Abstract
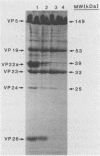
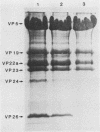
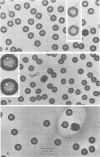
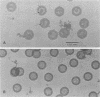
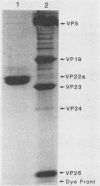
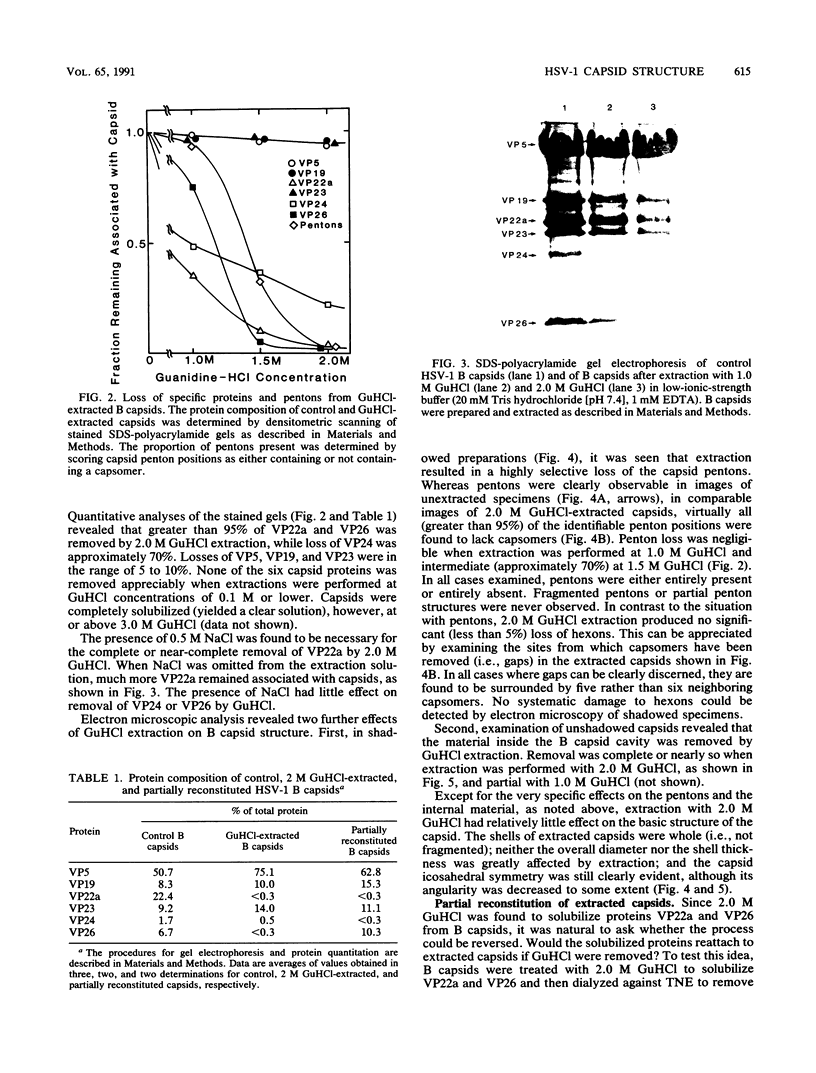
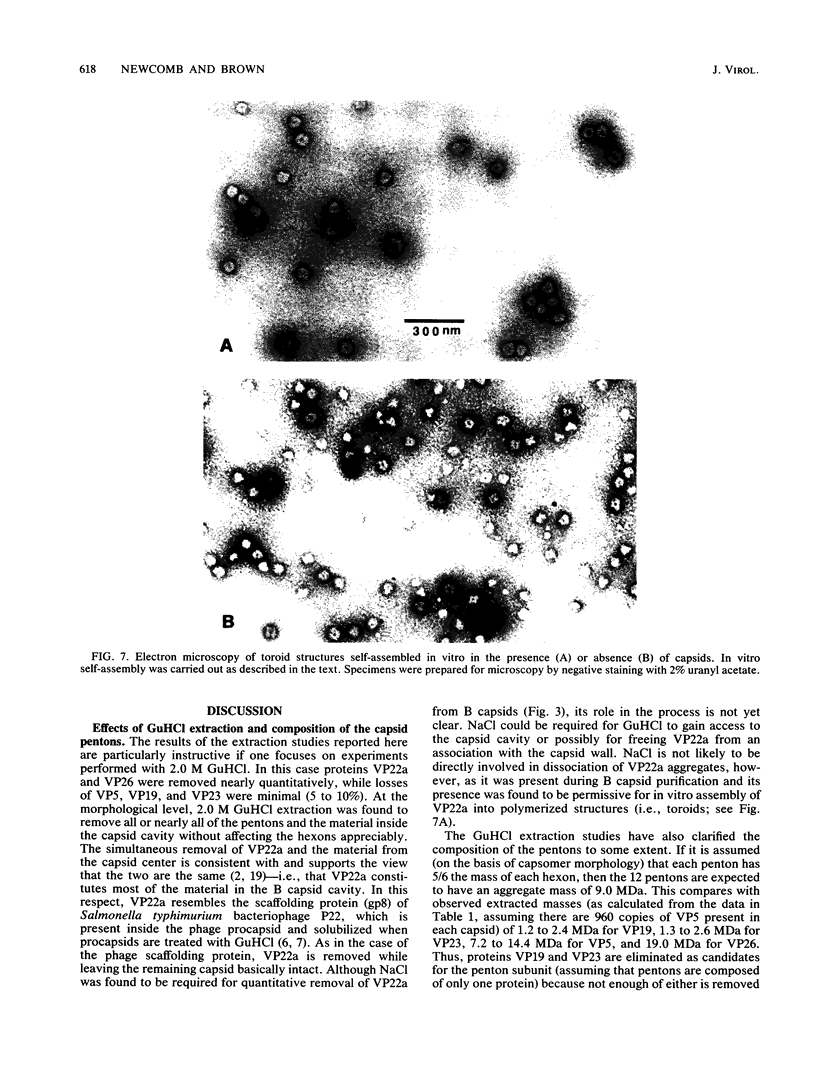
Viral B capsids were purified from cells infected with herpes simplex virus type 1 and extracted in vitro with 2.0 M guanidine hydrochloride (GuHCl). Sodium dodecyl sulfate-polyacrylamide gel analyses demonstrated that extraction resulted in the removal of greater than 95% of capsid proteins VP22a and VP26 while there was only minimal (less than 10%) loss of VP5 (the major capsid protein), VP19, and VP23. Electron microscopic analysis of extracted capsids revealed that the pentons and the material found inside the cavity of B capsids (primarily VP22a) were removed nearly quantitatively, but extracted capsids remained otherwise structurally intact. Few, if any, hexons were lost; the capsid diameter was not greatly affected; and its icosahedral symmetry was still clearly evident. The results demonstrate that neither VP19 nor VP23 could constitute the capsid pentons. Like the hexons, the pentons are most likely composed of VP5. When B capsids were treated with 2.0 M GuHCl and then dialyzed to remove GuHCl, two bands of viral material were separated by sucrose density gradient ultracentrifugation. The more rapidly migrating of the two consisted of capsids which lacked pentons and VP22a but had a full complement of VP26. Thus, VP26 must have reassociated with extracted capsids during dialysis. The more slowly migrating band consisted of torus-shaped structures approximately 60 nm in diameter which were composed entirely of VP22a. These latter structures closely resembled torus-shaped condensates often seen in the cavity of native B capsids. The results suggest a similarity between herpes simplex virus type 1 B capsids and procapsids of Salmonella bacteriophage P22. Both contain an internal protein (VP22a in the case of HSV-1 B capsids and gp8 or "scaffolding" protein in phage P22) that can be extracted in vitro with GuHCl and that is absent from mature virions.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aebi U., van Driel R., Bijlenga R. K., ten Heggeler B., van den Broek R., Steven A. C., Smith P. R. Capsid fine structure of T-even bacteriophages. Binding and localization of two dispensable capsid proteins into the P23* surface lattice. J Mol Biol. 1977 Mar 15;110(4):687–698. doi: 10.1016/s0022-2836(77)80084-3. [DOI] [PubMed] [Google Scholar]
- Baker T. S., Newcomb W. W., Booy F. P., Brown J. C., Steven A. C. Three-dimensional structures of maturable and abortive capsids of equine herpesvirus 1 from cryoelectron microscopy. J Virol. 1990 Feb;64(2):563–573. doi: 10.1128/jvi.64.2.563-573.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engel A., van Driel R., Driedonks R. A proposed structure of the prolate phage T4 prehead core. An electron microscopic study. J Ultrastruct Res. 1982 Jul;80(1):12–22. doi: 10.1016/s0022-5320(82)80028-2. [DOI] [PubMed] [Google Scholar]
- Friedmann A., Coward J. E., Rosenkranz H. S., Morgan C. Electron microscopic studies on assembly of herpes simplex virus upon removal of hydroxyurea block. J Gen Virol. 1975 Feb;26(2):171–181. doi: 10.1099/0022-1317-26-2-171. [DOI] [PubMed] [Google Scholar]
- Fuller M. T., King J. Purification of the coat and scaffolding proteins from procapsids of bacteriophage P22. Virology. 1981 Jul 30;112(2):529–547. doi: 10.1016/0042-6822(81)90300-7. [DOI] [PubMed] [Google Scholar]
- Fuller M. T., King J. Regulation of coat protein polymerization by the scaffolding protein of bacteriophage P22. Biophys J. 1980 Oct;32(1):381–401. doi: 10.1016/S0006-3495(80)84963-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furcinitti P. S., van Oostrum J., Burnett R. M. Adenovirus polypeptide IX revealed as capsid cement by difference images from electron microscopy and crystallography. EMBO J. 1989 Dec 1;8(12):3563–3570. doi: 10.1002/j.1460-2075.1989.tb08528.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson W., Roizman B. Proteins specified by herpes simplex virus. 8. Characterization and composition of multiple capsid forms of subtypes 1 and 2. J Virol. 1972 Nov;10(5):1044–1052. doi: 10.1128/jvi.10.5.1044-1052.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heine J. W., Honess R. W., Cassai E., Roizman B. Proteins specified by herpes simplex virus. XII. The virion polypeptides of type 1 strains. J Virol. 1974 Sep;14(3):640–651. doi: 10.1128/jvi.14.3.640-651.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imber R., Tsugita A., Wurtz M., Hohn T. Outer surface protein of bacteriophage lambda. J Mol Biol. 1980 May 25;139(3):277–295. doi: 10.1016/0022-2836(80)90131-x. [DOI] [PubMed] [Google Scholar]
- Ishii T., Yanagida M. Molecular organization of the shell of the Teven bacteriophage head. J Mol Biol. 1975 Oct 5;97(4):655–660. doi: 10.1016/s0022-2836(75)80065-9. [DOI] [PubMed] [Google Scholar]
- Laver W. G., Wrigley N. G., Pereira H. G. Removal of pentons from particles of adenovirus type 2. Virology. 1969 Nov;39(3):599–604. doi: 10.1016/0042-6822(69)90111-1. [DOI] [PubMed] [Google Scholar]
- Lee J. Y., Irmiere A., Gibson W. Primate cytomegalovirus assembly: evidence that DNA packaging occurs subsequent to B capsid assembly. Virology. 1988 Nov;167(1):87–96. doi: 10.1016/0042-6822(88)90057-8. [DOI] [PubMed] [Google Scholar]
- Lepault J., Dubochet J., Baschong W., Kellenberger E. Organization of double-stranded DNA in bacteriophages: a study by cryo-electron microscopy of vitrified samples. EMBO J. 1987 May;6(5):1507–1512. doi: 10.1002/j.1460-2075.1987.tb02393.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newcomb W. W., Brown J. C., Booy F. P., Steven A. C. Nucleocapsid mass and capsomer protein stoichiometry in equine herpesvirus 1: scanning transmission electron microscopic study. J Virol. 1989 Sep;63(9):3777–3783. doi: 10.1128/jvi.63.9.3777-3783.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newcomb W. W., Brown J. C. Use of Ar+ plasma etching to localize structural proteins in the capsid of herpes simplex virus type 1. J Virol. 1989 Nov;63(11):4697–4702. doi: 10.1128/jvi.63.11.4697-4702.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newcomb W. W., Brown J. C. Use of Ar+ plasma etching to localize structural proteins in viruses: studies with adenovirus 2. Anal Biochem. 1988 Mar;169(2):279–286. doi: 10.1016/0003-2697(88)90286-2. [DOI] [PubMed] [Google Scholar]
- Perdue M. L., Cohen J. C., Kemp M. C., Randall C. C., O'Callaghan D. J. Characterization of three species of nucleocapsids of equine herpesvirus type-1 (EHV-1). Virology. 1975 Mar;64(1):187–204. doi: 10.1016/0042-6822(75)90091-4. [DOI] [PubMed] [Google Scholar]
- Perdue M. L., Kemp M. C., Randall C. C., O'Callaghan D. J. Studies of the molecular anatomy of the L-M cell strain of equine herpes virus type 1: proteins of the nucleocapsid and intact virion. Virology. 1974 May;59(1):201–216. doi: 10.1016/0042-6822(74)90216-5. [DOI] [PubMed] [Google Scholar]
- Preston V. G., Coates J. A., Rixon F. J. Identification and characterization of a herpes simplex virus gene product required for encapsidation of virus DNA. J Virol. 1983 Mar;45(3):1056–1064. doi: 10.1128/jvi.45.3.1056-1064.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richards K. E., Williams R. C., Calendar R. Mode of DNA packing within bacteriophage heads. J Mol Biol. 1973 Aug 5;78(2):255–259. doi: 10.1016/0022-2836(73)90114-9. [DOI] [PubMed] [Google Scholar]
- Rixon F. J., Cross A. M., Addison C., Preston V. G. The products of herpes simplex virus type 1 gene UL26 which are involved in DNA packaging are strongly associated with empty but not with full capsids. J Gen Virol. 1988 Nov;69(Pt 11):2879–2891. doi: 10.1099/0022-1317-69-11-2879. [DOI] [PubMed] [Google Scholar]
- Russell W. C., Valentine R. C., Pereira H. G. The effect of heat on the anatomy of the adenovirus. J Gen Virol. 1967 Oct;1(4):509–522. doi: 10.1099/0022-1317-1-4-509. [DOI] [PubMed] [Google Scholar]
- Schaffer P. A., Brunschwig J. P., McCombs R. M., Benyesh-Melnick M. Electron microscopic studies of temperature-sensitive mutants of herpes simplex virus type 1. Virology. 1974 Dec;62(2):444–457. doi: 10.1016/0042-6822(74)90406-1. [DOI] [PubMed] [Google Scholar]
- Schrag J. D., Prasad B. V., Rixon F. J., Chiu W. Three-dimensional structure of the HSV1 nucleocapsid. Cell. 1989 Feb 24;56(4):651–660. doi: 10.1016/0092-8674(89)90587-4. [DOI] [PubMed] [Google Scholar]
- Sherman G., Bachenheimer S. L. Characterization of intranuclear capsids made by ts morphogenic mutants of HSV-1. Virology. 1988 Apr;163(2):471–480. doi: 10.1016/0042-6822(88)90288-7. [DOI] [PubMed] [Google Scholar]
- Steven A. C., Roberts C. R., Hay J., Bisher M. E., Pun T., Trus B. L. Hexavalent capsomers of herpes simplex virus type 2: symmetry, shape, dimensions, and oligomeric status. J Virol. 1986 Feb;57(2):578–584. doi: 10.1128/jvi.57.2.578-584.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas D., Newcomb W. W., Brown J. C., Wall J. S., Hainfeld J. F., Trus B. L., Steven A. C. Mass and molecular composition of vesicular stomatitis virus: a scanning transmission electron microscopy analysis. J Virol. 1985 May;54(2):598–607. doi: 10.1128/jvi.54.2.598-607.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vernon S. K., Ponce de Leon M., Cohen G. H., Eisenberg R. J., Rubin B. A. Morphological components of herpesvirus. III. Localization of herpes simplex virus type 1 nucleocapsid polypeptides by immune electron microscopy. J Gen Virol. 1981 May;54(Pt 1):39–46. doi: 10.1099/0022-1317-54-1-39. [DOI] [PubMed] [Google Scholar]
- Wurtz M., Kistler J., Hohn T. Surface structure of in vitro assembled bacteriophage lambda polyheads. J Mol Biol. 1976 Feb 15;101(1):39–56. doi: 10.1016/0022-2836(76)90065-6. [DOI] [PubMed] [Google Scholar]
- Yanagida M. Molecular organization of the shell of T-even bacteriophage head. II. Arrangement of subunits in the head shell of giant phages. J Mol Biol. 1977 Feb 5;109(4):515–537. doi: 10.1016/s0022-2836(77)80089-2. [DOI] [PubMed] [Google Scholar]
- van Driel R. Assembly of bacteriophage T4 head-related structures. IV. Isolation and association properties of T4 prehead proteins. J Mol Biol. 1980 Mar 25;138(1):27–42. doi: 10.1016/s0022-2836(80)80003-9. [DOI] [PubMed] [Google Scholar]