Abstract
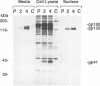
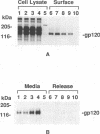
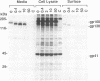
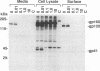
A biotinylation assay was used to detect the envelope glycoprotein of the simian immunodeficiency virus (SIV) envelope glycoprotein expressed by a recombinant vaccinia virus on the surface of HeLa T4 cells. The relationship between the detection of the envelope glycoprotein on the cell surface and its secretion from the cell was examined. It was found that much more gp120 was released into the culture medium than could be accounted for by shedding of the biotinylated SIV envelope protein from the cell surface. Treatment with the ionophore monensin showed that this drug did not block the secretion of gp120 into the culture medium even though the expression of gp120 on the cell surface was strongly downregulated. Similar results were observed for the secretion of gp120 in HUT78 cells infected with SIVmac251 virus. Brefeldin A, on the other hand, inhibited both the detection of gp120 on the cell surface and its secretion into the culture medium. On the basis of these results, we propose that gp120 can be secreted into the culture medium via at least two pathways. One pathway involves the dissociation of gp120 from membrane-associated gp41-gp120 complexes on the cell surface. However, the major pathway involves the secretion of gp120 without its transitory appearance on the cell surface as part of a gp41-gp120 complex.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alonso F. V., Compans R. W. Differential effect of monensin on enveloped viruses that form at distinct plasma membrane domains. J Cell Biol. 1981 Jun;89(3):700–705. doi: 10.1083/jcb.89.3.700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernstein H. B., Compans R. W. Sulfation of the human immunodeficiency virus envelope glycoprotein. J Virol. 1992 Dec;66(12):6953–6959. doi: 10.1128/jvi.66.12.6953-6959.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosch J. V., Schwarz R. T. Processing of gPr92env, the precursor to the glycoproteins of Rous sarcoma virus: use of inhibitors of oligosaccharide trimming and glycoprotein transport. Virology. 1984 Jan 15;132(1):95–109. doi: 10.1016/0042-6822(84)90094-1. [DOI] [PubMed] [Google Scholar]
- Crise B., Buonocore L., Rose J. K. CD4 is retained in the endoplasmic reticulum by the human immunodeficiency virus type 1 glycoprotein precursor. J Virol. 1990 Nov;64(11):5585–5593. doi: 10.1128/jvi.64.11.5585-5593.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dewar R. L., Vasudevachari M. B., Natarajan V., Salzman N. P. Biosynthesis and processing of human immunodeficiency virus type 1 envelope glycoproteins: effects of monensin on glycosylation and transport. J Virol. 1989 Jun;63(6):2452–2456. doi: 10.1128/jvi.63.6.2452-2456.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Earl P. L., Moss B., Doms R. W. Folding, interaction with GRP78-BiP, assembly, and transport of the human immunodeficiency virus type 1 envelope protein. J Virol. 1991 Apr;65(4):2047–2055. doi: 10.1128/jvi.65.4.2047-2055.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geyer H., Holschbach C., Hunsmann G., Schneider J. Carbohydrates of human immunodeficiency virus. Structures of oligosaccharides linked to the envelope glycoprotein 120. J Biol Chem. 1988 Aug 25;263(24):11760–11767. [PubMed] [Google Scholar]
- Ghosh-Choudhury N., Graham A., Ghosh H. P. Herpes simplex virus type 2 glycoprotein biogenesis: effect of monensin on glycoprotein maturation, intracellular transport and virus infectivity. J Gen Virol. 1987 Jul;68(Pt 7):1939–1949. doi: 10.1099/0022-1317-68-7-1939. [DOI] [PubMed] [Google Scholar]
- Griffiths G., Quinn P., Warren G. Dissection of the Golgi complex. I. Monensin inhibits the transport of viral membrane proteins from medial to trans Golgi cisternae in baby hamster kidney cells infected with Semliki Forest virus. J Cell Biol. 1983 Mar;96(3):835–850. doi: 10.1083/jcb.96.3.835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hallenberger S., Bosch V., Angliker H., Shaw E., Klenk H. D., Garten W. Inhibition of furin-mediated cleavage activation of HIV-1 glycoprotein gp160. Nature. 1992 Nov 26;360(6402):358–361. doi: 10.1038/360358a0. [DOI] [PubMed] [Google Scholar]
- Haseltine W. A. Molecular biology of the human immunodeficiency virus type 1. FASEB J. 1991 Jul;5(10):2349–2360. doi: 10.1096/fasebj.5.10.1829694. [DOI] [PubMed] [Google Scholar]
- Helseth E., Olshevsky U., Furman C., Sodroski J. Human immunodeficiency virus type 1 gp120 envelope glycoprotein regions important for association with the gp41 transmembrane glycoprotein. J Virol. 1991 Apr;65(4):2119–2123. doi: 10.1128/jvi.65.4.2119-2123.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holschbach C., Schneider J., Geyer H. Glycosylation of the envelope glycoprotein gp130 of simian immunodeficiency virus from sooty mangabey (Cercocebus atys). Biochem J. 1990 May 1;267(3):759–766. doi: 10.1042/bj2670759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson D. C., Schlesinger M. J. Vesicular stomatitis virus and sindbis virus glycoprotein transport to the cell surface is inhibited by ionophores. Virology. 1980 Jun;103(2):407–424. doi: 10.1016/0042-6822(80)90200-7. [DOI] [PubMed] [Google Scholar]
- Klausner R. D., Donaldson J. G., Lippincott-Schwartz J. Brefeldin A: insights into the control of membrane traffic and organelle structure. J Cell Biol. 1992 Mar;116(5):1071–1080. doi: 10.1083/jcb.116.5.1071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kowalski M., Potz J., Basiripour L., Dorfman T., Goh W. C., Terwilliger E., Dayton A., Rosen C., Haseltine W., Sodroski J. Functional regions of the envelope glycoprotein of human immunodeficiency virus type 1. Science. 1987 Sep 11;237(4820):1351–1355. doi: 10.1126/science.3629244. [DOI] [PubMed] [Google Scholar]
- Le Bivic A., Real F. X., Rodriguez-Boulan E. Vectorial targeting of apical and basolateral plasma membrane proteins in a human adenocarcinoma epithelial cell line. Proc Natl Acad Sci U S A. 1989 Dec;86(23):9313–9317. doi: 10.1073/pnas.86.23.9313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee W. R., Yu X. F., Syu W. J., Essex M., Lee T. H. Mutational analysis of conserved N-linked glycosylation sites of human immunodeficiency virus type 1 gp41. J Virol. 1992 Mar;66(3):1799–1803. doi: 10.1128/jvi.66.3.1799-1803.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKeating J. A., McKnight A., Moore J. P. Differential loss of envelope glycoprotein gp120 from virions of human immunodeficiency virus type 1 isolates: effects on infectivity and neutralization. J Virol. 1991 Feb;65(2):852–860. doi: 10.1128/jvi.65.2.852-860.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKeating J. A., Willey R. L. Structure and function of the HIV envelope. AIDS. 1989;3 (Suppl 1):S35–S41. doi: 10.1097/00002030-198901001-00005. [DOI] [PubMed] [Google Scholar]
- Mulligan M. J., Yamshchikov G. V., Ritter G. D., Jr, Gao F., Jin M. J., Nail C. D., Spies C. P., Hahn B. H., Compans R. W. Cytoplasmic domain truncation enhances fusion activity by the exterior glycoprotein complex of human immunodeficiency virus type 2 in selected cell types. J Virol. 1992 Jun;66(6):3971–3975. doi: 10.1128/jvi.66.6.3971-3975.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orenstein J. M., Meltzer M. S., Phipps T., Gendelman H. E. Cytoplasmic assembly and accumulation of human immunodeficiency virus types 1 and 2 in recombinant human colony-stimulating factor-1-treated human monocytes: an ultrastructural study. J Virol. 1988 Aug;62(8):2578–2586. doi: 10.1128/jvi.62.8.2578-2586.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pal R., Gallo R. C., Sarngadharan M. G. Processing of the structural proteins of human immunodeficiency virus type 1 in the presence of monensin and cerulenin. Proc Natl Acad Sci U S A. 1988 Dec;85(23):9283–9286. doi: 10.1073/pnas.85.23.9283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pal R., Mumbauer S., Hoke G. M., Takatsuki A., Sarngadharan M. G. Brefeldin A inhibits the processing and secretion of envelope glycoproteins of human immunodeficiency virus type 1. AIDS Res Hum Retroviruses. 1991 Aug;7(8):707–712. doi: 10.1089/aid.1991.7.707. [DOI] [PubMed] [Google Scholar]
- Sattentau Q. J., Moore J. P. Conformational changes induced in the human immunodeficiency virus envelope glycoprotein by soluble CD4 binding. J Exp Med. 1991 Aug 1;174(2):407–415. doi: 10.1084/jem.174.2.407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider J., Kaaden O., Copeland T. D., Oroszlan S., Hunsmann G. Shedding and interspecies type sero-reactivity of the envelope glycopolypeptide gp120 of the human immunodeficiency virus. J Gen Virol. 1986 Nov;67(Pt 11):2533–2538. doi: 10.1099/0022-1317-67-11-2533. [DOI] [PubMed] [Google Scholar]
- Smilowitz H. Routes of intracellular transport of acetylcholine receptor and esterase are distinct. Cell. 1980 Jan;19(1):237–244. doi: 10.1016/0092-8674(80)90405-5. [DOI] [PubMed] [Google Scholar]
- Srinivas R. V., Melsen L. R., Compans R. W. Effects of monensin on morphogenesis and infectivity of Friend murine leukemia virus. J Virol. 1982 Jun;42(3):1067–1075. doi: 10.1128/jvi.42.3.1067-1075.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stein B. S., Engleman E. G. Intracellular processing of the gp160 HIV-1 envelope precursor. Endoproteolytic cleavage occurs in a cis or medial compartment of the Golgi complex. J Biol Chem. 1990 Feb 15;265(5):2640–2649. [PubMed] [Google Scholar]
- Willey R. L., Bonifacino J. S., Potts B. J., Martin M. A., Klausner R. D. Biosynthesis, cleavage, and degradation of the human immunodeficiency virus 1 envelope glycoprotein gp160. Proc Natl Acad Sci U S A. 1988 Dec;85(24):9580–9584. doi: 10.1073/pnas.85.24.9580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willey R. L., Klimkait T., Frucht D. M., Bonifacino J. S., Martin M. A. Mutations within the human immunodeficiency virus type 1 gp160 envelope glycoprotein alter its intracellular transport and processing. Virology. 1991 Sep;184(1):319–329. doi: 10.1016/0042-6822(91)90848-6. [DOI] [PubMed] [Google Scholar]