Abstract
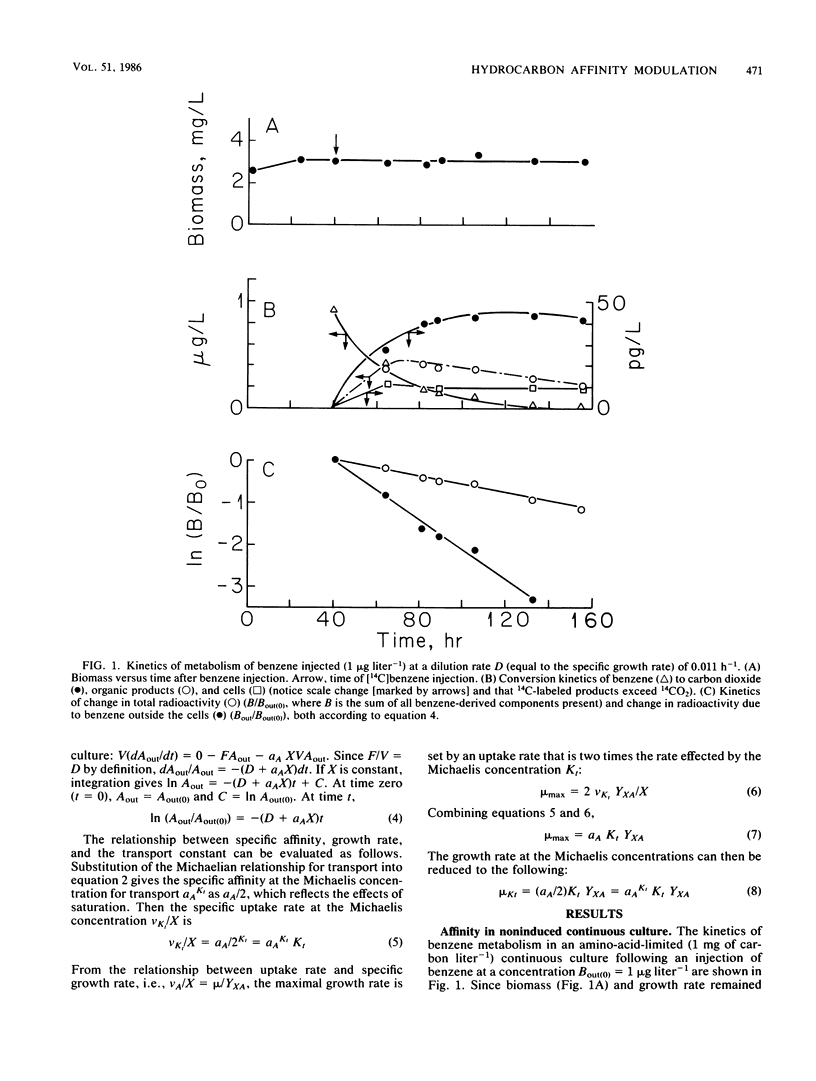
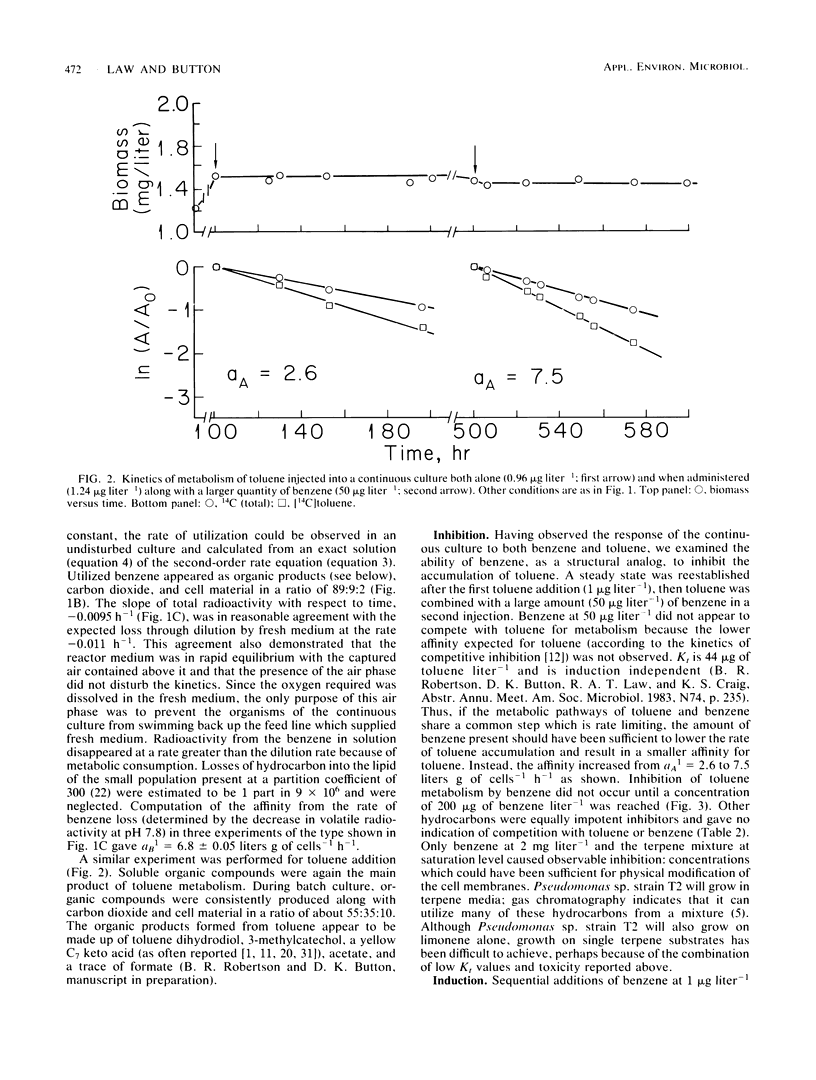
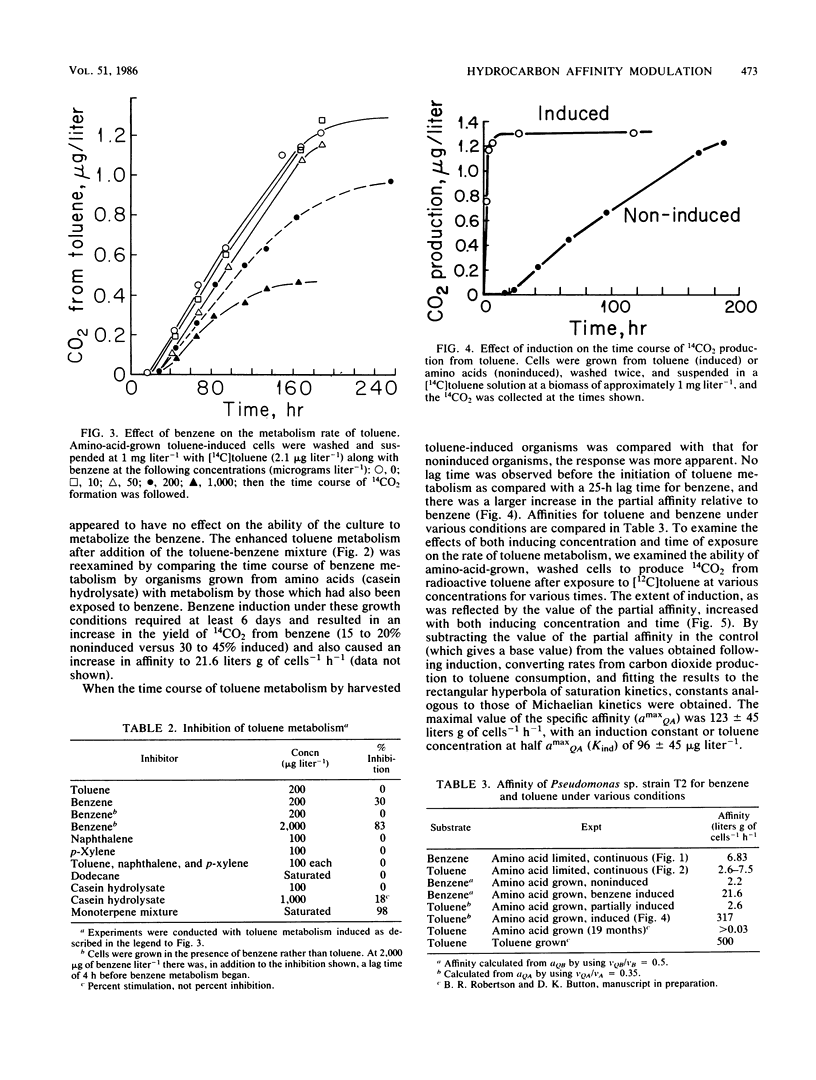
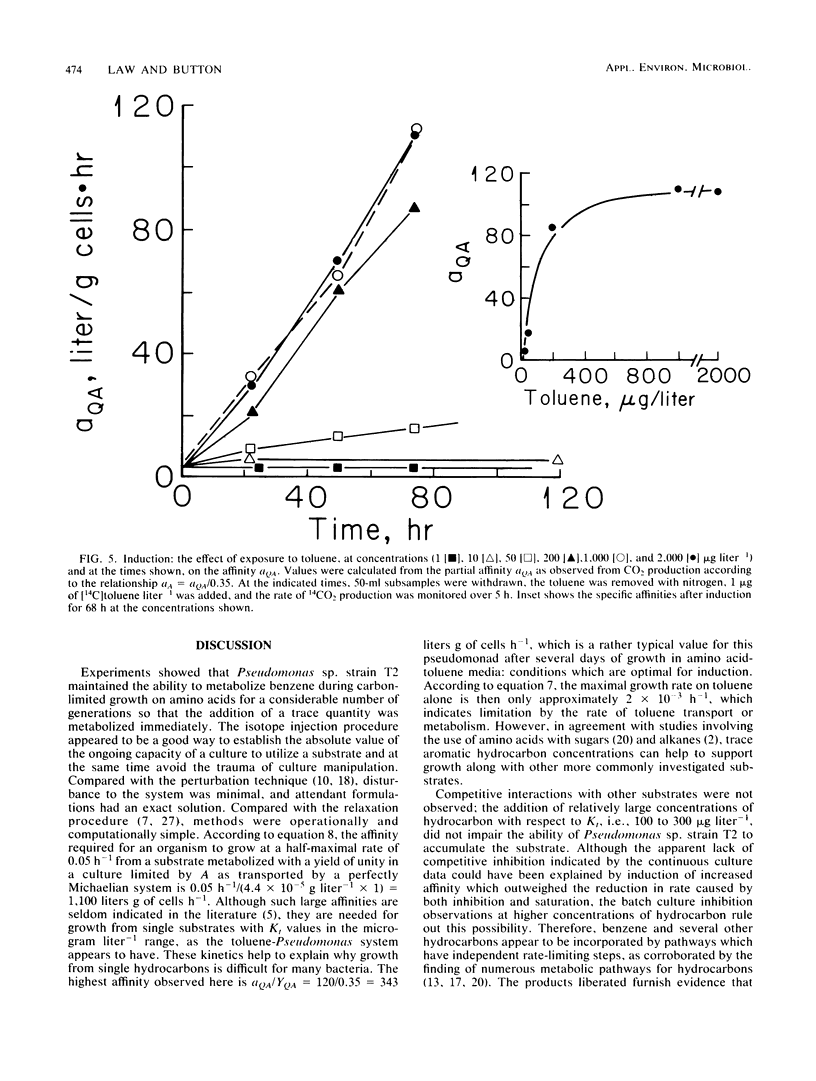
Trace (microgram liter−1) quantities of either toluene or benzene injected into an amino-acid-limited continuous culture of Pseudomonas sp. strain T2 were utilized immediately with affinities of 2.6 and 6.8 liters g of cells−1 h−1, respectively, and yielded large amounts of organic products, carbon dioxide, and cells. The immediate utilization of hydrocarbons by hydrocarbon-deprived organisms helps to establish the nutritional value of nonpolar substrates in the environment. The observation of small Michaelis constants for toluene transport led to tests of metabolic competition between hydrocarbons; however, competitive inhibition of toluene metabolism was not found for benzene, naphthalene, xylene, dodecane, or amino acids. Benzene and terpenes were inhibitory at milligram liter−1 concentrations. Toluene was metabolized by a strongly inducible system when compared with benzene. The capacity of toluene to effect larger affinity values increased with exposure time and concentration. The kinetics of induction suggested saturation phenomena, resulting in an induction constant, Kind, of 96 μg of toluene liter−1. Maximal induction of amino-acid-grown cells required about 80 h, with the affinity reaching 317 liters g of cells−1 h−1.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bayly R. C., Dagley S., Gibson D. T. The metabolism of cresols by species of Pseudomonas. Biochem J. 1966 Nov;101(2):293–301. doi: 10.1042/bj1010293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benson S., Fennewald M., Shapiro J., Huettner C. Fractionation of inducible alkane hydroxylase activity in Pseudomonas putida and characterization of hydroxylase-negative plasmid mutations. J Bacteriol. 1977 Nov;132(2):614–621. doi: 10.1128/jb.132.2.614-621.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Button D. K. Evidence for a terpene-based food chain in the gulf of alaska. Appl Environ Microbiol. 1984 Nov;48(5):1004–1011. doi: 10.1128/aem.48.5.1004-1011.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Button D. K. Kinetics of nutrient-limited transport and microbial growth. Microbiol Rev. 1985 Sep;49(3):270–297. doi: 10.1128/mr.49.3.270-297.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Button D. K., Robertson B. R., Craig K. S. Dissolved hydrocarbons and related microflora in a fjordal seaport: sources, sinks, concentrations, and kinetics. Appl Environ Microbiol. 1981 Oct;42(4):708–719. doi: 10.1128/aem.42.4.708-719.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Button D. K., Schell D. M., Robertson B. R. Sensitive and accurate methodology for measuring the kinetics of concentration-dependent hydrocarbon metabolism rates in seawater by microbial communities. Appl Environ Microbiol. 1981 Apr;41(4):936–941. doi: 10.1128/aem.41.4.936-941.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CLAUS D., WALKER N. THE DECOMPOSITION OF TOLUENE BY SOIL BACTERIA. J Gen Microbiol. 1964 Jul;36:107–122. doi: 10.1099/00221287-36-1-107. [DOI] [PubMed] [Google Scholar]
- DAGLEY S., GIBSON D. T. THE BACTERIAL DEGRADATION OF CATECHOL. Biochem J. 1965 May;95:466–474. doi: 10.1042/bj0950466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durham D. R., McNamee C. G., Stewart D. B. Dissimilation of aromatic compounds in Rhodotorula graminis: biochemical characterization of pleiotropically negative mutants. J Bacteriol. 1984 Nov;160(2):771–777. doi: 10.1128/jb.160.2.771-777.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fall R. R., Brown J. L., Schaeffer T. L. Enzyme recruitment allows the biodegradation of recalcitrant branched hydrocarbons by Pseudomonas citronellolis. Appl Environ Microbiol. 1979 Oct;38(4):715–722. doi: 10.1128/aem.38.4.715-722.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feist C. F., Hegeman G. D. Phenol and benzoate metabolism by Pseudomonas putida: regulation of tangential pathways. J Bacteriol. 1969 Nov;100(2):869–877. doi: 10.1128/jb.100.2.869-877.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson D. T. Microbial degradation of aromatic compounds. Science. 1967 Sep 13;161(3846):1093–1097. [PubMed] [Google Scholar]
- Kunz D. A., Chapman P. J. Isolation and characterization of spontaneously occurring TOL plasmid mutants of Pseudomonas putida HS1. J Bacteriol. 1981 Jun;146(3):952–964. doi: 10.1128/jb.146.3.952-964.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Law A. T., Button D. K. Multiple-carbon-source-limited growth kinetics of a marine coryneform bacterium. J Bacteriol. 1977 Jan;129(1):115–123. doi: 10.1128/jb.129.1.115-123.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray K., Williams P. A. Role of catechol and the methylcatechols as inducers of aromatic metabolism in Pseudomonas putida. J Bacteriol. 1974 Mar;117(3):1153–1157. doi: 10.1128/jb.117.3.1153-1157.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura L. K., Tyler D. D. Induction of D-aldohexoside:cytochrome c oxidoreductase in Agrobacterium tumefaciens. J Bacteriol. 1977 Feb;129(2):830–835. doi: 10.1128/jb.129.2.830-835.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfaender F. K., Bartholomew G. W. Measurement of aquatic biodegradation rates by determining heterotrophic uptake of radiolabeled pollutants. Appl Environ Microbiol. 1982 Jul;44(1):159–164. doi: 10.1128/aem.44.1.159-164.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robertson B. R., Button D. K. Phosphate-limited continuous culture of Rhodotorula rubra: kinetics of transport, leakage, and growth. J Bacteriol. 1979 Jun;138(3):884–895. doi: 10.1128/jb.138.3.884-895.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spain J. C., Pritchard P. H., Bourquin A. W. Effects of adaptation on biodegradation rates in sediment/water cores from estuarine and freshwater environments. Appl Environ Microbiol. 1980 Oct;40(4):726–734. doi: 10.1128/aem.40.4.726-734.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams P. A., Worsey M. J. Ubiquity of plasmids in coding for toluene and xylene metabolism in soil bacteria: evidence for the existence of new TOL plasmids. J Bacteriol. 1976 Mar;125(3):818–828. doi: 10.1128/jb.125.3.818-828.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeh W. K., Gibson D. T., Liu T. N. Toluene dioxygenase: a multicomponent enzyme system. Biochem Biophys Res Commun. 1977 Sep 9;78(1):401–410. doi: 10.1016/0006-291x(77)91268-2. [DOI] [PubMed] [Google Scholar]



