Abstract
We examined a panel of Sindbis virus mutants containing defined mutations in the 5' nontranslated region of the genome RNA, in the 3' nontranslated region, or in both for their growth in cultured cells and virulence in newborn mice. In cultured cells, these viruses all had defects in RNA synthesis and displayed a wide range of growth rates. The growth properties of the mutants were often very different in mouse cells from those in chicken cells or in mosquito cells. We hypothesize that host factors, presumably proteins, interact with these nontranslated regions to promote viral replication and that the mammalian protein and the chicken or mosquito protein are sufficiently divergent that alterations in the viral RNA sequence can affect the interactions with these different host proteins in different ways. Some of the mutants were temperature sensitive for plaque formation, whereas one mutant was slightly cold sensitive in its growth in chicken cells. Upon inoculation into mice, viruses that grew well in cultured mouse cells retained their virulence, but mice that succumbed usually had extended survival times. One virulent mutant that grew slightly less well in cultured mouse cells than did the parental virus produced eight times as much virus in mouse brain following intracerebral inoculation, suggesting that changes in these regulatory regions may have tissue-specific as well as host-specific effects. Viruses that were severely crippled in their growth in mouse cells in culture were usually, but not always, attenuated in their virulence. In particular, temperature sensitivity was correlated with attenuation. The effect of two mutations was found to be cumulative, and double mutants that contained mutations in both the 5' and 3' nontranslated regions were more attenuated than was either single mutant. Three of four double mutants tested were severely crippled for virus production in cultured cells and were avirulent for mice, even when inoculated intracerebrally.
Full text
PDF

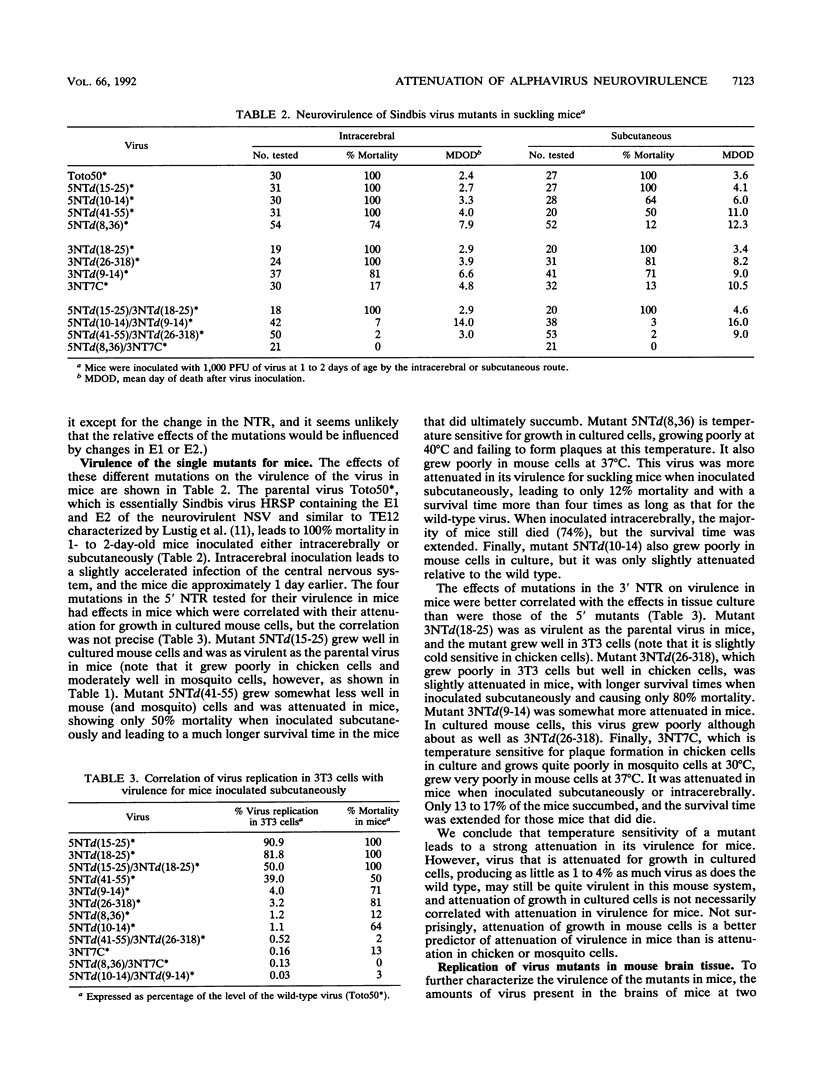


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Cohen J. I., Rosenblum B., Feinstone S. M., Ticehurst J., Purcell R. H. Attenuation and cell culture adaptation of hepatitis A virus (HAV): a genetic analysis with HAV cDNA. J Virol. 1989 Dec;63(12):5364–5370. doi: 10.1128/jvi.63.12.5364-5370.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen J. I., Rosenblum B., Ticehurst J. R., Daemer R. J., Feinstone S. M., Purcell R. H. Complete nucleotide sequence of an attenuated hepatitis A virus: comparison with wild-type virus. Proc Natl Acad Sci U S A. 1987 Apr;84(8):2497–2501. doi: 10.1073/pnas.84.8.2497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans D. M., Dunn G., Minor P. D., Schild G. C., Cann A. J., Stanway G., Almond J. W., Currey K., Maizel J. V., Jr Increased neurovirulence associated with a single nucleotide change in a noncoding region of the Sabin type 3 poliovaccine genome. Nature. 1985 Apr 11;314(6011):548–550. doi: 10.1038/314548a0. [DOI] [PubMed] [Google Scholar]
- Griffin D. E., Johnson R. T. Role of the immune response in recovery from Sindbis virus encephalitis in mice. J Immunol. 1977 Mar;118(3):1070–1075. [PubMed] [Google Scholar]
- Iizuka N., Kohara M., Hagino-Yamagishi K., Abe S., Komatsu T., Tago K., Arita M., Nomoto A. Construction of less neurovirulent polioviruses by introducing deletions into the 5' noncoding sequence of the genome. J Virol. 1989 Dec;63(12):5354–5363. doi: 10.1128/jvi.63.12.5354-5363.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson R. T., McFarland H. F., Levy S. E. Age-dependent resistance to viral encephalitis: studies of infections due to Sindbis virus in mice. J Infect Dis. 1972 Mar;125(3):257–262. doi: 10.1093/infdis/125.3.257. [DOI] [PubMed] [Google Scholar]
- Kuhn R. J., Hong Z., Strauss J. H. Mutagenesis of the 3' nontranslated region of Sindbis virus RNA. J Virol. 1990 Apr;64(4):1465–1476. doi: 10.1128/jvi.64.4.1465-1476.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lustig S., Jackson A. C., Hahn C. S., Griffin D. E., Strauss E. G., Strauss J. H. Molecular basis of Sindbis virus neurovirulence in mice. J Virol. 1988 Jul;62(7):2329–2336. doi: 10.1128/jvi.62.7.2329-2336.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakhasi H. L., Cao X. Q., Rouault T. A., Liu T. Y. Specific binding of host cell proteins to the 3'-terminal stem-loop structure of rubella virus negative-strand RNA. J Virol. 1991 Nov;65(11):5961–5967. doi: 10.1128/jvi.65.11.5961-5967.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakhasi H. L., Rouault T. A., Haile D. J., Liu T. Y., Klausner R. D. Specific high-affinity binding of host cell proteins to the 3' region of rubella virus RNA. New Biol. 1990 Mar;2(3):255–264. [PubMed] [Google Scholar]
- Niesters H. G., Strauss J. H. Defined mutations in the 5' nontranslated sequence of Sindbis virus RNA. J Virol. 1990 Sep;64(9):4162–4168. doi: 10.1128/jvi.64.9.4162-4168.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niesters H. G., Strauss J. H. Mutagenesis of the conserved 51-nucleotide region of Sindbis virus. J Virol. 1990 Apr;64(4):1639–1647. doi: 10.1128/jvi.64.4.1639-1647.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niklasson B., Espmark A., LeDuc J. W., Gargan T. P., Ennis W. A., Tesh R. B., Main A. J., Jr Association of a Sindbis-like virus with Ockelbo disease in Sweden. Am J Trop Med Hyg. 1984 Nov;33(6):1212–1217. doi: 10.4269/ajtmh.1984.33.1212. [DOI] [PubMed] [Google Scholar]
- Omata T., Kohara M., Kuge S., Komatsu T., Abe S., Semler B. L., Kameda A., Itoh H., Arita M., Wimmer E. Genetic analysis of the attenuation phenotype of poliovirus type 1. J Virol. 1986 May;58(2):348–358. doi: 10.1128/jvi.58.2.348-358.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardigon N., Strauss J. H. Cellular proteins bind to the 3' end of Sindbis virus minus-strand RNA. J Virol. 1992 Feb;66(2):1007–1015. doi: 10.1128/jvi.66.2.1007-1015.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rice C. M., Levis R., Strauss J. H., Huang H. V. Production of infectious RNA transcripts from Sindbis virus cDNA clones: mapping of lethal mutations, rescue of a temperature-sensitive marker, and in vitro mutagenesis to generate defined mutants. J Virol. 1987 Dec;61(12):3809–3819. doi: 10.1128/jvi.61.12.3809-3819.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirako Y., Niklasson B., Dalrymple J. M., Strauss E. G., Strauss J. H. Structure of the Ockelbo virus genome and its relationship to other Sindbis viruses. Virology. 1991 Jun;182(2):753–764. doi: 10.1016/0042-6822(91)90616-j. [DOI] [PubMed] [Google Scholar]
- Toyoda H., Kohara M., Kataoka Y., Suganuma T., Omata T., Imura N., Nomoto A. Complete nucleotide sequences of all three poliovirus serotype genomes. Implication for genetic relationship, gene function and antigenic determinants. J Mol Biol. 1984 Apr 25;174(4):561–585. doi: 10.1016/0022-2836(84)90084-6. [DOI] [PubMed] [Google Scholar]
- White B. A., Bancroft F. C. Cytoplasmic dot hybridization. Simple analysis of relative mRNA levels in multiple small cell or tissue samples. J Biol Chem. 1982 Aug 10;257(15):8569–8572. [PubMed] [Google Scholar]