Abstract
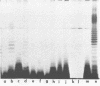
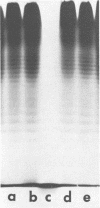
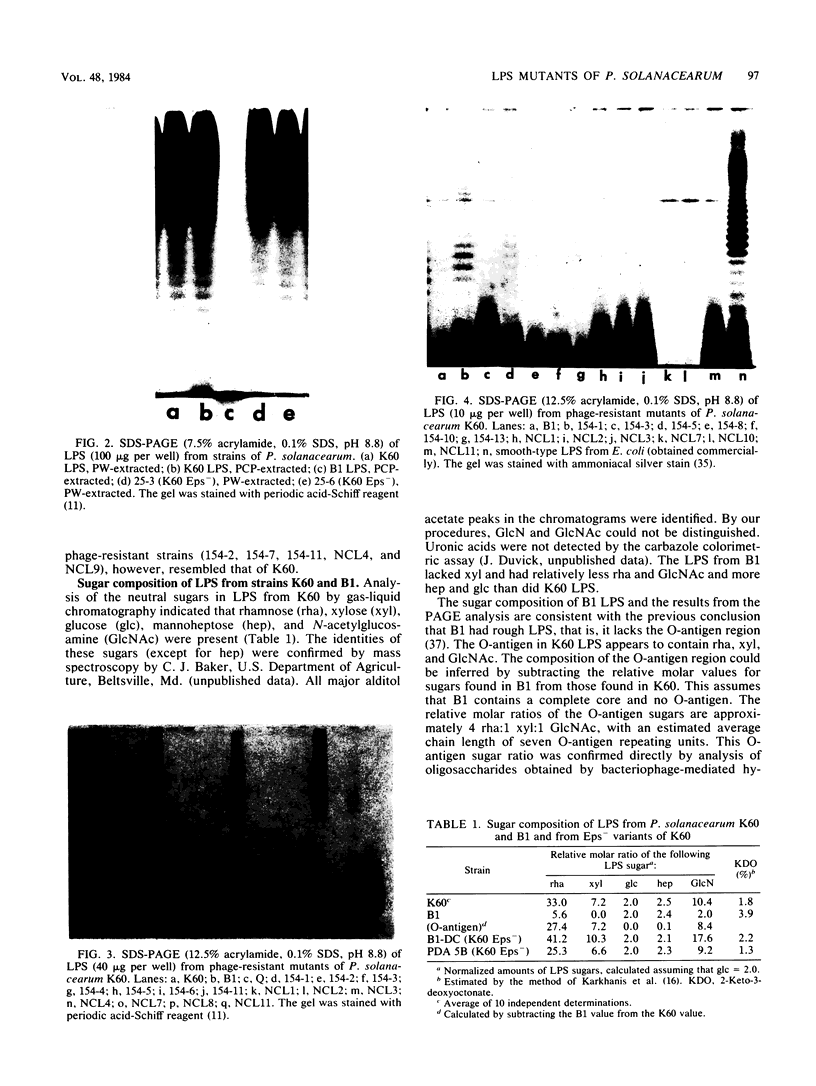
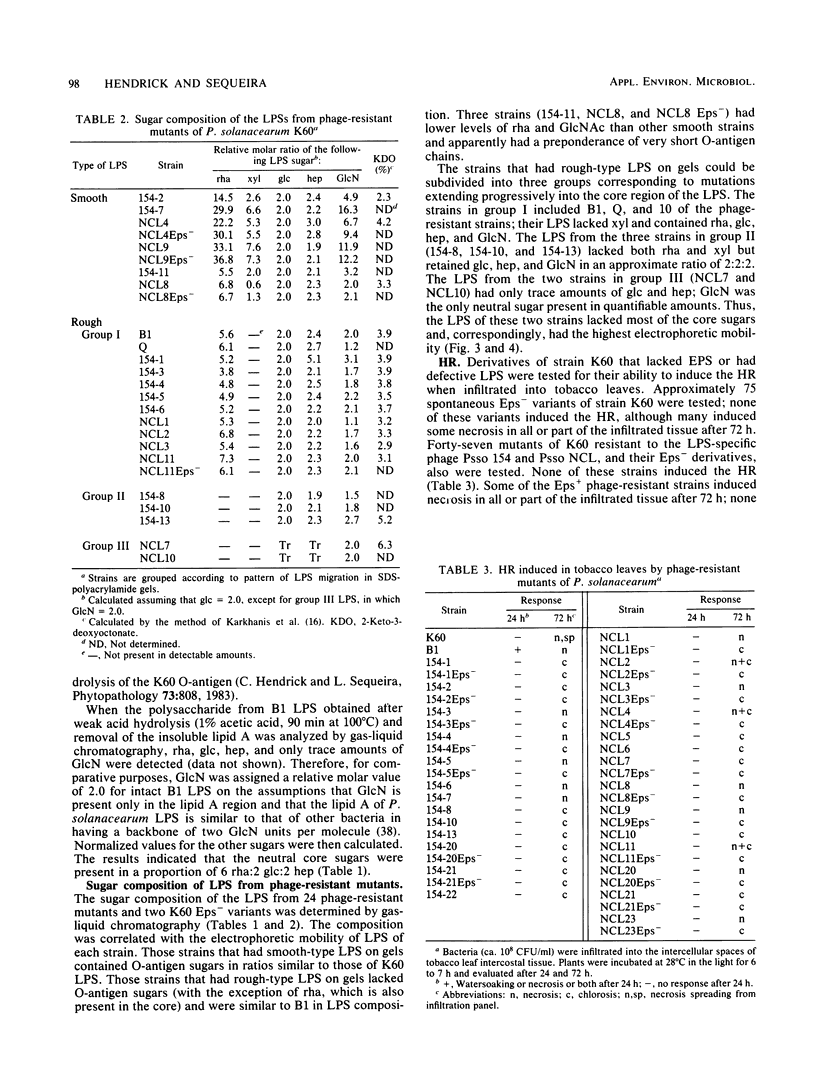
Lipopolysaccharide (LPS)-defective mutants of Pseudomonas solanacearum were used to test the hypothesis that differences in LPS structure are associated with the ability or inability of different strains to induce a hypersensitive response (HR) in tobacco. To obtain these mutants, LPS-specific bacteriophage of P. solanacearum were isolated and used to select phage-resistant mutants of the virulent, non-HR-inducing strain K60. The LPS of 24 of these mutants was purified and compared with that of K60 and its HR-inducing variant, B1. Upon sodium dodecyl sulfate-polyacrylamide gel electrophoresis, LPS from K60 and other smooth strains separated into many evenly spaced bands that migrated slowly, whereas LPS from B1 and most phage-resistant strains separated into one to three bands that migrated rapidly. Carbohydrate analysis showed that the LPS of the phage-resistant strains lacked O-antigen sugars (rhamnose, xylose, and N-acetylglucosamine) and could be grouped into (i) those that had all core sugars (rhamnose, glucose, heptose, and 2-keto-3-deoxyoctonate), (ii) those that had no core rhamnose, and (iii) those that lacked all core sugars except for 2-keto-3-deoxyoctonate. The LPS composition of 10 of the rough, phage-resistant mutants was similar to that of the HR-inducing strain, B1, yet none of them induced the HR. Only 2 of 13 mutant strains tested caused wilting of tobacco, and these had rough LPS but produced large amounts of extracellular polysaccharide, unlike most LPS-defective mutants. The evidence did not support the hypothesis that the initial interaction between rough LPS and tobacco cell walls is the determining factor in HR initiation.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ackermann H. W., Audurier A., Berthiaume L., Jones L. A., Mayo J. A., Vidaver A. K. Guidelines for bacteriophage characterization. Adv Virus Res. 1978;23:1–24. doi: 10.1016/s0065-3527(08)60096-2. [DOI] [PubMed] [Google Scholar]
- Ayers A. R., Ayers S. B., Goodman R. N. Extracellular Polysaccharide of Erwinia amylovora: a Correlation with Virulence. Appl Environ Microbiol. 1979 Oct;38(4):659–666. doi: 10.1128/aem.38.4.659-666.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradley D. E. Ultrastructure of bacteriophage and bacteriocins. Bacteriol Rev. 1967 Dec;31(4):230–314. doi: 10.1128/br.31.4.230-314.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradshaw-Rouse J. J., Whatley M. H., Coplin D. L., Woods A., Sequeira L., Kelman A. Agglutination of Erwinia stewartii Strains with a Corn Agglutinin: Correlation with Extracellular Polysaccharide Production and Pathogenicity. Appl Environ Microbiol. 1981 Aug;42(2):344–350. doi: 10.1128/aem.42.2.344-350.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fairbanks G., Steck T. L., Wallach D. F. Electrophoretic analysis of the major polypeptides of the human erythrocyte membrane. Biochemistry. 1971 Jun 22;10(13):2606–2617. doi: 10.1021/bi00789a030. [DOI] [PubMed] [Google Scholar]
- Galanos C., Lüderitz O., Westphal O. A new method for the extraction of R lipopolysaccharides. Eur J Biochem. 1969 Jun;9(2):245–249. doi: 10.1111/j.1432-1033.1969.tb00601.x. [DOI] [PubMed] [Google Scholar]
- Goldman R. C., Leive L. Heterogeneity of antigenic-side-chain length in lipopolysaccharide from Escherichia coli 0111 and Salmonella typhimurium LT2. Eur J Biochem. 1980;107(1):145–153. doi: 10.1111/j.1432-1033.1980.tb04635.x. [DOI] [PubMed] [Google Scholar]
- Hitchcock P. J., Brown T. M. Morphological heterogeneity among Salmonella lipopolysaccharide chemotypes in silver-stained polyacrylamide gels. J Bacteriol. 1983 Apr;154(1):269–277. doi: 10.1128/jb.154.1.269-277.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hrabak E. M., Urbano M. R., Dazzo F. B. Growth-phase-dependent immunodeterminants of Rhizobium trifolii lipopolysaccharide which bind trifoliin A, a white clover lectin. J Bacteriol. 1981 Nov;148(2):697–711. doi: 10.1128/jb.148.2.697-711.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KLEMENT Z. RAPID DETECTION OF THE PATHOGENICITY OF PHYTOPATHOGENIC PSEUDOMONADS. Nature. 1963 Jul 20;199:299–300. doi: 10.1038/199299b0. [DOI] [PubMed] [Google Scholar]
- Karkhanis Y. D., Zeltner J. Y., Jackson J. J., Carlo D. J. A new and improved microassay to determine 2-keto-3-deoxyoctonate in lipopolysaccharide of Gram-negative bacteria. Anal Biochem. 1978 Apr;85(2):595–601. doi: 10.1016/0003-2697(78)90260-9. [DOI] [PubMed] [Google Scholar]
- Kelman A., Hruschka J. The role of motility and aerotaxis in the selective increase of avirulent bacteria in still broth cultures of Pseudomonas solanacearum. J Gen Microbiol. 1973 May;76(1):177–188. doi: 10.1099/00221287-76-1-177. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Leach J. E., Cantrell M. A., Sequeira L. Hydroxyproline-rich bacterial agglutinin from potato : extraction, purification, and characterization. Plant Physiol. 1982 Nov;70(5):1353–1358. doi: 10.1104/pp.70.5.1353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munford R. S., Hall C. L., Rick P. D. Size heterogeneity of Salmonella typhimurium lipopolysaccharides in outer membranes and culture supernatant membrane fragments. J Bacteriol. 1980 Nov;144(2):630–640. doi: 10.1128/jb.144.2.630-640.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palva E. T., Mäkelä P. H. Lipopolysaccharide heterogeneity in Salmonella typhimurium analyzed by sodium dodecyl sulfate polyacrylamide gel electrophoresis. Eur J Biochem. 1980;107(1):137–143. doi: 10.1111/j.1432-1033.1980.tb04634.x. [DOI] [PubMed] [Google Scholar]
- Quirk A. V., Sletten A., Hignett R. C. Properties of phage-receptor lipopolysaccharide from Pseudomonas morsprunorum. J Gen Microbiol. 1976 Oct;96(2):375–381. doi: 10.1099/00221287-96-2-375. [DOI] [PubMed] [Google Scholar]
- Tsai C. M., Frasch C. E. A sensitive silver stain for detecting lipopolysaccharides in polyacrylamide gels. Anal Biochem. 1982 Jan 1;119(1):115–119. doi: 10.1016/0003-2697(82)90673-x. [DOI] [PubMed] [Google Scholar]
- Whatley M. H., Hunter N., Cantrell M. A., Hendrick C., Keegstra K., Sequeira L. Lipopolysaccharide Composition of the Wilt Pathogen, Pseudomonas solanacearum: CORRELATION WITH THE HYPERSENSITIVE RESPONSE IN TOBACCO. Plant Physiol. 1980 Mar;65(3):557–559. doi: 10.1104/pp.65.3.557. [DOI] [PMC free article] [PubMed] [Google Scholar]