Breuer M., Berns A. N-myc is frequently activated by proviral insertion in MuLV-induced T cell lymphomas. EMBO J. 1989 Jan;8(1):133–136. doi: 10.1002/j.1460-2075.1989.tb03357.x. [DOI] [PMC free article] [PubMed] [Google Scholar]