Abstract
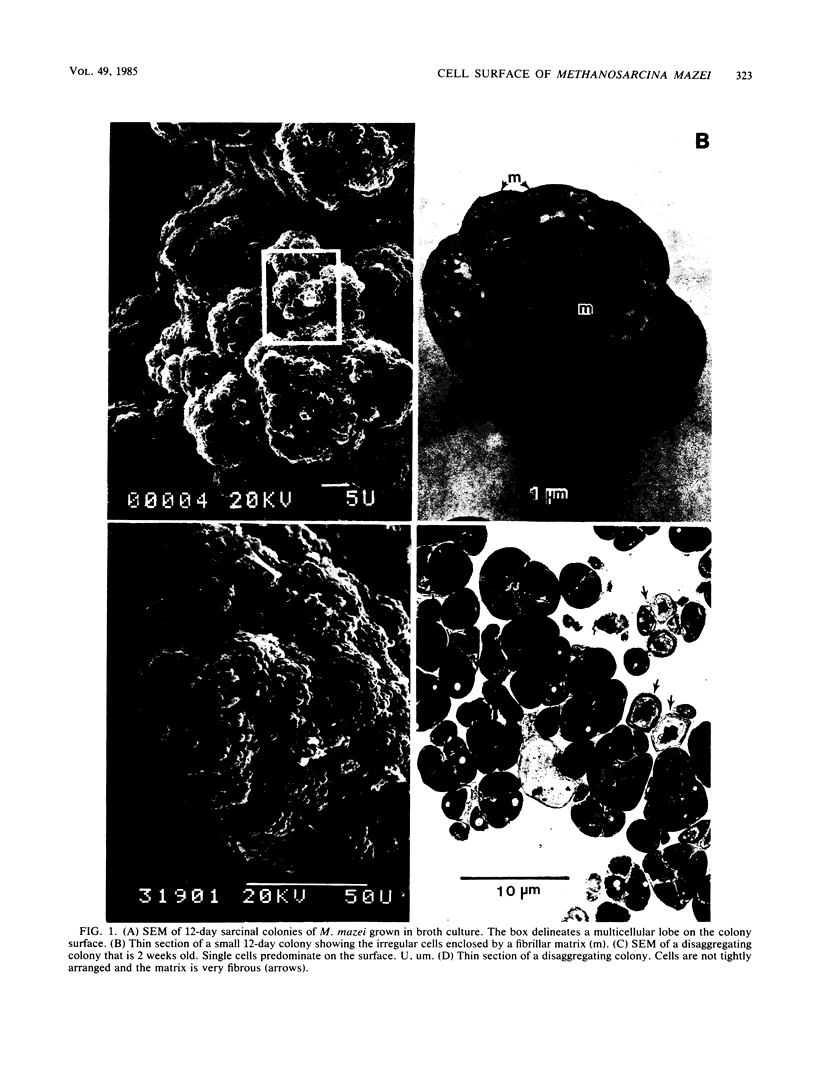
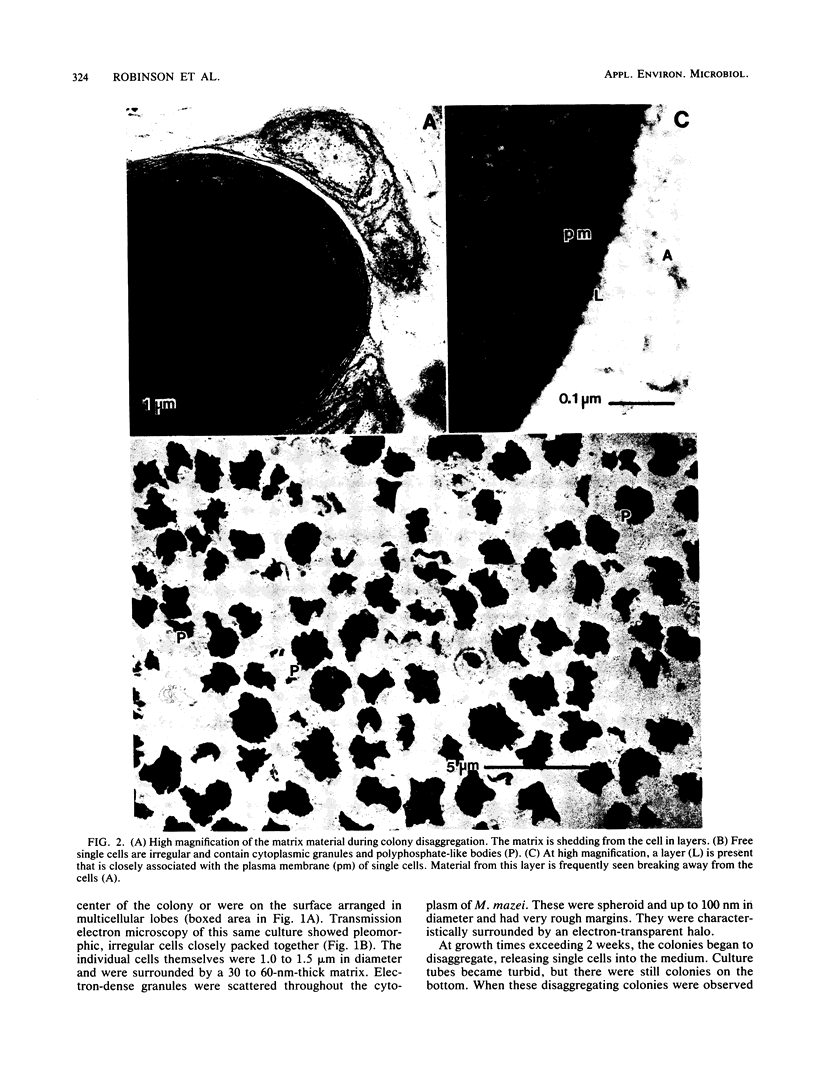
Colonial aggregates of Methanosarcina (= Methanococcus) mazei were examined with scanning and transmission electron microscopy. Cells are irregular and grouped into multicellular sarcinal colonies, which may disaggregate in older cultures. The protoplast is bounded by a typical trilaminar plasma membrane, outside of which is a matrix of loose fibrils. The presence and compactness of matrix material are responsible for the close packing of cells, and colony disaggregation seems to be the result of matrix shedding and degradation. The cell envelope contains complex hetero polysaccharides of N-acetylgalactosamine and galacturonic and glucuronic acids. Polymers extruded by M. mazei are likely quite adhesive in nature, accounting for its strong adherence to surfaces and hardiness compared with many other methanogens.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BLEIWEIS A. S., KARAKAWA W. W., KRAUSE R. M. IMPROVED TECHNIQUE FOR THE PREPARATION OF STREPTOCOCCAL CELL WALLS. J Bacteriol. 1964 Oct;88:1198–1200. doi: 10.1128/jb.88.4.1198-1200.1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balch W. E., Fox G. E., Magrum L. J., Woese C. R., Wolfe R. S. Methanogens: reevaluation of a unique biological group. Microbiol Rev. 1979 Jun;43(2):260–296. doi: 10.1128/mr.43.2.260-296.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Martino C., Zamboni L. Silver methenamine stain for electron microscopy. J Ultrastruct Res. 1967 Aug;19(3):273–282. doi: 10.1016/s0022-5320(67)80221-1. [DOI] [PubMed] [Google Scholar]
- Erlandsen S. L., Parsons J. A., Rodning C. B. Technical parameters of immunostaining of osmicated tissue in epoxy sections. J Histochem Cytochem. 1979 Sep;27(9):1286–1289. doi: 10.1177/27.9.90080. [DOI] [PubMed] [Google Scholar]
- Griggs L. J., Post A., White E. R., Finkelstein J. A., Moeckel W. E., Holden K. G., Zarembo J. E., Weisbach J. A. Identification and quantitation of alditol acetates of neutral and amino sugars from mucins by automated gas-liquid chromatography. Anal Biochem. 1971 Oct;43(2):369–381. doi: 10.1016/0003-2697(71)90266-1. [DOI] [PubMed] [Google Scholar]
- Horisberger M., Rosset J., Bauer H. Colloidal gold granules as markers for cell surface receptors in the scanning electron microscope. Experientia. 1975 Oct 15;31(10):1147–1149. doi: 10.1007/BF02326761. [DOI] [PubMed] [Google Scholar]
- Jones J. B., Bowers B., Stadtman T. C. Methanococcus vannielii: ultrastructure and sensitivity to detergents and antibiotics. J Bacteriol. 1977 Jun;130(3):1357–1363. doi: 10.1128/jb.130.3.1357-1363.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kandler O., Hippe H. Lack of peptidoglycan in the cell walls of Methanosarcina barkeri. Arch Microbiol. 1977 May 13;113(1-2):57–60. doi: 10.1007/BF00428580. [DOI] [PubMed] [Google Scholar]
- Kandler O., König H. Chemical composition of the peptidoglycan-free cell walls of methanogenic bacteria. Arch Microbiol. 1978 Aug 1;118(2):141–152. doi: 10.1007/BF00415722. [DOI] [PubMed] [Google Scholar]
- König H., Kandler O., Jensen M., Rietschel E. T. The primary structure of the glycan moiety of pseudomurein from Methanobacterium thermoautotrophicum. Hoppe Seylers Z Physiol Chem. 1983 Jun;364(6):627–636. doi: 10.1515/bchm2.1983.364.1.627. [DOI] [PubMed] [Google Scholar]
- König H., Kandler O. The amino acid sequence of the peptide moiety of the pseudomurein from Methanobacterium thermoautotrophicum. Arch Microbiol. 1979 Jun;121(3):271–275. doi: 10.1007/BF00425067. [DOI] [PubMed] [Google Scholar]
- MOLLENHAUER H. H. PLASTIC EMBEDDING MIXTURES FOR USE IN ELECTRON MICROSCOPY. Stain Technol. 1964 Mar;39:111–114. [PubMed] [Google Scholar]
- Mah R. A. The methanogenic bacteria, their ecology and physiology. Basic Life Sci. 1981;18:357–374. doi: 10.1007/978-1-4684-3980-9_21. [DOI] [PubMed] [Google Scholar]
- Pickett-Heaps J. D. Preliminary attempts at ultrastructural polysaccharide localization in root tip cells. J Histochem Cytochem. 1967 Aug;15(8):442–455. doi: 10.1177/15.8.442. [DOI] [PubMed] [Google Scholar]
- REYNOLDS E. S. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol. 1963 Apr;17:208–212. doi: 10.1083/jcb.17.1.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth J., Binder M. Coloidal gold, ferritin and peroxidase as markers for electron microscopic double labeling lectin techniques. J Histochem Cytochem. 1978 Mar;26(3):163–169. doi: 10.1177/26.3.632554. [DOI] [PubMed] [Google Scholar]
- Sowers K. R., Baron S. F., Ferry J. G. Methanosarcina acetivorans sp. nov., an Acetotrophic Methane-Producing Bacterium Isolated from Marine Sediments. Appl Environ Microbiol. 1984 May;47(5):971–978. doi: 10.1128/aem.47.5.971-978.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sowers K. R., Ferry J. G. Isolation and Characterization of a Methylotrophic Marine Methanogen, Methanococcoides methylutens gen. nov., sp. nov. Appl Environ Microbiol. 1983 Feb;45(2):684–690. doi: 10.1128/aem.45.2.684-690.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spurr A. R. A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res. 1969 Jan;26(1):31–43. doi: 10.1016/s0022-5320(69)90033-1. [DOI] [PubMed] [Google Scholar]
- Taylor R. L., Conrad H. E. Stoichiometric depolymerization of polyuronides and glycosaminoglycuronans to monosaccharides following reduction of their carbodiimide-activated carboxyl groups. Biochemistry. 1972 Apr 11;11(8):1383–1388. doi: 10.1021/bi00758a009. [DOI] [PubMed] [Google Scholar]
- Zeikus J. G., Bowen V. G. Comparative ultrastructure of methanogenic bacteria. Can J Microbiol. 1975 Feb;21(2):121–129. doi: 10.1139/m75-019. [DOI] [PubMed] [Google Scholar]