Abstract
The polycystic kidney diseases (PKD) are a group of genetic disorders causing renal failure and death from infancy to adulthood. Arginine vasopressin (AVP) V2 receptor antagonists inhibit cystogenesis in animal models of cystic kidney diseases, presumably by downregulating cAMP signaling, cell proliferation, and chloride-driven fluid secretion. For confirmation that the protective effect of these drugs is due to antagonism of AVP, PCK (Pkhd1−/−) and Brattleboro (AVP−/−) rats were crossed to generate rats with PKD and varying amounts of AVP. At 10 and 20 weeks of age, PCK AVP−/− rats had lower renal cAMP and almost complete inhibition of cystogenesis compared with PCK AVP+/+ and PCK AVP+/− rats. The V2 receptor agonist 1-deamino-8-d-arginine vasopressin increased renal cAMP and recovered the full cystic phenotype of PCK AVP−/− rats and aggravated the cystic disease of PCK AVP+/+ rats but did not induce cystic changes in wild-type rats. These observations indicate that AVP is a powerful modulator of cystogenesis and provide further support for clinical trials of V2 receptor antagonists in PKD.
Autosomal dominant polycystic kidney disease (ADPKD) is the most common of the inherited renal cystic diseases and a leading cause of ESRD. It is genetically heterogeneous with two genes identified, PKD1 and PKD2.1 Autosomal recessive PKD is less common than ADPKD but together with nephronophthisis is a leading cause of ESRD in childhood. It is caused by mutations to PKHD1. The proteins encoded by PKD1 (polycystin-1), PKD2 (polycystin-2), and PKHD1 (fibrocystin/polyductin) are membrane-associated proteins. The downstream signaling pathways associated with these proteins provide potential therapeutic targets.2 Arginine vasopressin (AVP) V2 receptor antagonists (OPC31260 and OPC41061) inhibit cystogenesis in animal models orthologous to human autosomal recessive PKD (PCK rat), ADPKD (Pkd2−/tm1Som), and adolescent nephronophthisis (pcy mouse) by downregulating cAMP signaling, cell proliferation, and chloride-driven fluid secretion.3–5 Clinical trials with V2 receptor antagonists have been initiated. The mechanism by which these drugs inhibit the development of polycystic kidney disease has not been fully elucidated. In addition to V2 antagonism, these drugs could exert an effect by favoring the action of AVP on V1a receptors or by an AVP unrelated mechanism.
RESULTS AND DISCUSSION
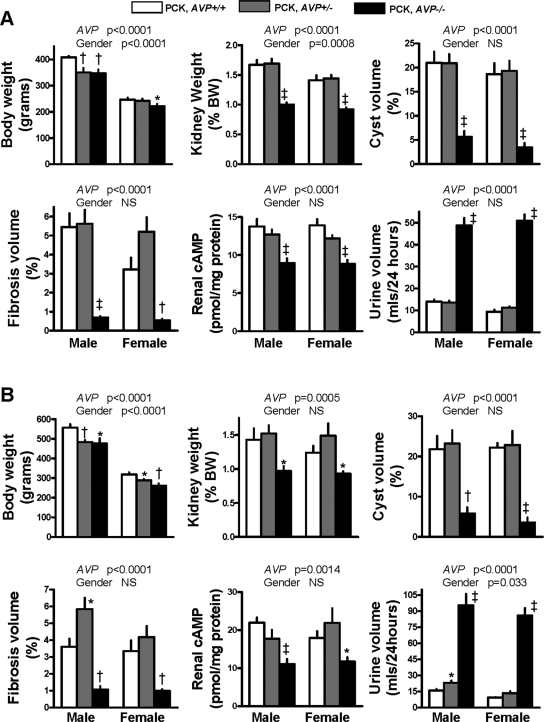
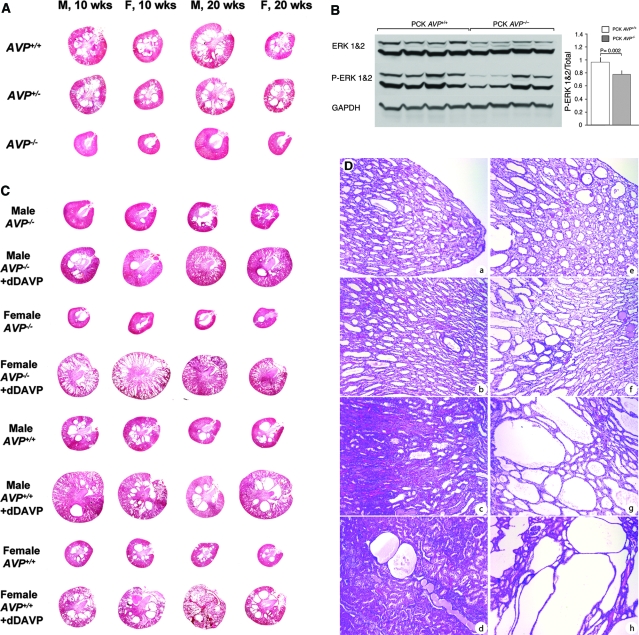
To confirm that inhibition of AVP action on the kidney accounts for the effect of V2 receptor antagonists on the development of PKD, we generated PCK AVP+/+, PCK AVP+/−, and PCK AVP−/− rats, as well as wild-type and Brattleboro controls, by breeding F1 rats resulting from PCK (Pkhd1−/−) and Brattleboro (AVP−/−) crosses. PCK rats are homozygous for a splicing mutation (IVS35-2A→T) that skips exon 36 and leads to a frame shift in Pkhd1.6 Brattleboro rats lack circulating AVP and are homozygous for a 1-bp deletion of a guanine nucleotide in the second exon of the AVP gene that results in a frame shift of the coding sequence for the carrier neurophysin II.7 Approximately 10 male and 10 female rats of each genotype were killed at 10 and 20 wk of age (Figures 1 and 2, A and B; Supplementary Tables 1 and 2). PCK AVP−/− rats and Brattleboro controls had marked polyuria and a slight increase in plasma sodium concentration. Renal cAMP content was markedly reduced in the PCK AVP−/− compared with the PCK AVP+/+ rats. This was accompanied by decreased phosphorylation of extracellular signal–regulated kinase (ERK) and a marked reduction in kidney weight and renal cyst and fibrosis volumes. No significant differences in liver or hepatic cyst or fibrosis volumes were detected, indicating that the protective effect of vasopressin deficiency is restricted to the kidneys. These results are consistent with the renal protective effects of AVP V2 receptor antagonists (OPC31260 and OPC41061)3–5 and inhibition of vasopressin by forced water ingestion in the PCK rat.8
Figure 1.
Body and kidney weights, indices of cystic disease severity, renal cAMP, and urine output, in 10-wk-old (A) and 20-wk-old (B) PCK AVP+/+, PCK AVP+/−, and PCK AVP−/− rats. Two-way ANOVA P values for AVP gene and gender effect are shown above the bar graphs. *P < 0.05, †P < 0.01, and ‡P < 0.001 versus PCK AVP+/+ rats.
Figure 2.
(A) Representative hematoxylin- and eosin-stained kidney sections from 10- and 20-wk-old male and female PCK AVP+/+, PCK AVP+/−, and PCK AVP−/− rats. PCK AVP−/− kidneys exhibit nearly complete protection from cystic disease. (B) Western blot of total and phosphorylated ERK1/2 and glyceraldehyde-3-phosphate dehydrogenase. ERK1/2 phosphorylation is significantly reduced in PCK AVP−/− compared with PCK AVP+/+ kidneys. (C) Hematoxylin- and eosin-stained kidney sections from male and female PCK AVP−/− and PCK AVP+/+ rats (n = 4 for each genotype and gender) treated continuously with dDAVP (10 ng/h per 100 g body wt) or vehicle alone between 12 and 20 wk of age. The administration of dDAVP recovered the cystic phenotype of the PCK AVP−/− and aggravated the cystic disease of the PCK AVP+/+ rats. (D) Representative photomicrographs of papilla, inner medulla, outer medulla, and cortex from male PCK AVP−/− rats treated continuously with vehicle (a through d) or dDAVP (e through h) between 12 and 20 wk of age. Note the mild focal dilation of collecting ducts in animals treated with vehicle alone. Administration of dDAVP results in diffuse dilation of collecting ducts, which is particularly prominent in the outer medulla and cortex. Magnification, ×100 (hematoxylin and eosin).
PCK AVP+/− rats, like PCK AVP−/− rats and Brattleboro controls, had slightly reduced body weights, but their renal cAMP levels, kidney weights, and cyst and fibrosis volumes were not lower than those in PCK AVP+/+ rats (Figures 1 and 2A, Supplementary Tables 1 and 2). Male PCK AVP+/− rats had slightly higher urine outputs and lower urine osmolalities than PCK AVP+/+ rats, particularly at 20 wk of age. This likely reflects a mild vasopressin deficiency that may become more evident with aging9 but is not sufficient to affect the renal concentration of cAMP or the development of PKD.
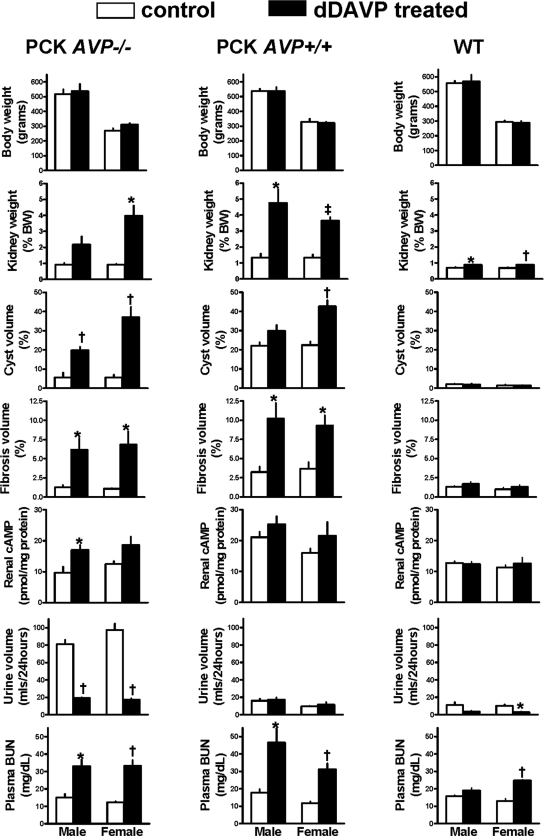
For confirmation that the protective effect of AVP deficiency on the development of PKD is due the lack of stimulation of the renal V2 receptors, PCK AVP−/−, PCK AVP+/+, and wild-type rats were treated with the V2 agonist 1-deamino-8-d-arginine vasopressin (dDAVP) administered via osmotic minipump at a dosage of 10 ng/h per 100 g body wt between 12 and 20 wk of age. This dosage is necessary to achieve urine osmolalities in Brattleboro rats similar to those observed in wild-type Sprague-Dawley rats.10 Administration of dDAVP to PCK AVP−/− reversed the polyuria, increased the renal concentration of cAMP, recovered the full cystic PCK phenotype as reflected by the kidney weights and cyst and fibrosis indices, and significantly increased the plasma blood urea nitrogen concentrations (Figures 2, C and D, and 3; Supplementary Table 3). Administration of dDAVP to wild-type rats at the dosage used in this study caused a slight but significant increase in renal mass per unit of body weight without inducing cystic changes or fibrosis. This is consistent with previous reports of selective AVP-induced hypertrophy of the medullary thick ascending limb in wild-type and Brattleboro rats.11,12 Consistent with the urine-concentrating defect and resistance to vasopressin associated with PKD,13 24-h urine volumes were significantly higher and antidiuretic responses to dDAVP blunted in PCK AVP+/+ compared with wild-type rats.
Figure 3.
Effects of continuous subcutaneous administration of dDAVP (10 ng/h per 100 g body wt) or vehicle alone from 12 to 20 wk of age on body and kidney weights, urine output, indices of cystic disease severity, and laboratory parameters in PCK AVP−/− and PCK AVP+/+ rats. The administration of dDAVP recovered the cystic phenotype of the PCK AVP−/− and aggravated the cystic disease of the PCK AVP+/+ rats. *P < 0.05, †P < 0.01, and ‡P < 0.001 versus control rats receiving vehicle alone.
As shown in previous studies,3–5,14 the renal cystic disease in untreated PCK rats is more severe in males than in females. The cause of this dimorphism is not known. Our study suggests a possible relationship to gender-related differences in the secretion of AVP and its subsequent actions on the kidney. Release of AVP into the circulation in response to osmotic stimulation,15 plasma and urine AVP levels, V2 receptor density and AVP-dependent cAMP production in collecting ducts,16–20 antidiuretic effects of endogenous (water deprivation)21 and exogenous AVP,16–19 and AVP-dependent (or dDAVP-dependent) susceptibility to develop desoxycorticosterone acetate-salt hypertension16–19 all are higher in male than in female rats. The administration of dDAVP in our study eliminated the sexual dimorphism of PKD, possibly because the circulating concentration of dDAVP achieved in our study exceeded the range where estradiol is capable of modulating its antidiuretic action.16–19
These observations may be relevant to patients with ADPKD. AVP secretion is more sensitive to osmotic stimuli,22 and plasma and/or urinary AVP levels23–25 and urine osmolality26 are higher in men than in women. Whether men have a higher or a lower renal sensitivity to AVP than women is not clear, because studies on this issue are not in agreement.20,27 Higher AVP levels and gender differences in urine concentration may contribute to the faster rate of cystic expansion28 and the earlier age of ESRD in men with ADPKD as observed in some29,30 but not all31,32 studies.
In summary, the results of this study indicate that AVP is a powerful modulator of cystogenesis. In the evolution from aquatic vertebrates to tetrapods, vasotocin in birds and vasopressin in mammals became essential for water homeostasis and survival.33 To avoid dehydration, humans live under the tonic influence of circulating AVP and persistent activation of adenylyl cyclase in the distal nephron and collecting duct. Only for short periods of time, after drinking large volumes of liquid, do plasma AVP levels decrease enough to render the urine more dilute than plasma. During most of the day, cyst epithelial cells are persistently stimulated to proliferate and secrete fluid. This may be further enhanced by the renal concentrating defect and increased circulating levels of AVP that occur in PKD. These observations provide further support for clinical trials of V2 receptor antagonists in this disease.
CONCISE METHODS
Experimental Animals and Study Design
The use of PCK rats (maintained at the animal facilities of the Mayo Clinic, Rochester, MN) and Brattleboro rats (Harlan Sprague-Dawley Inc., Indianapolis, IN) and the experimental protocols were approved by the Institutional Animal Care and Utilization Committee.
Genotyping
In PCK rats, the Pkhd1 exon 36 is missing and genomic sequencing showed the mutation to be an A (labeled with VIC)→T (labeled with FAM) transversion at the −2 position of IVS35.6 In Brattleboro rats, there is a single nucleotide deletion (G, labeled with VIC, and N, labeled with FAM) in exon B of the AVP gene.7 Genomic DNA was extracted from rat tail using QIAamp DNA Mini kit (Qiagen, Valencia, CA). Genotyping for the Pkhd1 substitution and a single base deletion in AVP was performed using commercially available custom-made TaqMan Genotyping assays. Real-time TaqMan PCR was performed according to the manufacturer's standard PCR. Twenty nanograms of total DNA was mixed with the 2× TaqMan Universal PCR Master Mix No AmpErase UNG and TaqMan Assay Mix to a final volume of 5 μl. Each sample underwent 40 amplification cycles on an ABI. Two fluorogenic minor groove binder probes were used for each locus using the dyes 6-carboxyfluorescein (FAM; excitation, 494 nm) and VIC (excitation, 538 nm), which are easily differentiated in the Applied Biosystems Prism 7900HT PCR system. The resulting cluster plot showed strong fluorescent signals for each allele and clear separation between the three clusters, easily discriminating the homozygous and heterozygous genotypes.
Experimental Protocol
The rats were maintained at the animal facilities of the Mayo Clinic. In some rats, osmotic minipumps (model 2ML4; Alzet, Palo Alto, CA) delivering vehicle alone or dDAVP at a rate of 10 ng/h per 100 g body wt were implanted subcutaneously through a small dorsal incision at 12 wk of age and replaced 4 wk later under anesthesia (ketamine 50 mg/kg and xylazine 10 mg/kg intraperitoneally). Twenty-four-hour urine outputs in metabolic cages and tail-cuff BP were obtained weekly on three consecutive weeks before rats were killed at 10 or 20 wk of age. After being killed, the rat were weighed and anesthetized with ketamine 50 mg/kg and xylazine 10 mg/kg, intraperitoneally. Blood was obtained by cardiac puncture for determination of serum electrolytes and blood urea nitrogen levels. The right kidney and the liver were placed into preweighed vials containing 10% formaldehyde in phosphate buffer (pH 7.4). The tissues were embedded in paraffin for histologic studies. The left kidneys were immediately frozen in liquid nitrogen for determination of cAMP.3–5
Histomorphometric Analysis
Four-micron transverse tissue sections, including cortex, medulla, and papilla, were stained with hematoxylin-eosin to measure cyst volumes and with picrosirius red stain for collagen to measure fibrosis. Image analysis procedures were performed with Meta-Morph software (Universal Imaging, West Chester, PA).3–5
cAMP Content of Whole Kidneys
The kidneys were ground to fine powder under liquid nitrogen in a stainless steel mortar and homogenized in 10 volumes of cold 5% TCA in a glass-Teflon tissue grinder. After centrifugation at 600 × g for 10 min, the supernatants were extracted with 3 Vol of water-saturated ether. After drying the aqueous extracts, the reconstituted samples were processed without acetylation using an enzyme immunoassay kit (Sigma-Aldrich, St. Louis, MO). The results were expressed in picomoles per milligram of protein.3–5
Western Blot Analysis
Total ERK1/2 and phosphorylated ERK1/2 were measured by Western blot analysis using anti-ERK1/2 (C16) and anti-phosphorylated ERK1/2 (E4) antibodies (Santa Cruz Biotechnology, Santa Cruz, CA).
Statistical Analyses
Comparisons between groups were made using one-way or two-way ANOVA with least significant difference comparisons of the means or t test as appropriate. Data are expressed as means ± SD.
DISCLOSURES
None.
Supplementary Material
Acknowledgments
This work was supported by grants from the PKD Foundation and by National Institutes of Health grant DK44863 (V.E.T.).
Published online ahead of print. Publication date available at www.jasn.org.
Supplemental information for this article is available online at http://www.jasn.org/.
See related editorial, “Therapy for Polycystic Kidney Disease? It's Water, Stupid!” on pages 1–2.
REFERENCES
- 1.Torres VE, Harris PC, Pirson Y: Autosomal dominant polycystic kidney disease. Lancet 369: 1287–1301, 2007 [DOI] [PubMed] [Google Scholar]
- 2.Torres VE, Harris PC: Mechanisms of disease: Autosomal dominant and recessive polycystic kidney diseases. Nat Clin Pract Nephrol 2: 40–54, 2006 [DOI] [PubMed] [Google Scholar]
- 3.Gattone VH, Wang X, Harris PC, Torres VE: Inhibition of renal cystic disease development and progression by a vasopressin V2 receptor antagonist. Nat Med 9: 1323–1326, 2003 [DOI] [PubMed] [Google Scholar]
- 4.Torres VE, Wang X, Qian Q, Somlo S, Harris PC, Gattone VH: Effective treatment of an orthologous model of autosomal dominant polycystic kidney disease. Nat Med 10: 363–364, 2004 [DOI] [PubMed] [Google Scholar]
- 5.Wang X, Gattone VH II, Harris PC, Torres VE: Effectiveness of vasopressin V2 receptor antagonists OPC-31260 and OPC-41061 on polycystic kidney disease development in the PCK rat. J Am Soc Nephrol 16: 846–851, 2005 [DOI] [PubMed] [Google Scholar]
- 6.Ward CJ, Hogan MC, Rossetti S, Walker D, Sneddon T, Wang X, Kubly V, Cunningham JM, Bacallao R, Ishibashi M, Milliner DS, Torres VE, Harris PC: The gene mutated in autosomal recessive polycystic kidney disease encodes a large, receptor-like protein. Nat Genet 30: 259–269, 2002 [DOI] [PubMed] [Google Scholar]
- 7.Schmale H, Richter D: Single base deletion in the vasopressin gene is the cause of diabetes insipidus in Brattleboro rats. Nature 308: 705–709, 1984 [DOI] [PubMed] [Google Scholar]
- 8.Nagao S, Kazuhiro N, Katsuyama M, Kurahashi H, Marunouchi T, Takahashi H, Wallace DP: Increased water intake decreases progression of polycystic kidney disease in the PCK rat. J Am Soc Nephrol 17: 228–235, 2006 [DOI] [PubMed] [Google Scholar]
- 9.Sladek CD, McNeill TH: The Brattleboro heterozygote rat as a model for neurohypophyseal aging: Vasopressin response to dehydration. Ann N Y Acad Sci 394: 173–176, 1982 [DOI] [PubMed] [Google Scholar]
- 10.Bouby N, Hassler C, Bankir L: Contribution of vasopressin to progression of chronic renal failure: Study in Brattleboro rats. Life Sci 65: 991–1004, 1999 [DOI] [PubMed] [Google Scholar]
- 11.Bouby N, Bankir L, Trinh-Trang-Tan MM, Minuth WW, Kriz W: Selective ADH-induced hypertrophy of the medullary thick ascending limb in Brattleboro rats. Kidney Int 28: 456–466, 1985 [DOI] [PubMed] [Google Scholar]
- 12.Bankir L, Fischer C, Fischer S, Jukkala K, Specht HC, Kriz W: Adaptation of the rat kidney to altered water intake and urine concentration. Pflugers Arch 412: 42–53, 1988 [DOI] [PubMed] [Google Scholar]
- 13.Seeman T, Dusek J, Vondrak K, Blahova K, Simkova E, Kreisinger J, Dvorak P, Kyncl M, Hribal Z, Janda J: Renal concentrating capacity is linked to blood pressure in children with autosomal dominant polycystic kidney disease. Physiol Res 53: 629–634, 2004 [PubMed] [Google Scholar]
- 14.Lager DJ, Qian Q, Bengal RJ, Ishibashi M, Torres VE: The pck rat: A new model that resembles human autosomal dominant polycystic kidney and liver disease. Kidney Int 59: 126–136, 2001 [DOI] [PubMed] [Google Scholar]
- 15.Ota M, Crofton JT, Liu H, Festavan G, Share L: Increased plasma osmolality stimulates peripheral and central vasopressin release in male and female rats. Am J Physiol 267: R923–R928, 1994 [DOI] [PubMed] [Google Scholar]
- 16.Wang YX, Crofton JT, Liu H, Sato K, Brooks DP, Share L: Estradiol attenuates the antidiuretic action of vasopressin in ovariectomized rats. Am J Physiol 268: R951–R957, 1995 [DOI] [PubMed] [Google Scholar]
- 17.Wang YX, Edwards RM, Nambi P, Stack EJ, Pullen M, Share L, Crofton JT, Brooks DP: Sex difference in the antidiuretic activity of vasopressin in the rat. Am J Physiol 265: R1284–R1290, 1993 [DOI] [PubMed] [Google Scholar]
- 18.Pettinger WA, Fallet R, Wang Y, Tam LT, Jeffries WB: Enhanced cAMP response to vasopressin in the CCT of DOCA-Na hypertensive rats. Am J Physiol 251: F1096–F1100, 1986 [DOI] [PubMed] [Google Scholar]
- 19.Ouchi Y, Share L, Crofton JT, Iitake K, Brooks DP: Sex difference in the development of deoxycorticosterone-salt hypertension in the rat. Hypertension 9: 172–177, 1987 [DOI] [PubMed] [Google Scholar]
- 20.Hatano T, Ogawa K, Kanda K, Seo H, Matsui N: Effect of ovarian steroids on cyclic adenosine 3′:5′-monophosphate production stimulated by arginine vasopressin in rat renal monolayer cultured cells. Endocrinol Jpn 35: 267–274, 1988 [DOI] [PubMed] [Google Scholar]
- 21.Wang YX, Crofton JT, Miller J, Sigman CJ, Liu H, Huber JM, Brooks DP, Share L: Sex difference in urinary concentrating ability of rats with water deprivation. Am J Physiol 270: R550–R555, 1996 [DOI] [PubMed] [Google Scholar]
- 22.Stachenfeld NS, Splenser AE, Calzone WL, Taylor MP, Keefe DL: Sex differences in osmotic regulation of AVP and renal sodium handling. J Appl Physiol 91: 1893–1901, 2001 [DOI] [PubMed] [Google Scholar]
- 23.Crofton JT, Dustan H, Share L, Brooks DP: Vasopressin secretion in normotensive black and white men and women on normal and low sodium diets. J Endocrinol 108: 191–199, 1986 [DOI] [PubMed] [Google Scholar]
- 24.Share L, Crofton JT, Ouchi Y: Vasopressin: Sexual dimorphism in secretion, cardiovascular actions and hypertension. Am J Med Sci 295: 314–319, 1988 [DOI] [PubMed] [Google Scholar]
- 25.Bakris G, Bursztyn M, Gavras I, Bresnahan M, Gavras H: Role of vasopressin in essential hypertension: racial differences. J Hypertens 15: 545–550, 1997 [DOI] [PubMed] [Google Scholar]
- 26.Perucca J, Bouby N, Valeix P, Bankir L: Sex difference in urine concentration across differing ages, sodium intake, and level of kidney disease. Am J Physiol Regul Integr Comp Physiol 292: R700–R705, 2007 [DOI] [PubMed] [Google Scholar]
- 27.Stachenfeld NS, Taylor HS, Leone CA, Keefe DL: Oestrogen effects on urine concentrating response in young women. J Physiol 552: 869–880, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Harris PC, Bae KT, Rossetti S, Torres VE, Grantham JJ, Chapman AB, Guay-Woodford LM, King BF, Wetzel LH, Baumgarten DA, Kenney PJ, Consugar M, Klahr S, Bennett WM, Meyers CM, Zhang QJ, Thompson PA, Zhu F, Miller JP: Cyst number but not the rate of cystic growth is associated with the mutated gene in autosomal dominant polycystic kidney disease. J Am Soc Nephrol 17: 3013–3019, 2006 [DOI] [PubMed] [Google Scholar]
- 29.Gabow P, Johnson A, Kaehny W, Kimberling W, Lezotte D, Duley I, Jones R: Factors affecting the progression of renal disease in autosomal-dominant polycystic kidney disease. Kidney Int 41: 1311–1319, 1992 [DOI] [PubMed] [Google Scholar]
- 30.Magistroni R, He N, Wang K, Andrew R, Johnson A, Gabow P, Dicks E, Parfrey P, Torra R, San-Millan JL, Coto E, Van Dijk M, Breuning M, Peters D, Bogdanova N, Ligabue G, Albertazzi A, Hateboer N, Demetriou K, Pierides A, Deltas C, St George-Hyslop P, Ravine D, Pei Y: Genotype-renal function correlation in type 2 autosomal dominant polycystic kidney disease. J Am Soc Nephrol 14: 1164–1174, 2003 [DOI] [PubMed] [Google Scholar]
- 31.Hateboer N, van Dijk MA, Bogdanova N, Coto E, Saggar-Malik AK, San Millan JL, Torra R, Breuning M, Ravine D: Comparison of phenotypes of polycystic kidney disease types 1 and 2. Lancet 353: 103–107, 1999 [DOI] [PubMed] [Google Scholar]
- 32.Rossetti S, Chauveau D, Kubly V, Slezak JM, Saggar-Malik AK, Pei Y, Ong AC, Stewart F, Watson ML, Bergstralh EJ, Winearls CG, Torres VE, Harris PC: Association of mutation position in polycystic kidney disease 1 (PKD1) gene and development of a vascular phenotype. Lancet 361: 2196–2201, 2003 [DOI] [PubMed] [Google Scholar]
- 33.Takei Y, Kawakoshi A, Tsukada T, Yuge S, Ogoshi M, Inoue K, Hyodo S, Bannai H, Miyano S: Contribution of comparative fish studies to general endocrinology: Structure and function of some osmoregulatory hormones. J Exp Zoolog A Comp Exp Biol 305: 787–798, 2006 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.