Abstract
Fluoroquinolone- and methicillin-resistant Staphylococcus pseudintermedius isolates harbor two new staphylococcal cassette chromosome mec (SCCmec) elements that belong to class A, allotype 3 (SCCmec II-III), and to the new allotype 5 (SCCmec VII). Analysis of the complete nucleotide sequences of the topoisomerase loci gyrB/gyrA and grlB/grlA revealed mutations involved in fluoroquinolone resistance.
Staphylococcus pseudintermedius is an opportunistic pathogen that primarily causes skin and nosocomial infections in dogs and cats but can also occasionally affect humans (1, 29). Fluoroquinolones and cephalosporins are widely used to treat staphylococcal infections in veterinary medicine (23). The frequent use of these antibiotics may augment the risk of rapidly selecting for bacteria resistant to both classes of antibiotics. In the past year, multidrug-resistant S. pseudintermedius strains have been isolated with increasing frequency from infection sites of dogs at our diagnostic unit. The antibiotic resistance mechanisms of these S. pseudintermedius strains were characterized with an emphasis on resistance to methicillin and fluoroquinolone.
Strain identification and antibiotic resistance profile.
The isolated strains (n = 15) were cultured on tryptone soy agar plates containing 5% sheep blood (Oxoid Ltd., Basingstoke, England) and were identified by catalase activity, Gram staining, and sequencing of the sodA gene as described previously (24, 26). All isolates harbored the leukocidin gene lukS (25) but not the Panton-Valentine leukocidin gene lukS-PV (28), as determined by PCR using the primers listed in Table 2. Antibiotic susceptibility was determined by broth microdilution using a custom Sensititre susceptibility plate, model NLV57 (Trek Diagnostics Systems, East Grinstead, England; MCS Diagnostics BV, Swalmen, The Netherlands), according to CLSI (formerly NCCLS) guidelines (4). Resistance genes were detected using a microarray (22) (Table 1). In addition to fluoroquinolone and oxacillin (methicillin) resistance, the isolates also displayed resistance to other antibiotics (Table 1).
TABLE 2.
Oligonucleotides used for PCR amplification of leukocidin genes, SCCmec, topoisomerase IV, and gyrase genes
| Gene | Primer name | Sequence | Primer design reference or source |
|---|---|---|---|
| Gyrase and topoisomerase IV | |||
| grlA | grlA-cons-F | GGTNCGTTTAAGTCAAGAa | This study |
| grlA-cons-R | CCTTCNACNATATGCATNCGa | ||
| grlB | grlB-sa-F2 | TATNCGNGAAGGTTTAACa | This study |
| grlB-cons-R | GTTTCNGGNTTCATNGTNGTTTCCCAa | ||
| grlB-grlA | grlB-si-PF | GCTATTTAGATTAGGCTTGC | This study |
| grlA-si-R0 | TGTGACCAAATCAAGTCG | ||
| gyrA | gyrA-cons-F | ACNGATTTACGTGATGAAACa | This study |
| gyrA-cons-R | ACGTCTNAAACGCATNTCTAAAa | ||
| gyrB | gyrB-sa-F4 | GTTGATATTCAAGAAAANATGGGa | This study |
| gyrB-sa-R2 | AGACCCCCCGGCAGAGTCa | ||
| gyrB-gyrA | gyrB-si-PF0 | GATGACGTCTTAAGTGAGTTGG | This study |
| gyrA-si-RV | ATTGGCGATAAGTTGTCAAAGG | ||
| SCCmec | |||
| ccrA | ccrA-F | AACGTGTCATTGCSACAC | This study |
| ccrA-R | GGGCGTAAGATTTATCAAGCTT | ||
| mecI | mecI-F | CCCTTTTTATACAATCTCGTT | 31 |
| mecI-R | ATATCATCTGCAGAATGGG | ||
| orfX | ORFX1r | AACGTTTAGGCCCATACACCA | 9 |
| IR-L SRImec III | SccmecIR-F | CTTATCAGTTGATGATGCG | This study |
| lukS | lukSi-F1 | ATGGTAAAAAATAAATTATTAGCCG | This study |
| lukSi-R0 | TTAATTATGCCCCTTTACTTTAATTTC | ||
| lukS-PV | luk-PV-1 | ATCATTAGGTAAAATGTCTGGACATGATCCA | 17 |
| luk-PV-2 | GCATCAASTGTATTGGATAGCAAAAGC |
Primers designed from conserved regions found after alignment of DNA sequences of grlB/grlA and gyrB/gyrA from S. aureus (GenBank accession no. L25288 and BA000018), S. epidermidis (GenBank accession no. CP000029 and AE015929), and S. haemolyticus (GenBank accession no. AY341071, AY341072, AY341073, and AY341074).
TABLE 1.
Antibiotic resistance profiles, lukS gene, SCCmec type, and clonal relatedness of 15 Staphylococcus pseudintermedius isolates from dogs and of S. pseudintermedius type strain CCUG49543T
| Strain | Source | MIC (μg/ml) for the following antibiotica (resistance breakpointb)
|
Antibiotic resistance genes detectedc | Leukocidin | SCCmec type | gyr/grl group | ST | agr type | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AMK (≥64) | AMX (≥8/4) | CEF (≥32) | CHL (≥32) | ENRO (≥4) | CLI (≥4) | ERY (≥8) | GEN (≥16) | KAN (≥64) | LZD (−) | NIT (≥128) | OXA (≥4) | PEN (≥0.25) | STR (≥32) | SXT (≥4/76) | Q/D (≥4) | TET (≥16) | VAN (≥16) | ||||||||
| CCUG49543T (LMG22219T) | Type strain | ≤16 | ≤2/1 | ≤2 | 8 | ≤0.25 | >8 | >16 | ≤2 | ≤1,000 | ≤0.5 | ≤16 | 0.5 | 0.25 | >32 | ≤0.5/9.5 | ≤0.5 | 32 | ≤1 | ant(6′)-Ia, aph(3′)-III, blaZ, erm(B), sat4, tet(M) | lukS | ST63 | IV | ||
| KM241 | Otitis externa | ≤16 | ≤2/1 | ≤2 | ≤4 | 4 | ≤0.5 | ≤0.25 | ≤2 | ≤1,000 | ≤0.5 | ≤16 | 16 | ≤0.12 | ≤4 | ≤0.5/9.5 | ≤0.5 | 32 | ≤1 | mecA, tet(M) | lukS | VII | 1 | ST73 | II |
| KM336 | Osteitis after surgery | ≤16 | ≤2/1 | ≤2 | 32 | 16 | >8 | >16 | 4 | ≤1,000 | ≤0.5 | ≤16 | 16 | 2 | >32 | 4/76 | ≤0.5 | ≤1 | ≤1 | aac(6′)-Ie, ant(6′)-Ia, aph(3′)-III, blaZ, catpC221, erm(B),mecA, sat4, dfr(G) | lukS | II-III | 5 | ST71 | III |
| KM337 | Otitis externa | ≤16 | ≤2/1 | ≤2 | ≤4 | ≤0.25 | ≤0.5 | ≤0.25 | ≤2 | ≤1,000 | ≤0.5 | ≤16 | ≤0.25 | ≤0.12 | ≤4 | ≤0.5/9.5 | ≤0.5 | ≤1 | ≤1 | lukS | ST74 | II | |||
| KM571 | Infected wound after surgery | ≤16 | 8/4 | ≤2 | 8 | >16 | >8 | >16 | 16 | ≤1,000 | 2 | ≤16 | >16 | >8 | >32 | 8/152 | 1 | >32 | 2 | aac(6′)-Ie, ant(6′)-Ia, aph(3′)-III, blaZ, erm(B), mecA, sat4, tet(K), dfr(G) | lukS | II-III | 5 | ST71 | III |
| KM631 | Osteitis after surgery | ≤16 | 4/2 | 8 | 64 | 16 | >8 | >16 | 32 | ≤1,000 | ≤0.5 | ≤16 | >16 | >8 | >32 | 8/152 | 1 | >32 | ≤1 | ant(6′)-Ia, aph(3′)-III, blaZ, catpC221, erm(B), mecA, sat4, tet(K), dfr(G) | lukS | II-III | 5 | ST71 | III |
| KM1087 | Vaginitis | ≤16 | ≤2/1 | ≤2 | ≤4 | ≤0.25 | ≤0.5 | ≤0.25 | ≤2 | ≤1,000 | ≤0.5 | ≤16 | ≤0.25 | ≤0.12 | ≤4 | ≤0.5/9.5 | ≤0.5 | 32 | ≤1 | blaZ, tet(M) | lukS | ST75 | I | ||
| KM1250 | Infected wound | ≤16 | ≤2/1 | ≤2 | 8 | ≤0.25 | ≤0.5 | ≤0.25 | ≤2 | ≤1,000 | 1 | ≤16 | ≤0.25 | ≤0.12 | ≤4 | ≤0.5/9.5 | ≤0.5 | ≤1 | ≤1 | lukS | ST41 | II | |||
| KM1381 | Fistula after surgery | ≤16 | ≤2/1 | ≤2 | 64 | 16 | >8 | >16 | 16 | ≤1,000 | ≤0.5 | ≤16 | >16 | ≤0.12 | >32 | 4/76 | ≤0.5 | >32 | ≤1 | aph(3′)-III, aph(2′)-Ia, catpC221, erm(B), mecA, sat4, tet(K), dfr(G) | lukS | II-III | 5 | ST71 | III |
| KM1395 | Otitis externa | ≤16 | ≤2/1 | ≤2 | 8 | >16 | >8 | >16 | 32 | ≤1,000 | 1 | ≤16 | >16 | ≤0.12 | >32 | 8/152 | 1 | >32 | ≤1 | aac(6′)-Ie, ant(6′)-Ia, aph(3′)-III, blaZ, erm(B), mecA, sat4, tet(K), dfr(G) | lukS | II-III | 4 | ST71 | III |
| KM1542 | Seroma | ≤16 | 4/2 | ≤2 | 64 | >16 | >8 | >16 | 500 | ≤1,000 | 1 | ≤16 | 2 | 0.5 | >32 | 8/152 | 1 | >32 | ≤1 | aac(6′)-Ie, ant(6′)-Ia, aph(3′)-III, blaZ, catpC221, erm(B), mecA, sat4, tet(K), dfr(G) | lukS | II-III | 5 | ST71 | III |
| KM1591 | Pyoderma | ≤16 | ≤2/1 | ≤2 | ≤4 | 8 | >8 | >16 | ≤2 | ≤1,000 | ≤0.5 | ≤16 | ≤0.25 | ≤0.12 | >32 | ≤0.5/9.5 | ≤0.5 | 32 | ≤1 | ant(6′)-Ia, aph(3′)-III, erm(B), sat4, tet(M) | lukS | 2 | ST51 | I | |
| KM1832 | Osteitis after surgery | ≤16 | ≤2/1 | ≤2 | 32 | 16 | >8 | >16 | 16 | ≤1,000 | ≤0.5 | ≤16 | >16 | 0.25 | >32 | 4/76 | ≤0.5 | >32 | ≤1 | aac(6′)-Ie, ant(6′)-Ia, aph(3′)-III, blaZ, catpC221, erm(B), mecA, sat4, tet(K), dfr(G) | lukS | II-III | 5 | ST71 | III |
| KM1896 | Gingivitis | ≤16 | ≤2/1 | ≤2 | 32 | 16 | >8 | >16 | 16 | ≤1,000 | ≤0.5 | ≤16 | >16 | ≤0.12 | >32 | 4/76 | 1 | >32 | ≤1 | ant(6′)-Ia, aph(3′)-III, catpC221, erm(B), mecA, sat4, tet(K), dfr(G) | lukS | II-III | 5 | ST71 | III |
| SD91 | Ear | ≤16 | ≤2/1 | ≤2 | 64 | 16 | >8 | >16 | 500 | ≤1,000 | ≤0.5 | ≤16 | 4 | >8 | >32 | 4/76 | ≤0.5 | >32 | ≤1 | ant(6′)-Ia, aph(3′)-III, blaZ, catpC221, erm(B), mecA, sat4, tet(K), dfr(G) | lukS | II-III | 5 | ST71 | III |
| SD1071 | Nasal cavities | ≤16 | ≤2/1 | ≤2 | ≤4 | 8 | 2d | >16 | ≤2 | ≤1,000 | 1 | ≤16 | ≤0.25 | ≤0.12 | >32 | ≤0.5/9.5 | ≤0.5 | 32 | ≤1 | ant(6′)-Ia, aph(3′)-III, blaZ, erm(B), sat4, tet(M) | lukS | 3 | ST76 | IV | |
AMK, amikacin; AMX, amoxicillin; CEF, cephalotin; CHL, chloramphenicol; CLI, clindamycin; ENRO, enrofloxacin; ERY, erythromycin; GEN, gentamicin; KAN, kanamycin; LZD, linezolid; NIT, nitrofurantoin; OXA, oxacillin; PEN, penicillin; STR, streptomycin; SXT, trimethoprim-sulfamethoxazole; Q/D, quinupristin-dalfopristin; TET, tetracycline; VAN, vancomycin.
The MIC breakpoints (in micrograms per milliliter) determining resistance were those recommended for S. aureus in CLSI supplement M100-S16 (6) except for enrofloxacin and streptomycin, for which breakpoints of CLSI from supplement M31-S1 for bacteria from animals (3) and from the French Society for Microbiology (www.sfm.asso.fr) were used. MICs above resistance breakpoints are boldfaced.
Antibiotic resistance genes and their functions are as follows: erm(B), macrolides, lincosamides, and streptogramin B methylase; blaZ, β-lactamase; catpC221, chloramphenicol acetyltransferase; dfr(G), dihydrofolate reductase (trimethoprim resistance); tet(K), tetracycline efflux; aac(6′)-Ie and aph(3′)-III, aminoglycoside acetyltransferase and phosphotransferase; ant(6′)-Ia, streptomycin adenylyltransferase; sat4, streptothricin acetyltransferase; mecA, penicillin-binding protein PBP2′.
Inducible clindamycin resistance as determined according to CLSI guidelines (5).
Characterization of mutations in topoisomerase genes.
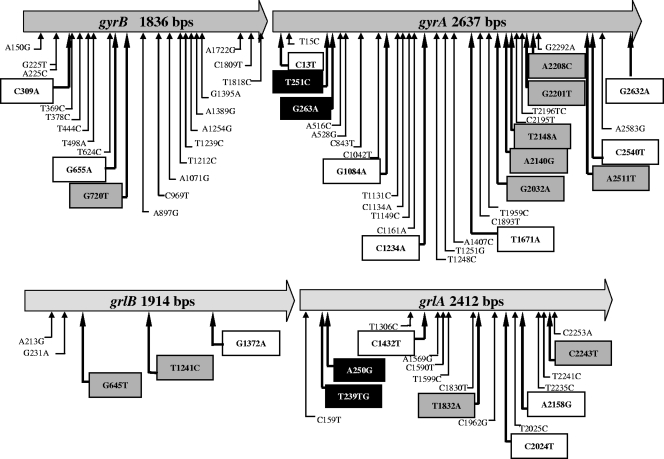
The mechanism of resistance to fluoroquinolones was investigated by sequence analysis of the topoisomerase II (gyrA and gyrB) and IV (grlA and grlB) genes, since mutations in these genes have been shown to confer resistance to fluoroquinolones on Staphylococcus aureus (8, 11, 13, 14, 18, 27, 30). Fragments of gyrA, gyrB, grlA, and grlB of type strains S. pseudintermedius CCUG49543T (also known as LMG 22219T [10]) (CCUG, Culture Collection, University of Göteborg, Göteborg, Sweden) and Staphylococcus intermedius DSM20373T (also known as NCTC 11048T) (DSMZ, German Collection of Microorganisms and Cell Cultures, Braunschweig, Germany) were amplified by PCR and sequenced on an ABI Prism 3100 genetic analyzer (Applied Biosystems, Foster City, CA) using oligonucleotide primers (Table 2) designed from conserved regions found after alignment of DNA sequences of grlB/grlA and gyrB/gyrA from S. aureus (GenBank accession no. L25288 and BA000018), Staphylococcus epidermidis (GenBank accession no. CP000029 and AE015929), and Staphylococcus haemolyticus (GenBank accession no. AY341071, AY341072, AY341073, and AY341074) using MultAlin (7). The nucleotide sequences flanking the amplified fragments were determined by genome walking using the Universal Vectorette system according to the manufacturer's protocol (Sigma-Genosys, St. Louis, MO) and genomic DNA digested with Sau3A, EcoRI, HindIII, and ApoI. The nucleotide sequences of the grlB/grlA and gyrB/gyrA loci of S. pseudintermedius CCUG49543 share 89% and 94% identity, respectively, with those of S. intermedius DSM20373. The entire gyrB/gyrA and grlB/grlA loci of fluoroquinolone-resistant and fluoroquinolone-susceptible S. pseudintermedius strains were then amplified using the Expand Long Template PCR system (Roche Applied Science, Indianapolis, IN) (annealing at 54°C for 30 s and extension at 68°C for 5 min) with specific primer pairs gyrB-si-PF0-gyrA-si-RV and grlB-si-PF-grlA-si-R0 and were sequenced. Nucleotide sequence comparison revealed high levels of nucleotide polymorphism in the gyrase and topoisomerase IV genes. Most of the mutations are silent (Fig. 1). Mutations that cause amino acid substitutions in the topoisomerases were found in both resistant and susceptible strains and cannot, therefore, be responsible for resistance. However, other amino acid mutations were found only in fluoroquinolone-resistant strains (Table 3). Some of these amino acid changes occurred at the same positions as those reported for fluoroquinolone-resistant S. aureus, S. intermedius, and Staphylococcus schleiferi strains, including positions 251 (Ser84Leu) and 263 (Glu88Gly) on gyrA and positions 239 (Ser80Ile) and 250 (Asp84Asn) on grlA (8, 13-15, 18, 27, 30) (Table 3). S. pseudintermedius strains harboring at least one of these mutations showed decreased susceptibility to enrofloxacin (MICs, 4 to 8 μg/ml). Higher MICs (≥16 μg/ml) were observed when two additional mutations (Thr678Ala and Glu714Lys) were present in gyrA (Table 3). Additional amino acid substitutions were found in the topoisomerase genes, but their roles in fluoroquinolone resistance remain to be determined (Fig. 1).
FIG. 1.
Consensus nucleotide mutations present in the topoisomerase IV and gyrase genes of 12 fluoroquinolone-resistant and 4 fluoroquinolone-susceptible strains. Silent mutations (not boxed) are indicated with thin arrows. Open boxes, mutations that cause amino acid substitutions in both fluoroquinolone-resistant and fluoroquinolone-susceptible strains; solid boxes, mutations that cause amino acid substitutions at the same positions as those described for fluoroquinolone-resistant S. aureus; shaded boxes, additional mutations that cause amino acid substitutions in fluoroquinolone-resistant S. pseudintermedius.
TABLE 3.
Nucleotide mutations that cause amino acid substitutions in gyrase and topoisomerase IV in fluoroquinolone-resistant S. pseudintermedius
| Strain | Presence or absence of the indicated mutation in the following gene:
|
MIC (μg/ml) for enrofloxacin | gyr/grl group | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
grlA
|
grlB
|
gyrA
|
gyrB T720G (Phe240Leu) | |||||||||||||
| G239T (Ser80Ile)a | G250A (Asp84Asn)a | A1832T (Gln611Leu) | T2243C (Val748Ala) | T645G (Asp215Glu) | T1241C (Leu414Ser) | C251T (Ser84Leu)a | A263G (Glu88Gly)a | A2032G (Thr678Ala) | G2140A (Glu714Lys) | T2201G (Leu734Arg) | C2208A (Asn736Lys) | T2511A (Asp837Glu) | ||||
| Fluoroquinolone susceptible | ||||||||||||||||
| CCUG 49543T | − | − | − | − | − | − | − | − | − | − | − | − | − | − | ≤0.25 | − |
| KM337 | − | − | − | − | − | − | − | − | − | − | − | − | − | − | ≤0.25 | − |
| KM1087 | − | − | − | − | − | − | − | − | − | − | − | − | − | − | ≤0.25 | − |
| KM1250 | − | − | − | − | − | − | − | − | − | − | − | − | − | − | ≤0.25 | − |
| Fluoroquinolone resistant | ||||||||||||||||
| KM241 | + | − | + | − | − | − | − | + | − | − | − | − | + | + | 4 | 1 |
| KM1591 | − | + | − | − | − | + | + | − | − | − | − | − | − | − | 8 | 2 |
| SD1071 | + | − | − | + | − | − | + | − | − | − | + | + | − | − | 8 | 3 |
| KM1395 | + | − | − | − | + | − | + | − | + | + | − | − | − | − | >16 | 4 |
| SD91 | + | − | − | − | − | − | + | − | + | + | − | − | − | − | 16 | 5 |
| KM1381 | + | − | − | − | − | − | + | − | + | + | − | − | − | − | 16 | 5 |
| KM631 | + | − | − | − | − | − | + | − | + | + | − | − | − | − | 16 | 5 |
| KM1832 | + | − | − | − | − | − | + | − | + | + | − | − | − | − | 16 | 5 |
| KM1896 | + | − | − | − | − | − | + | − | + | + | − | − | − | − | 16 | 5 |
| KM336 | + | − | − | − | − | − | + | − | + | + | − | − | − | − | 16 | 5 |
| KM571 | + | − | − | − | − | − | + | − | + | + | − | − | − | − | >16 | 5 |
| KM1542 | + | − | − | − | − | − | + | − | + | + | − | − | − | − | >16 | 5 |
Characterization of two new SCCmec elements.
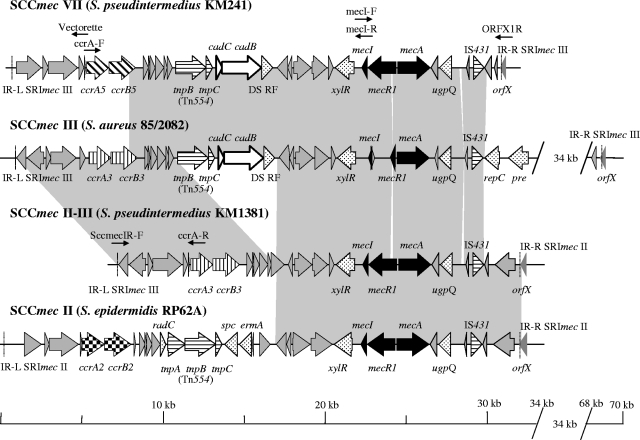
Methicillin resistance is mediated by the mecA gene, which is located on a large staphylococcal cassette chromosome mec (SCCmec) element. SCCmec elements are characterized by the completeness of the methicillin resistance regulon containing mecA, by the allotype of the recombinase genes ccrA and ccrB, and by the general genetic structure (2, 12, 20). SCCmec are delineated by two inverted repeats, IR-L and IR-R (Fig. 2). Six different SCCmec (I to VI) have been described to date for Staphylococcus (2, 12, 16, 20, 21). In the course of our study, SCCmec of S. pseudintermedius could not be classified using PCR methods previously developed to determine the SCCmec class (19, 31) and were therefore sequenced. First, the regions spanning orfX to mecI and mecI to ccrA of all methicillin-resistant S. pseudintermedius strains were amplified using the Expand Long Template PCR system (Roche Applied Science, Indianapolis, IN) with primers ccrA4-F1 and mecI-R (fragment A) and mecI-F and ORFX1r (9) (fragment B) (annealing at 50°C for 30 s; extension at 68°C for 15 min) (Table 2; Fig. 2). Restriction analysis of fragments A and B, digested with HindIII and PstI, respectively, revealed two types of SCCmec. The SCCmec of KM241 had a unique profile, whereas all the other profiles were identical to that of KM1381. The fragments were sequenced using a primer-walking strategy. To complete the entire nucleotide sequence of the cassette, the 5′ end of the SCCmec of KM1381 situated upstream of ccrA was amplified using primer ccrA-R and primer SccmecIR-F, which is specific to S. aureus SCCmec III IR-L (GenBank accession no. AB037671). The 5′-end sequence of the SCCmec of KM241 was determined using the Universal Vectorette system (Sigma-Genosys Co., St. Louis, MO). Sequence analysis of the two entire cassettes revealed two new SCCmec, SCCmec II-III in KM1381 and SCCmec VII in KM241 (Fig. 2). SCCmec II-III consists of a combination of S. aureus SCCmec III (accession no. AB037671) (100% nucleotide identity from IR-L to ORF12) and S. epidermidis SCCmec II (GenBank accession no. CP000029) (98.9% nucleotide identity from ORF13 to IR-R) (Fig. 2). SCCmec VII contains new recombinase genes, ccrA5 and ccrB5, classifying it as a new allotype, allotype 5. The amino acid sequences of ccrA5 and ccrB5 showed 75.6% identity overall to S. aureus CcrA, allotype 3 (GenBank accession no. BAA88754), and 92.3% identity overall to S. aureus CcrB, allotype 3 (GenBank accession no. BAA88755). The rest of the SCCmec VII downstream of the ccrA5 and ccrB5 loci until IS431 showed 99% nucleotide identity to S. aureus SCCmec III (GenBank accession no. AB037671) (Fig. 2) but differed from SCCmec III by a complete mecA regulon. Both SCCmec II-III and SCCmec VII are new cassettes, which belong to class A, allotype 3, and class A, allotype 5, respectively.
FIG. 2.
Alignment of S. pseudintermedius SCCmec II-III and SCCmec VII of with S. aureus SCCmec III and S. epidermidis SCCmec II. Solid arrows represent the mecA regulon. Diagonally lined, vertically lined, and checked arrows represent the different cassette chromosome recombinases A and B. Horizontally lined arrows, transposases; open arrows, cadmium resistance genes; shaded arrows, hypothetical proteins. Cassettes are delimited by inverted repeats IR-L and IR-R (dashed lines).
Differentiation of strains by ST.
Sequence typing (ST) of five gene loci (the 16S rRNA gene, tuf, cpn60, pta, and agrD) and examination of the allelic variation of agrD (1) showed that all methicillin-resistant strains containing SCCmec II-III were clonally related. They belong to ST71, agr type III (Table 1), which is the predominant clonal group in North and Central Europe (1). They also contain the same mutations in the gyrB/gyrA and grlB/grlA genes (gyr/grl group 5), except for strain KM1395. S. pseudintermedius strain KM241 (SCCmec VII) belongs to a new group (ST73, agr IV). The methicillin- and fluoroquinolone-susceptible strains belong to the distinct ST group ST41, agr II, and to new ST groups ST74, agr II, and ST75, agr I (Table 1).
New SCCmec with new recombinase genes in S. pseudintermedius represent a new reservoir of the mecA gene for methicillin-sensitive Staphylococcus species. Until now, mainly SCCmec III has been detected in S. pseudintermedius (26). Additionally, sequence analysis of topoisomerase genes has allowed us to determine, for the first time for S. pseudintermedius, mutations that play a role in fluoroquinolone resistance.
The presence of multidrug-resistant Staphylococcus of animal origin is a further demonstration that the use of antibiotics in veterinary medicine selects for resistant strains. Guidelines regarding the use and choice of antibiotics should be followed in veterinary medicine to suppress the rapid, nationwide dissemination of multidrug-resistant S. pseudintermedius clones.
Nucleotide sequence accession numbers.
Nucleotide sequences were deposited in the EMBL/GenBank/DDBJ databases. SCCmec II-III and SCCmec VII were assigned accession no. AM904732 and AM904731. The gyrB/gyrA and grlB/grlA loci of S. pseudintermedius CCUG49543T and KM1381 and of S. intermedius DSM20373T were assigned accession no. AM262968 and AM262971, AM262969 and AM262972, and AM262967 and AM262970, respectively.
Acknowledgments
We thank the personnel of the diagnostic unit of the Institute of Veterinary Bacteriology of the University of Bern who isolated the strains. We also thank J. Ross Fitzgerald for helpful advice on sequence typing.
Footnotes
Published ahead of print on 27 February 2008.
REFERENCES
- 1.Bannoehr, J., N. L. Ben Zakour, A. S. Waller, L. Guardabassi, K. L. Thoday, A. H. M. van den Broek, and J. R. Fitzgerald. 2007. Population genetic structure of the Staphylococcus intermedius group: insights into agr diversification and the emergence of methicillin-resistant strains. J. Bacteriol. 1898685-8692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chongtrakool, P., T. Ito, X. X. Ma, Y. Kondo, S. Trakulsomboon, C. Tiensasitorn, M. Jamklang, T. Chavalit, J. H. Song, and K. Hiramatsu. 2006. Staphylococcal cassette chromosome mec (SCCmec) typing of methicillin-resistant Staphylococcus aureus strains isolated in 11 Asian countries: a proposal for a new nomenclature for SCCmec elements. Antimicrob. Agents Chemother. 501001-1012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Clinical and Laboratory Standards Institute. 2004. Performance standards for antimicrobial disk and dilution susceptibility tests for bacteria isolated from animals; informational supplement M31-S1, vol. 24, no. 17. Clinical and Laboratory Standards Institute, Wayne, PA.
- 4.Clinical and Laboratory Standards Institute. 2006. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically, 7th ed., vol. 26, no. 2. Approved standard M7-A7. Clinical and Laboratory Standards Institute, Wayne, PA.
- 5.Clinical and Laboratory Standards Institute. 2006. Performance standards for antimicrobial disk susceptibility tests, 9th ed., vol. 26, no. 1. Approved standard M2-A9. Clinical and Laboratory Standards Institute, Wayne, PA. [Google Scholar]
- 6.Clinical and Laboratory Standards Institute. 2006. Performance standards for antimicrobial susceptibility testing; sixteenth informational supplement M100-S16, vol. 26, no. 3. Clinical and Laboratory Standards Institute, Wayne, PA. [Google Scholar]
- 7.Corpet, F. 1988. Multiple sequence alignment with hierarchical clustering. Nucleic Acids Res. 1610881-10890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Couzinet, S., J. Yugueros, C. Barras, N. Visomblin, P. Francois, B. Lacroix, G. Vernet, D. Lew, A. Troesch, J. Schrenzel, and C. Jay. 2005. Evaluation of a high-density oligonucleotide array for characterization of grlA, grlB, gyrA and gyrB mutations in fluoroquinolone resistant Staphylococcus aureus isolates. J. Microbiol. Methods 60275-279. [DOI] [PubMed] [Google Scholar]
- 9.Cuny, C., and W. Witte. 2005. PCR for the identification of methicillin-resistant Staphylococcus aureus (MRSA) strains using a single primer pair specific for SCCmec elements and the neighbouring chromosome-borne orfX. Clin. Microbiol. Infect. 11834-837. [DOI] [PubMed] [Google Scholar]
- 10.Devriese, L. A., M. Vancanneyt, M. Baele, M. Vaneechoutte, E. De Graef, C. Snauwaert, I. Cleenwerck, P. Dawyndt, J. Swings, A. Decostere, and F. Haesebrouck. 2005. Staphylococcus pseudintermedius sp. nov., a coagulase-positive species from animals. Int. J. Syst. Evol. Microbiol. 551569-1573. [DOI] [PubMed] [Google Scholar]
- 11.Ferrero, L., B. Cameron, B. Manse, D. Lagneaux, J. Crouzet, A. Famechon, and F. Blanche. 1994. Cloning and primary structure of Staphylococcus aureus DNA topoisomerase IV: a primary target of fluoroquinolones. Mol. Microbiol. 13641-653. [DOI] [PubMed] [Google Scholar]
- 12.Hanssen, A. M., and J. U. Ericson Sollid. 2006. SCCmec in staphylococci: genes on the move. FEMS Immunol. Med. Microbiol. 468-20. [DOI] [PubMed] [Google Scholar]
- 13.Horii, T., Y. Suzuki, A. Monji, M. Morita, H. Muramatsu, Y. Kondo, M. Doi, A. Takeshita, T. Kanno, and M. Maekawa. 2003. Detection of mutations in quinolone resistance-determining regions in levofloxacin- and methicillin-resistant Staphylococcus aureus: effects of the mutations on fluoroquinolone MICs. Diagn. Microbiol. Infect. Dis. 46139-145. [DOI] [PubMed] [Google Scholar]
- 14.Iihara, H., T. Suzuki, Y. Kawamura, K. Ohkusu, Y. Inoue, W. Zhang, M. M. Shah, Y. Atagiri, Y. Hashi, and T. Zaki. 2006. Emerging multiple mutations and high-level fluoroquinolone resistance in methicillin-resistant Staphylococcus aureus isolated from ocular infections. Diagn. Microbiol. Infect. Dis. 56297-303. [DOI] [PubMed] [Google Scholar]
- 15.Intorre, L., M. Vanni, D. Di Bello, C. Pretti, V. Meucci, R. Tognetti, G. Soldani, G. Cardini, and O. Jousson. 2007. Antimicrobial susceptibility and mechanism of resistance to fluoroquinolones in Staphylococcus intermedius and Staphylococcus schleiferi. J. Vet. Pharmacol. Ther. 30464-469. [DOI] [PubMed] [Google Scholar]
- 16.Ito, T., X. X. Ma, F. Takeuchi, K. Okuma, H. Yuzawa, and K. Hiramatsu. 2004. Novel type V staphylococcal cassette chromosome mec driven by a novel cassette chromosome recombinase, ccrC. Antimicrob. Agents Chemother. 482637-2651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lina, G., Y. Piémont, F. Godail-Gamot, M. Bes, M.-O. Peter, V. Gauduchon, F. Vandenesch, and J. Etienne. 1999. Involvement of Panton-Valentine leukocidin-producing Staphylococcus aureus in primary skin infections and pneumonia. Clin. Infect. Dis. 291128-1132. [DOI] [PubMed] [Google Scholar]
- 18.Noguchi, N., T. Okihara, Y. Namiki, Y. Kumaki, Y. Yamanaka, M. Koyama, K. Wakasugi, and M. Sasatsu. 2005. Susceptibility and resistance genes to fluoroquinolones in methicillin-resistant Staphylococcus aureus isolated in 2002. Int. J. Antimicrob. Agents 25374-379. [DOI] [PubMed] [Google Scholar]
- 19.Oliveira, D. C., and H. de Lencastre. 2002. Multiplex PCR strategy for rapid identification of structural types and variants of the mec element in methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 462155-2161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Oliveira, D. C., C. Milheiriço, and H. de Lencastre. 2006. Redefining a structural variant of staphylococcal cassette chromosome mec, SCCmec type VI. Antimicrob. Agents Chemother. 503457-3459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Oliveira, D. C., C. Milheiriço, S. Vinga, and H. de Lencastre. 2006. Assessment of allelic variation in the ccrAB locus in methicillin-resistant Staphylococcus aureus clones. J. Antimicrob. Chemother. 5823-30. [DOI] [PubMed] [Google Scholar]
- 22.Perreten, V., L. Vorlet-Fawer, P. Slickers, R. Ehricht, P. Kuhnert, and J. Frey. 2005. Microarray-based detection of 90 antibiotic resistance genes of gram-positive bacteria. J. Clin. Microbiol. 432291-2302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Piriz, S., J. Valle, E. M. Mateos, R. de la Fuente, D. Cid, J. A. Ruiz-Santaquiteria, and S. Vadillo. 1996. In vitro activity of fifteen antimicrobial agents against methicillin-resistant and methicillin-susceptible Staphylococcus intermedius. J. Vet. Pharmacol. Ther. 19118-123. [DOI] [PubMed] [Google Scholar]
- 24.Poyart, C., G. Quesne, C. Boumaila, and P. Trieu-Cuot. 2001. Rapid and accurate species-level identification of coagulase-negative staphylococci by using the sodA gene as a target. J. Clin. Microbiol. 394296-4301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Prévost, G., T. Bouakham, Y. Piémont, and H. Monteil. 1995. Characterisation of a synergohymenotropic toxin produced by Staphylococcus intermedius. FEBS Lett. 376135-140. [DOI] [PubMed] [Google Scholar]
- 26.Sasaki, T., K. Kikuchi, Y. Tanaka, N. Takahashi, S. Kamata, and K. Hiramatsu. 2007. Methicillin-resistant Staphylococcus pseudintermedius in a veterinary teaching hospital. J. Clin. Microbiol. 451118-1125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sierra, J. M., J. G. Cabeza, M. Ruiz Chaler, T. Montero, J. Hernandez, J. Mensa, M. Llagostera, and J. Vila. 2005. The selection of resistance to and the mutagenicity of different fluoroquinolones in Staphylococcus aureus and Streptococcus pneumoniae. Clin. Microbiol. Infect. 11750-758. [DOI] [PubMed] [Google Scholar]
- 28.Tristan, A., T. Ferry, G. Durand, O. Dauwalder, M. Bes, G. Lina, F. Vandenesch, and J. Etienne. 2007. Virulence determinants in community and hospital methicillin-resistant Staphylococcus aureus. J. Hosp. Infect. 65(S2)105-109. [DOI] [PubMed] [Google Scholar]
- 29.Van Hoovels, L., A. Vankeerberghen, A. Boel, K. Van Vaerenbergh, and H. De Beenhouwer. 2006. First case of Staphylococcus pseudintermedius infection in a human. J. Clin. Microbiol. 444609-4612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Vickers, A. A., A. J. O'Neill, and I. Chopra. 2007. Emergence and maintenance of resistance to fluoroquinolones and coumarins in Staphylococcus aureus: predictions from in vitro studies. J. Antimicrob. Chemother. 60269-273. [DOI] [PubMed] [Google Scholar]
- 31.Zhang, K., J.-A. McClure, S. Elsayed, T. Louie, and J. M. Conly. 2005. Novel multiplex PCR assay for characterization and concomitant subtyping of staphylococcal cassette chromosome mec types I to V in methicillin-resistant Staphylococcus aureus. J. Clin. Microbiol. 435026-5033. [DOI] [PMC free article] [PubMed] [Google Scholar]