Abstract
The expression of GnRH (GnRH-I, LHRH) and its receptor as a part of an autocrine regulatory system of cell proliferation has been demonstrated in a number of human malignant tumors, including cancers of the ovary. The proliferation of human ovarian cancer cell lines is time- and dose-dependently reduced by GnRH and its superagonistic analogs. The classical GnRH receptor signal-transduction mechanisms, known to operate in the pituitary, are not involved in the mediation of antiproliferative effects of GnRH analogs in these cancer cells. The GnRH receptor rather interacts with the mitogenic signal transduction of growth-factor receptors and related oncogene products associated with tyrosine kinase activity via activation of a phosphotyrosine phosphatase resulting in downregulation of cancer cell proliferation. In addition GnRH activates nucleus factor κB (NFκB) and protects the cancer cells from apoptosis. Furthermore GnRH induces activation of the c-Jun N-terminal kinase/activator protein-1 (JNK/AP-1) pathway independent of the known AP-1 activators, protein kinase (PKC) or mitogen activated protein kinase (MAPK/ERK).
Recently it was shown that human ovarian cancer cells express a putative second GnRH receptor specific for GnRH type II (GnRH-II). The proliferation of these cells is dose- and time-dependently reduced by GnRH-II in a greater extent than by GnRH-I (GnRH, LHRH) superagonists. In previous studies we have demonstrated that in ovarian cancer cell lines except for the EFO-27 cell line GnRH-I antagonist Cetrorelix has comparable antiproliferative effects as GnRH-I agonists indicating that the dichotomy of GnRH-I agonists and antagonists might not apply to the GnRH-I system in cancer cells. After GnRH-I receptor knock down the antiproliferative effects of GnRH-I agonist Triptorelin were abrogated while the effects of GnRH-I antagonist Cetrorelix and GnRH-II were still existing. In addition, in the ovarian cancer cell line EFO-27 GnRH-I receptor but not putative GnRH-II receptor expression was found. These data suggest that in ovarian cancer cells the antiproliferative effects of GnRH-I antagonist Cetrorelix and GnRH-II are not mediated through the GnRH-I receptor.
Introduction
The hypothalamic decapeptide gonadotropin releasing hormone (GnRH, GnRH-I), also called luteinizing hormone releasing hormone (LHRH), plays a key role in the regulation of mammalian reproduction [1-3]. It is released from the hypothalamus in a pulsatile manner and stimulates the synthesis and release of luteinizing hormone (LH) and follicle stimulating hormone (FSH). In addition to this classic hypophysiotropic actions, GnRH functions as a modulator of the activity of diverse systems in the brain and many peripheral organs [for review see [4]]. An autocrine/paracrine function of GnRH has been suggested to exist, for instance, in the placenta, granulosa cells, myometrium, and lymphoid cells [for review see [4,5]]. In addition, it is probable that such GnRH-I-based autocrine systems are present in a number of human malignant tumors including cancers of the ovary, endometrium, breast and prostate [for review see [4,5]].
The GnRH-I system in human ovarian cancers
In earlier studies the expression of GnRH-I and its receptor (GnRH-I receptor) as well as direct antiproliferative effects of GnRH-I and its analogs have been demonstrated in a number of malignant human tumors, including cancers of the ovary [6-14]. Data available today suggest that approximately 80% of ovarian cancers express high-affinity GnRH-I receptors [4,5,15]. These findings suggested the presence of a local regulatory system based on GnRH-I. The same situation was found in endometrial, breast and prostate cancer cells [4,5,15,16].
The in vitro proliferation of a variety of human tumor cell lines, including those from ovarian cancers can be inhibited by GnRH-I and its agonistic analogs in a dose- and time-dependent manner [4,9,11,12,17-20]. In most human ovarian cancer cells except for the ovarian cancer cell line EFO-27 GnRH-I antagonists act like agonists indicating that the dichotomy of GnRH-I agonists and antagonists does not exist in tumor cells [5,9]. Using human ovarian cancer cell line OV-1063 xenografted into nude mice, Yano et al. [12] demonstrated a significant inhibition of tumor growth by chronic treatment with the GnRH-I antagonist Cetrorelix but not with the GnRH-I agonist Triptorelin. As both GnRH-I analogs induced a comparable suppression of the pituitary-gonadal axis, the authors speculated that in vivo anti tumor effects of Cetrorelix were exerted directly on GnRH-I receptors in tumors [8]. The findings on direct anti tumor effects of GnRH-I analogs in ovarian and endometrial cancer reported by several other groups are completely or partly in agreement with the results described earlier [4,8,21,22]. In contrast, other investigators failed to detect direct anti tumor effects of GnRH-I analogs in human ovarian and endometrial cancer cell lines or observed them only at extremely high GnRH-I analog concentrations [23-25]. These discrepancies might be due to the fact that probably the majority of the cell lines used by these authors did not express high-affinity GnRH-I receptors [4,26]. Alternatively, differences in culture or experimental conditions as well as in the types of GnRH-I analogs used might be responsible for the observed variance. In the case of prostate cancer, several groups reported direct antiproliferative effects of GnRH-I analogs in vitro and in animal in vivo models, which could be mediated through specific GnRH-I-binding sites [1,27-32].
The proliferation of human ovarian cancer cells was significantly increased after treatment with an antiserum to GnRH-I, suggesting that GnRH-I produced by human ovarian cancer cells acts as a negative autocrine regulator of proliferation [33]. In contrast, Arencibia and Schally [34] have recently reported that in ES-2 human ovarian cancer cells GnRH-I agonist Triptorelin at 10 ng/ml stimulated the proliferation in vitro after 48 h, but was inhibitory after 72 h and at concentrations of 1000 ng/ml. GnRH-I antagonist Cetrorelix inhibited growth of ES-2 cell line only at 1000 ng/ml. The incubation of ES-2 ovarian cancer cells in vitro with an GnRH-I antibody inhibited cell proliferation in a time and concentration-dependent manner. These results suggest that GnRH-I may function as an autocrine growth factor in this ovarian cancer cell line [34]. Differences between tumor cell lines e.g. variances in G-protein coupling and signaling might explain these discrepancies.
Signaling mechanisms mediating the direct anti-tumor effects of GnRH-I
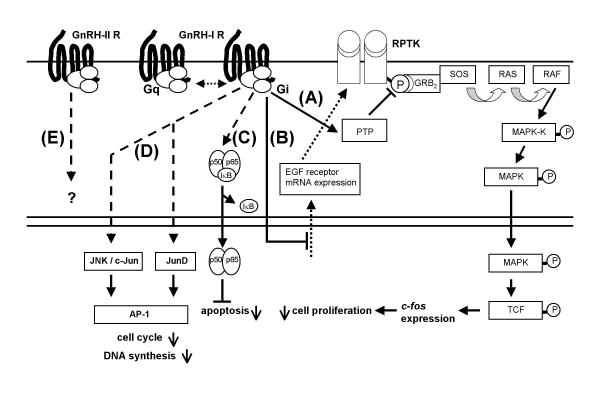
During the last ten years, the signaling mechanisms mediating the antiproliferative effects of GnRH-I analogs in ovarian, endometrial and breast cancer cells have been elucidated. The signaling mechanism of GnRH-I receptor in human cancers is quite different from that in pituitary gonadotrophs, where GnRH-I receptors couple to G-protein αq and activate phospholipase C (PLC), protein kinase C (PKC), and adenylyl cyclase (AC) [reviewed in [4]]. Although we could clearly demonstrate the activation of PLC, PKC, and AC in these tumor cells by pharmacological stimuli [35], the signaling pathways induced by GnRH-I in pituitary gonadotrophs were not activated by GnRH-I agonist Triptorelin in ovarian, endometrial and breast cancer cell lines [35,36]. We found, however, that after binding of its ligand, the GnRH-I receptor in these cancers couples to G-protein αi and activates a phosphotyrosine phosphatase (PTP) [35-40] (Fig. 1A). This PTP dephosphorylates EGF receptors [36]. As a result, mitogenic signaling induced by EGF binding to its receptor is abrogated leading to a suppression of EGF-induced activation of mitogen-activated protein kinase (MAPK) [35], c-fos expression [41], and EGF-induced proliferation [35] (Fig. 1A). These findings are in accord with reports that GnRH-I analogs reduce expression of growth factor receptors and their mRNA [12,42,43] (Fig. 1B) and/or growth factor induced tyrosine kinase activity [35,37-39,42,44-46]. The reason for the differences of the GnRH-I receptor signaling between pituitary gonadotrophs and tumor cells remains unclear, as we could not find mutations or splice variants in the tumor GnRH-I receptor which might explain this phenomenon.
Figure 1.
GnRH-I and GnRH-II signaling in human gynecological cancer cells: A) GnRH-I activates a phosphotyrosine phosphatase (PTP) inhibiting the mitogenic signal transduction of growth factor receptors resulting in downregulation of cell proliferation. B) GnRH-I downregulates epidermal growth factor (EGF) receptor mRNA expression. C) Activated GnRH-I receptor induces nucleus factor κB (NFκB) activation and nuclear translocation of activated NFκB. Activated NFκB now couples to κB DNA binding sites and induces expression of anti-apoptotic mechanisms. D) GnRH-I activates c-Jun N-terminal kinase (JNK), induces JunD-DNA binding and stimulates activator protein (AP-1) activity, resulting in reduced proliferation as indicated by increased G0/1 phase of cell cycle and decreased DNA synthesis. E) Unknown signal transduction of a putative human GnRH-II receptor. In human gynecological cancer cells GnRH-I analogs mediate antiproliferative actions via inhibition of growth factor-induced mitogenic signal transduction. In addition GnRH-I induces growth factor receptor downregulation. GnRH-I protects the cancer cells from apoptosis via activation of NFκB, stimulates AP-1 activity and extends cell cycle. PPTK, receptor protein tyrosine kinase; GRB2, adaptor protein; SOS, guanine nucleotide exchange factor; RAS, small GTPase; RAF, a protein-serine/threonine kinase; MAPK-K, mitogen activated kinase kinase; MAPK, mitogen activated kinase; TCF, transcription factor; IκB, inhibitory κB; Gq, G-protein αq; Gi, G-protein αi; p50, p65, NFκB subunits.
Recently, it was speculated that induction of apoptosis might be involved in the antiproliferative activity of GnRH-I and its analogs [47-49]. However, though we have tried to show induction of apoptosis by GnRH-I analogs, we have found it only in one (Ca-Ov-3) of nine (EFO-21, EFO-27, OVCAR-3, AN-3-CA, Ca-Ov-3, SK-OV-3) ovarian cancer cell lines [[50] and unpublished results]. In contrast, we found that GnRH-I agonist Triptorelin reduced apoptosis induced by the cytotoxic agent doxorubicin. Since Triptorelin-induced reduction of Doxorubicin-induced apoptosis was blocked by inhibition of nucleus factor kappa B (NFκB) translocation into the nucleus and Triptorelin was shown to induce NFκB activation (Fig. 1C), we concluded that GnRH-I has an antiapoptotic effect mediated through NFκB activation in these human ovarian cancer cells [50]. This possibility to protect ovarian cancer cells from programmed cell death is a new and important feature in GnRH-I signaling in ovarian tumors apart from the inhibitory interference with the mitogenic pathway.
Recently, it became evident that it is not only mitogenic signaling of growth factor receptors that is modulated by GnRH-I in human cancers. In human ovarian and endometrial cancer cells GnRH-I agonist Triptorelin stimulates the activity of activator protein-1 (AP-1) mediated through pertussis toxin-sensitive G-protein αi (Fig. 1D). In addition, Triptorelin activates JNK, known to activate AP-1 [51] (Fig. 1D). In earlier investigations we have shown that Triptorelin does not activate phospholipase C (PLC) and protein kinase C (PKC) in endometrial and ovarian cancer cells [35]. In addition, it has been demonstrated that Triptorelin inhibits growth factor-induced mitogen activated protein kinase (MAPK, ERK) activity [35]. Thus Triptorelin-induced activation of the JNK/AP-1 pathway in endometrial cancer cells is independent of the known AP-1 activators, PKC or MAPK (ERK) (Fig. 1D).
In ovarian and endometrial cancer cells GnRH-I analogs mediate antiproliferative actions via inhibition of growth factor-induced mitogenic signal transduction. GnRH-I agonist Triptorelin protects the cancer cells from apoptosis via activation of NFκB, and Triptorelin stimulates AP-1 and JNK activity. Recently Yamauchi et al. [52] found that JNK is involved in inhibition of cell proliferation induced by α1B-adrenergic receptor in human embryonic kidney cells. In a study in rats, c-jun mRNA depression and endometrial epithelial cell proliferation were suggested to be linked [53]. In UT-OC-3 ovarian cancer cells cytokines have inhibitory effects on cell proliferation and activate AP-1 and NFκB [54]. Since the antiproliferative GnRH-I agonist Triptorelin activates the JNK/c-jun pathway and JNK/c-jun was found to be involved in downregulation of cell proliferation in different systems, it seems reasonable to speculate that the JNK/c-jun pathway is involved in the antiproliferative actions of the GnRH agonist Triptorelin. In addition, we have shown that GnRH-I agonist Triptorelin induces JunD-DNA binding, resulting in reduced proliferation as indicated by increased G0/1 phase of cell cycle and decreased DNA synthesis (Fig. 1D). Since GnRH-I activates NFκB and protects ovarian cancer cells from Doxorubicin-induced apoptosis and JunD is shown to decrease cell cycle and cell proliferation, we propose that JunD activated by GnRH-I acts as a modulator of cell proliferation and cooperates with the anti-apoptotic and anti-mitogenic functions of GnRH-I [55].
GnRH-II and its receptor
In non-mammalian vertebrates it became evident that three structural variants of GnRH were present in individual species [56,57]. A similar situation seems to exist in mammals. One of these GnRH variants is GnRH-II, which is totally conserved in structure in the evolution from fish to mammals [58,59]. In human granulosa-luteal cells expression of GnRH-II was found [60]. In these cells GnRH-I agonists exerted a biphasic effect on GnRH-I receptor density, while GnRH-II agonists induced a downregulation of GnRH-I receptor expression and of GnRH-II itself [60]. Recently Millar et al. cloned a type II GnRH receptor from the marmoset monkey which is highly selective for GnRH-II [61]. At the same time Neill et al. cloned the GnRH-II receptor from the rhesus monkey [62]. Only 41% (marmoset GnRH-II receptor) and 39% (rhesus monkey GnRH-II receptor) identities with the GnRH-I receptor have been reported [61,62]. In contrast to the GnRH-I receptor the GnRH-II receptor in the marmoset and rhesus monkey has a C-terminal, cytoplasmatic tail resulting in a more rapid internalization [61,62].
Using RT-PCR and Southern blot analysis we could recently show that human ovarian and endometrial cancer cells express a putative second GnRH receptor specific for GnRH-II [63] (Fig. 1E). The proliferation of these cell lines was reduced in a dose- and time-dependent manner by native GnRH-II. These effects were significantly higher than the antiproliferative effects of equimolar doses of GnRH-I agonist Triptorelin [63]. In the GnRH-II receptor mRNA positive but GnRH-I receptor negative ovarian cancer cell line SK-OV-3 native GnRH-II but not GnRH-I agonist Triptorelin had antiproliferative effects [63]. In previous studies we have demonstrated that in ovarian cancer cell lines except for the EFO-27 cell line GnRH-I antagonist Cetrorelix has comparable antiproliferative effects as GnRH-I agonists indicating that the dichotomy of GnRH-I agonists and antagonists might not apply to the GnRH-I system in cancer cells [9] After GnRH-I receptor knock down in EFO-21 and OVCAR-3 human ovarian cancer cell lines the antiproliferative effects of GnRH-I agonist Triptorelin were abrogated while the effects of GnRH-I antagonist Cetrorelix and GnRH-II were still existing [unpublished results]. In addition, in the ovarian cancer cell line EFO-27 GnRH-I receptor but not putative GnRH-II receptor expression was found [unpublished results]. These data suggest that in ovarian and endometrial cancer cells the antiproliferative effects of GnRH-I antagonist Cetrorelix and GnRH-II are not mediated through the GnRH-I receptor. It is possible that these antiproliferative effects are mediated through a putative GnRH-II receptor. However, the human GnRH-II receptor is expressed as a variety of splice variants [64] and a functional human GnRH-II receptor transcript has not been found until now. Further investigations are required to determine whether these GnRH-II receptor splice variants translate to functional proteins.
Gonadotropin biosynthesis and secretion by GnRH-I can be mediated by activation of MAP kinases. Therefore, Millar et al. [61] assessed the capacity of both human GnRH-I receptor and marmoset GnRH-II receptor to activate the MAP kinases ERK2, JNK, and p38α in COS-7 cells transfected with either the human GnRH-I receptor or the marmoset GnRH-II receptor. At the GnRH-I receptor, GnRH-I was considerably more potent than GnRH-II in activating ERK2 whereas at the GnRH-II receptor, GnRH-II was markedly more potent than GnRH-I [61]. Neither GnRH-I receptor nor GnRH-II receptor stimulation resulted in activation of JNK [61]. Activation of p38α was detected on stimulation of GnRH-II receptor with GnRH-II but not with stimulation of GnRH-I receptor with GnRH-I [61]. These data suggest that there are distinct differences in the signal transduction by the two GnRH receptors. However, the signal transduction mechanisms mediating the antiproliferative activity of GnRH-II in human ovarian cancer cells are not known (Fig. 1E).
As the antiproliferative activity of native GnRH-II is significantly superior to that of the GnRH-I superagonists, superactive agonists of GnRH-II might become efficacious drugs for the therapy of human cancers. However, the more widely distributed expression pattern of GnRH-II receptor [61] could limit the effectiveness of GnRH-II agonists.
Acknowledgments
Acknowledgements
Our work was supported by the Deutsche Forschungsgemeinschaft (SFB 215-B10 and GR 1895/2-1), the PE Kempkes Foundation, Marburg, Germany, the Bundesministerium für Bildung und Forschung, the German-Israeli Foundation for Scientific Research and Development (I-684-176.2/2000), Ferring Pharmaceuticals, Copenhagen, Denmark, Asta Medica AG, Frankfurt, Germany.
Contributor Information
Carsten Gründker, Email: grundker@med.uni-goettingen.de.
Günter Emons, Email: emons@med.uni-goettingen.de.
References
- Schally AV. Hypothalamic hormones from neuroendocrinology to cancer therapy. Anticancer Drugs. 1994;5:115–130. [PubMed] [Google Scholar]
- Stojilkovic SS, Catt KJ. Expression and signal transduction pathways of gonadotropin-releasing hormone receptors. Recent Prog Horm Res. 1995;30:161–205. doi: 10.1016/b978-0-12-571150-0.50012-3. [DOI] [PubMed] [Google Scholar]
- Stanislaus D, Pinter JH, Janovick JA, Conn PM. Mechanisms mediating multiple physiological responses to gonadotropin-releasing hormone. Mol Cell Endocrinol. 1998;144:1–10. doi: 10.1016/S0303-7207(98)00126-9. [DOI] [PubMed] [Google Scholar]
- Gründker C, Günthert AR, Westphalen S, Emons G. Biology of the GnRH system in human gynecological cancers. Eur J Endocrinol. 2002;146:1–14. doi: 10.1530/eje.0.1460001. [DOI] [PubMed] [Google Scholar]
- Emons G, Ortmann O, Schulz KD, Schally AV. Growth-inhibitory actions of analogues of luteinizing hormone releasing hormone on tumor cells. Trends Endocrinol Metab. 1997;8:155–362. doi: 10.1016/s1043-2760(97)00155-0. [DOI] [PubMed] [Google Scholar]
- Emons G, Pahwa GS, Brack C, Sturm R, Oberheuser F, Knuppen R. Gonadotropin releasing hormone binding sites in human epithelial ovarian carcinomata. Eur J Cancer Clin Oncol. 1989;25:215–221. doi: 10.1016/0277-5379(89)90011-4. [DOI] [PubMed] [Google Scholar]
- Pahwa GS, Vollmer G, Knuppen R, Emons G. Photoaffinity labelling of gonadotropin releasing hormone binding sites in human epithelial ovarian carcinomata. Biochem Biophys Res Commun. 1989;161:1086–1092. doi: 10.1016/0006-291x(89)91354-5. [DOI] [PubMed] [Google Scholar]
- Thompson MA, Adelson MD, Kaufman LM. Lupron retards proliferation of ovarian tumor cells cultured in serum-free medium. J Clin Endocrinol Metab. 1991;72:1036–1041. doi: 10.1210/jcem-72-5-1036. [DOI] [PubMed] [Google Scholar]
- Emons G, Ortmann O, Becker M, Irmer G, Springer B, Laun R, Hölzel F, Schulz KD, Schally AV. High affinity binding and direct antiproliferative effects of LHRH analogues in human ovarian cancer cell lines. Cancer Res. 1993;54:5439–5446. [PubMed] [Google Scholar]
- Ohno T, Imai A, Furui T, Takahashi K, Tamaya T. Presence of gonadotropin-releasing hormone and his messenger ribunucleic acid in human ovarian epithelial carcinoma. Am J Obstet Gynecol. 1993;169:605–610. doi: 10.1016/0002-9378(93)90630-2. [DOI] [PubMed] [Google Scholar]
- Yano T, Pinski J, Radulovic S, Schally AV. Inhibition of human epithelial ovarian cancer cell growth in vitro by agonistic and antagonistic analogues of luteinizing hormone-releasing hormone. Proc Natl Acad Sci USA. 1994;91:1701–1704. doi: 10.1073/pnas.91.5.1701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yano T, Pinski J, Halmos G, Szepeshazi K, Schally AV. Inhibition of growth of OV-1063 human epithelial ovarian cancer xenografts in nude mice by treatment with luteinizing hormone-releasing hormone antagonist SB-75. Proc Natl Acad Sci USA. 1994;91:7090–7094. doi: 10.1073/pnas.91.15.7090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kakar SS, Grizzle WE, Neill JD. The nucleotide sequence of human GnRH receptors in breast and ovarian tumors are identical with that found in pituitary. Mol Cell Endocrinol. 1994;106:145–149. doi: 10.1016/0303-7207(94)90196-1. [DOI] [PubMed] [Google Scholar]
- Irmer G, Bürger C, Müller R, Ortmann O, Peter U, Kakar S, Neill JD, Schulz KD, Emons G. Expression of the messenger RNAs for luteinizing hormone-releasing hormone (LHRH) and its receptor in human ovarian epithelial carcinoma. Cancer Res. 1995;55:817–822. [PubMed] [Google Scholar]
- Völker P, Gründker C, Schmidt O, Schulz KD, Emons G. Expression of receptors for luteinizing hormone-releasing hormone in human ovarian and endometrial cancers: frequency, autoregulation and correlation with direct antiproliferative activity of LHRH analogues. Am J Obstet Gynecol. 2002;186:171–179. doi: 10.1067/mob.2002.119633. [DOI] [PubMed] [Google Scholar]
- Dondi D, Limonta P, Moretti RM, Marelli MM, Garattini E, Motta M. Antiproliferative effects of luteinizing hormone-releasing hormone (LHRH) agonists on human androgen-independent prostate cancer cell line DU 145: Evidence for an autocrine-inhibitory LHRH loop. Cancer Res. 1994;54:4091–4095. [PubMed] [Google Scholar]
- Emons G, Schally AV. The use of luteinizing hormone-releasing hormone agonists and antagonists in gynecological cancers. Hum Reprod. 1994;9:1364–1379. doi: 10.1093/oxfordjournals.humrep.a138714. [DOI] [PubMed] [Google Scholar]
- Emons G, Schröder B, Ortmann O, Westphalen S, Schulz KD, Schally AV. High affinity binding and direct antiproliferative effects of luteinizing hormone-releasing hormone analogs in human endometrial cancer cell lines. J Clin Endocrinol Metab. 1993;77:1458–1464. doi: 10.1210/jc.77.6.1458. [DOI] [PubMed] [Google Scholar]
- Miller WR, Scott WN, Morris R, Fraser HM, Sharpe RM. Growth of human breast cancer cells inhibited by luteinizing hormone-releasing hormone agonist. Nature. 1985;313:231–233. doi: 10.1038/313231a0. [DOI] [PubMed] [Google Scholar]
- Shibata S, Sato H, Ota H, Karube A, Takahashi O, Tanaka T. Involvement of annexin V in antiproliferative effects of gonadotropin-releasing hormone agonists on human endometrial cancer cell line. Gynecol Oncol. 1997;66:217–221. doi: 10.1006/gyno.1997.4746. [DOI] [PubMed] [Google Scholar]
- Kleinman D, Douvdevani A, Schally AV, Levy J, Sharoni Y. Direct growth inhibition of human endometrial cancer cells by the gonadotropin-releasing hormone antagonist SB-75: role of apoptosis. Am J Obst Gynecol. 1994;170:96–102. doi: 10.1016/s0002-9378(94)70391-4. [DOI] [PubMed] [Google Scholar]
- Pályi I, Vincze B, Kálnay A, Turi G, Mezo I, Teplan I, Seprodi J, Pato J, Mora M. Effect of gonadotropin releasing hormone analogs and their conjugates on gonadotropin-releasing hormone receptor-positive human cancer cell lines. Cancer Detect Prev. 1996;20:146–152. [PubMed] [Google Scholar]
- Connor JP, Buller RE, Conn PM. Effects of GnRH analogs on six ovarian cancer cell lines in culture. Gynecol Oncol. 1994;54:3215–3221. doi: 10.1006/gyno.1994.1170. [DOI] [PubMed] [Google Scholar]
- Manetta A, Gamboa-Vujicic L, Paredes P, Emma D, Liao S, Leong L, Asch B, Schally AV. Inhibition of growth of human ovarian cancer in nude mice by luteinizing hormone-releasing hormone antagonist Cetrorelix (SB-75) Fertil Steril. 1995;63:282–287. [PubMed] [Google Scholar]
- Chatzaki E, Bax CMR, Eidne KA, Anderson L, Grudzinskas JG, Gallagher CJ. The expression of gonadotropin-releasing hormone and its receptor in endometrial cancer and its relevance as an autocrine growth factor. Cancer Res. 1996;56:2055–2065. [PubMed] [Google Scholar]
- Emons G, Ortmann O, Irmer G, Müller V, Schulz KD, Schally AV. Treatment of ovarian cancer with LHRH antagonists. In: Filicori M, Flamigni C, editor. Treatment with GnRH Analogs: Controversies and Perspectives. Carnforth: Parthenon Publishing; 1996. pp. 165–172. [Google Scholar]
- Loop SM, Gorder CA, Lewis SM, Saiers JH, Drivdahl RH, Ostenson RC. Growth inhibition of human prostatic cancer cells by an agonist of gonadotropin-releasing hormone. Prostate. 1995;26:179–188. doi: 10.1002/pros.2990260403. [DOI] [PubMed] [Google Scholar]
- Motta M, Dondi D, Moretti M, Montagnani-Marelli M, Pimpinelli F, Maggi R, Limonta P. Role of growth factors, steroid and peptide hormones in the regulation of human prostatic tumor growth. J Steroid Biochem Mol Biol. 1996;56:107–111. doi: 10.1016/0960-0760(95)00240-5. [DOI] [PubMed] [Google Scholar]
- Jungwirth A, Galvan G, Pinski J, Halmos G, Szepeshazi K, Cai RZ, Groot K, Schally AV. Luteinizing hormone releasing hormone antagonist Cetrorelix (SB-75) and bombesin antagonist RC-3940-II inhibit the growth of androgen-independent PC-3 prostate cancer in nude mice. Prostate. 1997;32:164–172. doi: 10.1002/(SICI)1097-0045(19970801)32:3<164::AID-PROS2>3.3.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- Jungwirth A, Pinski J, Galvan G, Halmos G, Szepeshazi K, Cai RZ, Groot K, Vadillo-Buenfil M, Schally AV. Inhibition of growth of androgen-independent DU-145 prostate cancer in vivo by luteinizing hormone-releasing hormone antagonist Cetrorelix and bombesin antagonists RC-3940-II and RC-3950-II. Eur J Cancer. 1997;33:1141–1148. doi: 10.1016/S0959-8049(97)00072-5. [DOI] [PubMed] [Google Scholar]
- Montagnani-Marelli M, Moretti RM, Dondi D, Limonta P, Motta M. Effects of LHRH agonists on the growth of human prostatic tumor cells: "in vitro" and "in vivo" studies. Arch Ital Urol Androl. 1997;69:257–263. [PubMed] [Google Scholar]
- Qayum A, Gullick W, Clayton RC, Sikora K, Waxman J. The effects of gonadotropin-releasing hormone analogues in prostate cancer are mediated through specific tumor receptors. Br J Cancer. 1990;62:96–99. doi: 10.1038/bjc.1990.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emons G, Weiβ S, Ortmann O, Gründker C, Schulz KD. Luteinizing hormone-releasing hormone (LHRH) might act as a negative autocrine regulator of proliferation of human ovarian cancer. Eur J Endocrinol. 2000;142:665–670. doi: 10.1530/eje.0.1420665. [DOI] [PubMed] [Google Scholar]
- Arencibia JM, Schally AV. Luteinizing hormone-releasing hormone as an autocrine growth factor in ES-2 ovarian cancer cell line. Int J Oncol. 2000;16:1009–1013. doi: 10.3892/ijo.16.5.1009. [DOI] [PubMed] [Google Scholar]
- Emons G, Müller V, Ortmann O, Grossmann G, Trautner U, von Stuckrad B, Schulz KD, Schally AV. Luteinizing hormone-releasing hormone agonist triptorelin antagonizes signal transduction and mitogenic activity of epidermal growth factor in human ovarian and endometrial cancer cell lines. Int J Oncol. 1996;9:1129–1137. doi: 10.3892/ijo.9.6.1129. [DOI] [PubMed] [Google Scholar]
- Gründker C, Völker P, Günthert AR, Emons G. Antiproliferative signaling of LHRH in human endometrial and ovarian cancer cells through G-protein αi-mediated activation of phosphotyrosine phosphatase. Endocrinology. 2001;142:2369–2380. doi: 10.1210/en.142.6.2369. [DOI] [PubMed] [Google Scholar]
- Lee MT, Liebow C, Kramer AR, Schally AV. Effects of epidermal growth factor and analogues of luteinizing hormone-releasing hormone and somatostatin on phosphorylation of tyrosine residues of specific substrates in various tumors. Proc Natl Acad Sci USA. 1991;88:1656–1660. doi: 10.1073/pnas.88.5.1656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furui T, Imai A, Takagi H, Horibe S, Fuseya T, Tamaya T. Phosphotyrosine phosphatase activity in membranes from endometrial carcinoma. Oncology Reports. 1995;2:1055–1057. doi: 10.3892/or.2.6.1055. [DOI] [PubMed] [Google Scholar]
- Imai A, Takagi H, Furui T, Horibe S, Fuseya T, Tamaya T. Evidence for coupling of phosphotyrosine phosphatase to gonadotropin-releasing hormone receptor in ovarian carcinoma membrane. Cancer. 1996;77:132–137. doi: 10.1002/(SICI)1097-0142(19960101)77:1<132::AID-CNCR22>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- Imai A, Takagi H, Horibe S, Fuseya T, Tamaya T. Coupling of gonadotropin releasing hormone receptor to Gi protein in human reproductive tract tumors. J Clin Endocrinol Metab. 1996;81:3249–3253. doi: 10.1210/jc.81.9.3249. [DOI] [PubMed] [Google Scholar]
- Gründker C, Völker P, Schulz KD, Emons G. Luteinizing Hormone-releasing Hormone (LHRH) Agonist Triptorelin and Antagonist Cetrorelix inhibit EGF-induced c-fos Expression in Human Gynecological Cancers. Gynecol Oncol. 2000;78:194–202. doi: 10.1006/gyno.2000.5863. [DOI] [PubMed] [Google Scholar]
- Moretti RM, Montagnani-Marelli M, Dondi D, Poletti A, Martini L, Motta M, Limonta P. Luteinizing hormone-releasing hormone agonists interfere with the stimulatory actions of epidermal growth factor in human prostatic cancer cell lines, LNCaP and DU 145. J Clin Endocrinol Metab. 1996;81:3930–3937. doi: 10.1210/jc.81.11.3930. [DOI] [PubMed] [Google Scholar]
- Shirahige Y, Cook C, Pinski J, Halmos G, Nair R, Schally AV. Treatment with luteinizing hormone-releasing hormone antagonist SB-75 decreases levels of epidermal growth factor receptor and its mRNA in OV-1063 human epithelial ovarian cancer xenografts in nude mice. Int J Oncol. 1994;5:1031–1035. doi: 10.3892/ijo.5.5.1031. [DOI] [PubMed] [Google Scholar]
- Kéri G, Balogh A, Szöke B, Téplan J, Csika O. Gonadotropin-releasing hormone analogues inhibit cell proliferation and activate signal transduction pathways in MDA-MB-231 human breast cancer cell lines. Tumour Biol. 1991;12:61–67. [PubMed] [Google Scholar]
- Liebow C, Lee MT, Kramer AR, Schally AV. Regulation of luteinizing hormone-releasing hormone receptor binding by heterologous receptor-stimulated tyrosine phosphorylation. Proc Natl Acad Sci USA. 1991;88:2244–2248. doi: 10.1073/pnas.88.6.2244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hershkovitz E, Marbach M, Bosin M, Levy J, Roberts C, Jr, Le Roith D, Schally AV, Sharoni Y. Luteinizing hormone-releasing hormone antagonists interfere with autocrine and paracrine growth stimulation of MCF-7 mammary cancer cells by insulin like growth factors. J Clin Endocrinol Metab. 1993;77:963–968. doi: 10.1210/jc.77.4.963. [DOI] [PubMed] [Google Scholar]
- Imai A, Horibe S, Takagi A, Ohno T, Tamaya T. Frequent expression of Fas in gonadotropin-releasing hormone receptor-bearing tumors. Eur J Obstet Gynecol Reprod Biol. 1997;74:73–78. doi: 10.1016/S0301-2115(97)02750-4. [DOI] [PubMed] [Google Scholar]
- Imai A, Takagi A, Horibe S, Takagi H, Tamaya T. Evidence for tight coupling of gonadotropin-releasing hormone receptor to stimulate Fas ligand expression in reproductive tumors: possible mechanism for hormonal control of apoptotic cell death. J Clin Endocrinol Metab. 1998;83:127–431. doi: 10.1210/jcem.83.2.4530. [DOI] [PubMed] [Google Scholar]
- Imai A, Takagi A, Horibe S, Takagi H, Tamaya T. Fas and Fas-ligand system may mediate antiproliferative activity of gonadotropin-releasing hormone receptor in endometrial cancer cells. Int J Oncol. 1998;13:97–100. doi: 10.3892/ijo.13.1.97. [DOI] [PubMed] [Google Scholar]
- Gründker C, Schulz K, Günthert AR, Emons G. Luteinizing hormone-releasing hormone induces nuclear factor kappaB-activation and inhibits apoptosis in ovarian cancer cells. J Clin Endocrinol Metab. 2000;85:3815–3820. doi: 10.1210/jc.85.10.3815. [DOI] [PubMed] [Google Scholar]
- Gründker C, Schlotawa L, Viereck V, Emons G. Protein kinase C (PKC)-independent stimulation of activator protein-1 (AP-1) and c-Jun N-terminal kinase (JNK) activity in human endometrial cancer cells by luteinizing hormone-releasing hormone (LHRH) agonist Triptorelin. Eur J Endocrinol. 2001;145:651–658. doi: 10.1530/eje.0.1450651. [DOI] [PubMed] [Google Scholar]
- Yamauchi J, Itoh H, Shinoura H, Miyamoto Y, Hirasawa A, Kaziro Y, Tsujimoto G. Involvement of c-Jun N-terminal kinase and p38 mitogen-activated protein kinase in α1B-adrenergic receptor/Gαq-induced inhibition of cell proliferation. Biochem Biophys Res Commun. 2001;281:1019–1023. doi: 10.1006/bbrc.2001.4472. [DOI] [PubMed] [Google Scholar]
- Bigsby RM, Li A. Differentially regulated immediate early genes in the rat uterus. Endocrinology. 1994;134:1820–1826. doi: 10.1210/en.134.4.1820. [DOI] [PubMed] [Google Scholar]
- Seppänen M, Lin L, Punnonen J, Grénman S, Punnonen R, Vihko K. Regulation of UT-OC-3 ovarian carcinoma cells by cytokines: inhibitory effects on cell proliferation and activation of transcription factors AP-1 and NF-kappaB. Eur J Endocrinol. 2000;142:393–401. doi: 10.1530/eje.0.1420393. [DOI] [PubMed] [Google Scholar]
- Günthert AR, Gründker C, Hollmann K, Emons G. Luteinizing hormone-releasing hormone induces JunD-DNA binding and extends cell cycle in human ovarian cancer cells. Biochem Biophys Res Commun. 2002;294:11–15. doi: 10.1016/S0006-291X(02)00427-8. [DOI] [PubMed] [Google Scholar]
- Miyamoto K, Hasegawa Y, Nomura M, Igarashi M, Kangawa K, Matsuo H. Identification of the second gonadotropin-releasing hormone in chicken hypothalamus: evidence that gonadotropin secretion is probably controlled by two distinct gonadotropin-releasing hormones in avian species. Proc Natl Acad Sci USA. 1984;81:3874–3878. doi: 10.1073/pnas.81.12.3874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sower SA, Chiang YC, Lovas S, Conlon JM. Primary structure and biological activity of a third gonadotropin-releasing hormone from lamprey brain. Endocrinology. 1993;132:1125–1131. doi: 10.1210/en.132.3.1125. [DOI] [PubMed] [Google Scholar]
- White RB, Eisen JA, Kasten TL, Fernald RD. Second gene for gonadotropin-releasing hormone in humans. Proc Natlional Acad Sci USA. 1998;95:305–309. doi: 10.1073/pnas.95.1.305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urbanski HF, White RB, Fernald RD, Kahoma SG, Garyfallou VT, Densmore VS. Regional expression of mRNA encoding a second form of gonadotropin-releasing hormone in the macaque brain. Endocrinology. 1999;140:1945–1948. doi: 10.1210/en.140.4.1945. [DOI] [PubMed] [Google Scholar]
- Kang SK, Tai CJ, Nathwani PS, Leung PC. Differential regulation of two forms of gonadotropin-releasing hormone messenger ribonucleic acid in human granulosa-luteal cells. Endocrinology. 2001;142:182–192. doi: 10.1210/en.142.1.182. [DOI] [PubMed] [Google Scholar]
- Millar RP, Lowe S, Conklin D, Pawson A, Maudsley S, Troskie B, Ott T, Millar M, Lincoln G, Sellar R, Faurholm B, Scobie G, Kuestner R, Terasawa E, Katz A. A novel mammalian receptor for the evolutionarily conserved type II GnRH. Proc Natl Acad Sci U S A. 2001;98:9636–9641. doi: 10.1073/pnas.141048498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neill JD, Duck LW, Sellers JC, Musgrove LC. A gonadotropin-releasing hormone (GnRH) receptor specific for GnRH II in primates. Biochem Biophys Res Commun. 2001;282:1012–1018. doi: 10.1006/bbrc.2001.4678. [DOI] [PubMed] [Google Scholar]
- Gründker C, Günthert AR, Millar RP, Emons G. Expression of gonadotropin-releasing hormone II (GnRH-II) receptor in human endometrial and ovarian cancer cells and effects of GnRH-II on tumor cell proliferation. J Clin Endocrinol Metab. 2002;87:1427–1430. doi: 10.1210/jc.87.3.1427. [DOI] [PubMed] [Google Scholar]
- Morgan K, Conklin D, Pawson AJ, Sellar R, Ott TR, Millar RP. A transcriptionally active human type II gonadotropin-releasing hormone receptor gene homolog overlaps two genes in the antisense orientation on chromosome 1q.12. Endocrinology. 2003;144:423–436. doi: 10.1210/en.2002-220622. [DOI] [PubMed] [Google Scholar]