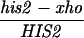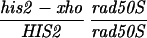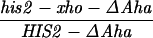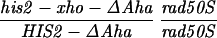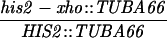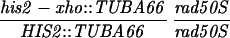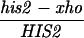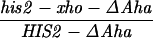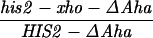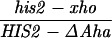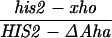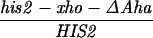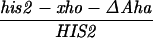Abstract
Double strand breaks (DSBs) have been found at several meiotic recombination hot spots in Saccharomyces cerevisiae; more global studies have found that they occur at many places along several yeast chromosomes during meiosis. Indeed, the number of breaks found is consistent with the number of recombination events predicted from the genetic map. We have previously demonstrated that the HIS2 gene is a recombination hot spot, exhibiting a high frequency of gene conversion and associated crossing over. This paper shows that DSBs occur in meiosis at a site in the coding region and at a site downstream of the HIS2 gene and that the DSBs are dependent upon genes required for recombination. The frequency of DSBs at HIS2 increases when the gene conversion frequency is increased by alterations in the DNA around HIS2, and vice versa. A deletion that increases both DSBs and conversion can stimulate both when heterozygous; that is, it is semidominant and acts to stimulate DSBs in trans. These data are consistent with the view that homologous chromosomes associate with each other before the formation of the DSBs.
Keywords: meiosis, chromosomes, gene conversion, pairing
The mechanism whereby homologous chromosomes find each other during meiosis and initiate recombination has been the source of considerable speculation and experimentation. Several models have been proposed to account for the genetic and, more recently, molecular data that have been obtained about meiotic recombination. The classical Holliday model (1) suggested that nicks are made in strands of the same polarity in two homologs, after which the broken strands are exchanged from one duplex to the other. The Meselson–Radding model (2) begins recombination with a single strand nick on one homolog, after which one end of the broken strand invades the homologous duplex. More recently, the double strand break (DSB) model for recombination was proposed by Szostak et al. (3); in this scheme, both ends of the broken duplex invade the homolog to initiate recombination. In the latter two models, one can imagine that the mechanism whereby homologs find each other involves recA-like (4) synaptase-assisted searching by the single strands created at the initiating breaks or nicks.
Since the publication of the DSB model, experiments have been done that demonstrate that DSBs occur during meiosis in yeast and that they are associated with recombination. These experiments may be sorted into two groups: (i) experiments that examine recombination hot spots both for recombination and DSBs, and (ii) experiments that look at the more global distribution of DSBs along entire chromosomes. Within the first class of experiments, Sun et al. (5) first demonstrated that a meiotic DSB occurs at the ARG4 locus. The location of the DSB was in the DNA sequences that had been shown to be necessary (6) for conversion at ARG4. Furthermore, the time at which the breaks appeared was consistent with the time at which commitment to recombination occurred. In these early experiments, the DSBs were difficult to detect, in part because they were transient, as would be expected for an intermediate in the recombination process. This problem was solved by the discovery of a particular allele of the recombination gene RAD50 (7). This allele (rad50S) allows DSBs to form, but prevents processing; thus, DSBs accumulate in meiosis, allowing them to be quantified. Cao, Alani, and Kleckner (8) demonstrated that DSBs occurring at an artificial hot spot (created by the fusion of the HIS4 and LEU2 genes) had the same general kinetics of appearance as DSBs at ARG4. In addition, DSB loci were eliminated by null mutations in either of the early recombination genes SPO11 or RAD50 (8). Thus, the DSBs occurring at both the ARG4 and HIS4::LEU2 hot spots exhibited the properties expected if they were involved in the initiation of recombination as predicted by the DSB model (3). More recently, meiotic DSBs have been found at the HIS4 gene in strains that exhibit very high frequencies of recombination at this locus (9). Furthermore, these authors have shown a linear relationship between the amount of conversion and the amount of DSBs, leading them to suggest that DSBs initiate recombination at HIS4.
Not only did these known recombination hot spots have meiotic DSBs, but several studies have shown that DSBs occur in meiosis along many Saccharomyces cerevisiae chromosomes (10–12). Pulse field gel analysis demonstrates that breaks occur in meiosis at discrete locations. The numbers and locations of DSBs were generally consistent with known genetic map distances, compatible with the hypothesis that DSBs are involved in meiotic recombination.
We have been studying meiotic recombination at the HIS2 gene in S. cerevisiae. This gene has a gradient of gene conversion that is high at the 3′ end and low at the 5′ end of the gene (13, 14); this polarity is opposite to that found in the well-studied ARG4 and HIS4 genes (6, 9). Consistent with the 3′ to 5′ gradient, the promoter region of the HIS2 gene is not required for the high frequency of recombination observed (13, 14). In fact, at least some of the sequences necessary for high frequencies of conversion at HIS2 appear to be located some 700 bp downstream of the coding region (14). The experiments in this paper demonstrate that there are meiosis-specific DSBs at HIS2 and determine their properties.
MATERIALS AND METHODS
Yeast and Bacterial Strains.
All yeast strains used in these experiments are derived from RM96-15AX (MATa his2-xho ura3-13 met2-1 trp1-1) and RM182-55C (MATα cly3 cdc14-1 lys2-1 ade5 ade2-1 can1r). The diploid made by crossing RM96-15AX and RM182-55C is RM169. Mutations in the HIS2 gene or other alterations (e.g., rad50S) were introduced by transformation as described below. The genotype of each strain at HIS2 (and at RAD50, if appropriate) is shown in the appropriate table or figure. Two types of diploids were used. “Isogenic” diploids were made by directly crossing appropriate transformants of RM96-15AX and RM182-55C. “Congenic” diploids were made as follows. (i) A transformant of either RM96-15AX or RM182-55C (called P1) was crossed with the other parent (P2). (ii) A segregant from this cross with the appropriate genotype was then backcrossed at least three times with the P1 parent. (iii) A final segregant (which should be at least 94% identical to the P1 parent) was then crossed to the appropriate P2 parent to make the diploid. Each diploid used is denoted in the appropriate figure or table as isogenic or congenic. All tetrads dissected to determine the frequency of gene conversion at the HIS2 locus were checked for false tetrads by examining the segregation of at least five other loci. The bacterial strain DH5α (Bethesda Research Laboratories) was used for all DNA manipulations.
Mutations in the HIS2 Gene.
All mutations in the HIS2 region were made in vitro and placed in yeast by two-step gene replacement (15). The new mutation (his2-hpa2) used in this paper was made using oligonucleotide-directed mutagenesis as detailed in ref. 14. his2-hpa2 is the second mutation we created at this HpaI site, hence the -hpa2 extension. The his2-hpa2 mutation is a single-base pair substitution (A → T) at position +947, which ablates the HpaI site and creates a stop codon. The his2-2 mutation used to monitor heteroallelic recombination is located at +290 (14). A complete description of the construction of all other his2 mutations, the ΔAha deletion, and the TUBA66 insertion may be found in ref. 14. All mutations and alterations created in vitro and placed in yeast were confirmed by Southern blot analysis. A map of relevant loci is shown in Fig. 1.
Figure 1.
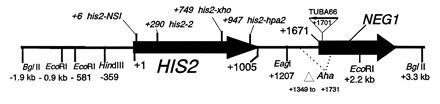
Map of the HIS2 gene and surrounding region. Features shown are those used in the experiments described in this paper. All locations are numbered relative to the A of the first ATG of the HIS2 coding region (designated +1). The arrow indicates the direction of transcription. The location of the downstream gene (NEG1) is also shown.
Analysis of DSBs at HIS2.
To study DSBs in and around the HIS2 locus, the separation of function mutation rad50S-KI81 (referred to henceforth as rad50S) was introduced by one-step gene replacement (15) as described in ref. 16 using the plasmid pNKY349 (provided by Nancy Kleckner, Harvard University). The URA3 marker is inserted downstream of the RAD50 coding region. All transformants were verified genetically and by Southern analysis as in ref. 16. To obtain cells in meiosis, diploids were grown to a concentration of 2 × 107 cells per ml in YPA, washed, and concentrated 2-fold in sporulation medium at 30°C. Media are defined in refs. 14 and 16. Samples (20 ml) were taken at the indicated times, and the cells were mixed with equal volumes of 95% ethanol at 4°C and 50 mM EDTA and stored at −20°C. To verify the progression of the cells in meiosis, aliquots were taken at each timepoint and counted for sporulation and 4′,6-diamidino-2-phenylindole (DAPI)-stained (7) to check the fraction of multinucleate cells. DNA was made as described in ref. 8. Gels were run at 50 V for 33–38 hr in TBE buffer in a 0.8% agarose gel at 4°C, as described (16, 17). All imaging and quantitation was done using a Molecular Dynamics PhosphorImager model 445SI as per instructions of the manufacturer.
RESULTS
The Gene Conversion Gradient at the HIS2 Locus.
All crosses in this paper were examined for five markers that were heterozygous, in addition to HIS2 (see Materials and Methods). There were no significant differences in the frequencies of conversion of the other markers in diploids with or without alterations in or near the HIS2 gene (data not shown). We have previously demonstrated that the polarity gradient of conversion in the HIS2 gene is 3′ → 5′ (13, 14). For example, a single base mutation that created a NsiI site (+6) had a gene conversion frequency of 4.9%, and a 4-bp insertion mutation at the XhoI site (+749) converted with a frequency of 14.1% (Fig. 1). Interestingly, an 8-bp linker insertion mutation located further 3′ at the HpaI site (+947) converted at a lower frequency of 11.7% (14). In previous work (14), we suggested that the lower frequency observed at the HpaI site was due to marker effects of the insertion. An alternative possibility was that the gradient reached its peak within the HIS2 gene near the XhoI site and declined toward the HpaI site. We constructed a single base change mutation in the HpaI site (see Materials and Methods); the frequency of conversion is 20.7% (62 conversions in 299 tetrads). Thus, the frequency of conversion appears to increase all the way across the HIS2 gene (see Discussion).
DSBs at the HIS2 Locus.
To determine whether DSBs occur during meiosis in the HIS2 region, we constructed rad50S strains (8). The data in Fig. 2 indicate that two DSBs occur in the HIS2 region, and they appear with the kinetics that would be expected if they were associated with recombination. The location of the breaks is consistent with the gene conversion polarity gradient (see Discussion). We have labeled the break in the coding region (at position 650 ± 30) as break C and the one 200-bp downstream of the coding region (at position 1200 ± 30) as break B. To compare the time at which DSBs appear to the time of commitment to recombination, we measured the appearance of His+ recombinants in a RAD50 derivative of the rad50S diploid (Fig. 3). From these results, it appears that the DSBs at HIS2 appear at a time consistent with their being involved in meiotic recombination. However, we note that the experiments have to be done in two different strains (rad50S and RAD50), and it is possible that the timing of their progression through meiosis varies. Finally, we examined the effect of a mutation in an early Rec gene (REC104) on the DSBs. A rec104 mutation eliminates all meiotic recombination (18) and, as Fig. 2 demonstrates, DSBs at HIS2 cannot be detected in the rec104 mutant. We have obtained the same results with rec102 and rec114 mutations (data not shown). We conclude that the DSBs found at the HIS2 locus have many of the properties of meiotic DSBs found at other S. cerevisiae recombination hot spots.
Figure 2.
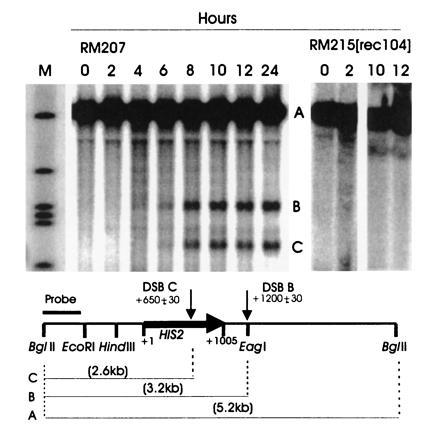
Southern analysis of DSBs during meiosis at the HIS2 locus. Cells were removed from sporulation medium at the times indicated, and DNA was made, digested with BglII, and analyzed as described. The numbers above the lanes refer to the time in meiosis. The parental band A and the DSB bands B and C are illustrated in the figure. The probe was the BglII–EcoRI fragment shown on the figure. The diploid used for the wild-type lanes was RM207 (his2-xho/HIS2 rad50S/rad50S, congenic to RM169). On the right side of the figure are four time points from a congenic rec104-Δ1/rec104-Δ1 rad50S/rad50S diploid (RM215). The lane labeled M contains markers of sizes 5.2 kb, 3.9 kb, 3.2 kb, 3.0 kb, 2.9 kb, and 1.9 kb, from top to bottom. All lanes shown were run on the same gel.
Figure 3.
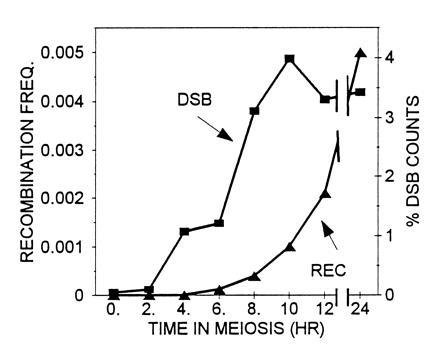
Time course of commitment to recombination and DSB formation in meiosis at the HIS2 locus. Recombination frequency is the number of His+ recombinants divided by the total number of colonies. The percent DSB counts is the amount of counts found in the DSB bands (at time t) divided by the total number of counts in the parental and DSB bands (at time t). The time course of DSBs (▪) was from one experiment using RM207 (his2-xho/HIS2 rad50S/rad50S, congenic to RM169; see also Table 1). The recombination values shown are the frequency of His+ prototrophs (▴) in a return to growth experiment in a congenic diploid of genotype his2-xho/his2-2 RAD50/RAD50.
Relationship of DSBs and Gene Conversion.
Our previous work has demonstrated that sequences necessary for the high frequency of conversion at HIS2 are located some 700 bp downstream of the coding region (i.e., downstream of position +1701; ref. 14). The separation between the sites where the breaks occur and sequences necessary for conversion provided an opportunity to ask how conversion at HIS2 is related to the DSBs observed. This separation allowed us to alter the frequency of conversion without altering the sequences near the DSB sites. In our previous studies of conversion at HIS2, we examined a deletion (ΔAha) of 382 bp from position +1349 to position +1731, and an insertion (TUBA66) of 5.5 kb at position +1701 (ref. 14; Fig. 1). ΔAha increases gene conversion of the his2-xho allele from 14.1% to 23.8%, and the TUBA66 insertion decreases conversion from 14.1% to 2.2% (Table 1). [We note that the effects of both the ΔAha and TUBA66 alterations are limited to the HIS2 locus; neither alteration significantly changed gene conversion at any of five other loci (data not shown).] The data in Fig. 4 show that the presence of ΔAha increases the amount of both DSB bands, but does not change their position or the kinetics of their appearance (see Discussion). For example, in the experiment shown in Fig. 4B, DSB B, located at +1200, is increased from the 10- and 12-hr average of 1.8% to 4.0%, and DSB C, at +630, is increased from 2.5% to 9.1%. In two independent experiments, we could not detect (<0.5%) any DSB bands in the HIS2 region in the diploid containing the TUBA66 insertion (data not shown). Thus, there is a correlation, although not a linear one, between the amount of conversion and the amount of DSBs at HIS2 observed during meiosis (Table 1; see Discussion).
Table 1.
Correlation between gene conversion levels and the amount of DSBs
| Gene conversion
|
DSB
measurements
|
||||||||
|---|---|---|---|---|---|---|---|---|---|
| Diploid | Genotype | No. of tetrads | GC content, % | Diploid | Genotype | No. of experiments | DSB, % | ||
| RM169 |
|
391 | 14.1 | RM207 |
|
5 | 4.3 ± 0.8 | ||
| RM198 |
|
319 | 23.8 | RM206 |
|
3 | 12.2 ± 0.6 | ||
| RM187 |
|
316 | 2.2 | RM216 |
|
2 | <0.5* | ||
The percent DSB = (the counts in the DSB bands divided by the total number of counts) × 100. RM207(rad50S/rad50S) is congenic to RM169; RM206 (rad50S/rad50S) is congenic to RM198; and RM216 (rad50S/rad50S) is congenic to RM187. DSBs were measured at 10 and 12 hr. The DSB values presented are the average of the number of experiments shown.
No bands at the normal DSB positions were detected in two independent experiments. Reconstruction experiments indicate that bands consisting of 0.5% of the total DNA could be detected.
Figure 4.
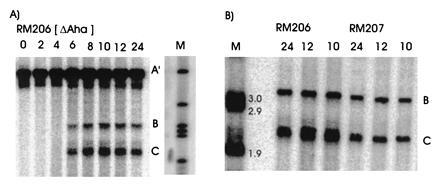
Southern blot analysis of DSBs in a diploid homozygous for ΔAha. (A) The diploid RM206 is homozygous for ΔAha and the rad50S mutation and is congenic to RM169. Cells were removed from sporulation medium at the time noted at the top of each lane. Markers (M) shown are 5.2 kb, 3.9 kb, 3.2 kb, 3.0 kb, 2.9 kb, and 1.9 kb, from top to bottom. Labels and the probe are as in Fig. 2 except band A′ is the parental band from RM206 and is 382 bp smaller than the normal (wild-type) band, which is designated as A (see Fig. 2). (B) Evidence that DSB bands from the ΔAha diploid are the same size as the normal diploid. An enlargement of the DSB bands is shown. DNA from the times in meiosis indicated were analyzed as in A. RM206 is homozygous for ΔAha; RM207 does not contain the deletion. The markers (lane M) in B are a subset of those used in A. [The total amount of counts in the bands A (not shown), B, and C (for RM207) was 1.3 × 106 at 10 hr, 1.4 × 106 at 12 hr, and 2.0 × 106 at 24 hr; the total amount of counts in bands A′ (not shown), B, and C (for RM206) was 1.4 × 106 at 10 hr, 1.2 × 106 at 12 hr, and 1.5 × 106 at 24 hr.]
Analysis of Chromosome Preference for DSBs in Strains Heterozygous for ΔAha.
The data above indicate that there is 1.7 times as much gene conversion and 2.8 times as many DSBs in diploids homozygous for ΔAha as there are in normal strains (Table 1). If the Aha deletion were capable of stimulating DSBs and recombination only on the chromosome in which it was present, then recombination in a strain heterozygous for ΔAha should have both a physical and a genetic consequence. First, more DSBs should occur on the homolog containing ΔAha (the “hot” chromosome) than on the chromosome without ΔAha (the “cold” chromosome). Second, the DSB model suggests that there should be a loss of parity in gene conversion; more recombination should initiate on the hot chromosome, leading to preferential loss of the marker on that chromosome (19). The data in Table 2 demonstrate no significant disparity in four independent experiments with diploids heterozygous for ΔAha. We also note that the overall frequency of gene conversion in all diploids heterozygous for ΔAha was significantly (95% confidence level; G test) elevated over RM169 (Table 2). This indicates that the presence of the Aha deletion stimulates recombination even when heterozygous (see Discussion).
Table 2.
Genetic analysis of heterozygous Aha deletion
| Diploid | Genotype | Total no. of tetrads | GC, % | 3:1/1:3 (parity) | |
|---|---|---|---|---|---|
| RM169 |
|
391 | 14.1 | 25/26 (0.96) | |
| RM198 |
|
319 | 23.8 | 33/41 (0.80) | |
| RM212 |
|
303 | 21.1 | 32/31 (1.03) | |
| RM197 |
|
274 | 22.6 | 29/30 (0.97) | |
| RM211 |
|
318 | 18.9 | 26/31 (0.84) | |
| RM203 |
|
296 | 23.6 | 36/31 (1.16) | |
| RM213 |
|
290 | 19.0 | 27/27 (1.00) |
None of the parity values were significantly different from 1.0 using the χ2 test at the 95% confidence level. In fact, the two most different values (RM198 and RM203) were not significantly different (χ2 = 0.838; P = 0.36). RM212, RM211, and RM213 are isogenic to RM169. RM198, RM197, and RM203 are congenic to RM169.
To test the first prediction made above, we examined DSBs in diploids heterozygous for ΔAha that were homozygous for the rad50S allele. If the Aha deletion were capable of stimulating recombination only in the chromosome on which it was located, there should be 2.8 times more breaks on the hot chromosome. To differentially detect breaks on the hot versus the cold chromosome, we used a probe located downstream of the deletion (i.e, the EcoRI–BglII fragment shown to the right in Fig. 1). [When used in an experiment with either a normal or homozygous ΔAha strain, the frequency and location of DSBs detected by this “downstream” probe were the same as those detected by the normal “upstream” probe (data not shown).] Thus, in Fig. 5, the DSB fragments generated from the two homologous chromosomes differ by 382 bp in length. The data in Fig. 5 have been quantitated in Table 3, along with two additional experiments. The data clearly indicate that breaks occur equally on both homologs. The percentage of total DNA broken in the four experiments averages 9.4%; this value is elevated over the average value of 4.3% found in the strain with no deletion (Table 1). Thus, as was found above for gene conversion, the hot chromosome with ΔAha is capable of stimulating DSBs in the heterozygote; interestingly, it does so equally on both homologs.
Figure 5.
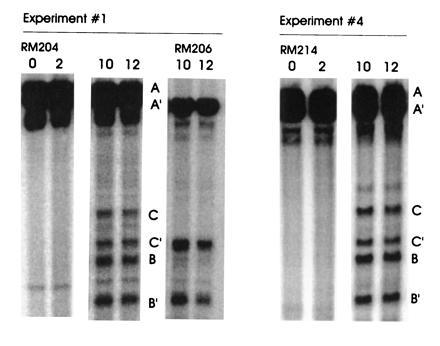
Southern blot analysis of diploids heterozygous for ΔAha. The experiments were done as in Fig. 2, except that the probe was the EcoRI–BglII fragment shown on the right of Fig. 1. Bands from the ΔAha chromosome are A′, B′, and C′. Bands from the normal chromosome are denoted A, B, and C. Because the break bands are probed from the right, they include the deletion. The difference in sizes between the bands A and A′, B and B′, and C and C′ is 382 bp. The DNA from RM206 (homozygous for ΔAha) was run on the same gel as the DNA from RM204 (heterozygous for ΔAha); the positions of the bands in RM206 allow the four bands from RM204 to be assigned to the hot or cold chromosome. Both diploids were homozygous for rad50S. RM204 is congenic to RM169; RM214 is isogenic to RM169.
Table 3.
DSBs in diploids heterozygous for the Aha deletion
| Experiment (strain) | Chromosome broken | 10 hr
|
12 hr
|
Average ratio hot/cold ± SD | |||
|---|---|---|---|---|---|---|---|
| DSB, % | Ratio, hot/cold | DSB, % | Ratio, hot/cold | ||||
| 1 (RM204) | Cold | B | 2.67 | 2.61 | |||
| C | 1.99 | 2.02 | |||||
| (B+C) | 4.66 | 4.63 | |||||
| 1.09 | 1.03 | 1.06 ± 0.04 | |||||
| Hot | B′ | 2.95 | 2.73 | ||||
| C′ | 2.11 | 2.05 | |||||
| (B′+C′) | 5.06 | 4.78 | |||||
| 2 (RM204) | Cold | B | 2.76 | 2.63 | |||
| C | 2.20 | 2.18 | |||||
| (B+C) | 4.96 | 4.81 | |||||
| 1.00 | 1.06 | 1.03 ± 0.04 | |||||
| Hot | B′ | 2.72 | 2.76 | ||||
| C′ | 2.25 | 2.35 | |||||
| (B′+C′) | 4.97 | 5.11 | |||||
| 3 (RM204) | Cold | B | 2.28 | 2.02 | |||
| C | 1.70 | 1.60 | |||||
| (B+C) | 3.98 | 3.62 | |||||
| 0.92 | 1.04 | 0.98 ± 0.08 | |||||
| Hot | B′ | 2.33 | 2.62 | ||||
| C′ | 1.34 | 1.16 | |||||
| (B′+C′) | 3.67 | 3.78 | |||||
| 4 (RM214) | Cold | B | 2.27 | 3.25 | |||
| C | 1.99 | 2.43 | |||||
| (B+C) | 4.26 | 5.68 | |||||
| 0.97 | 0.91 | 0.94 ± 0.04 | |||||
| Hot | B′ | 2.30 | 3.07 | ||||
| C′ | 1.81 | 2.12 | |||||
| (B′+C′) | 4.11 | 5.19 | |||||
| Overall average | 1.00 ± 0.07 | 1.01 ± 0.07 | 1.00 ± 0.2 | ||||
RM204 has the genotype his2-xho-ΔAha/his2-NSI rad50S/rad50S and is congenic to RM169. RM214 has the genotype his2-xho-ΔAha/HIS2 rad50S/rad50S and is isogenic to RM169. The hot chromosome refers to the homolog containing the Aha deletion; the cold chromosome refers to the homolog without the Aha deletion. B, B′, C, and C′ are defined as in Fig. 5. The times refer to cells taken at 10 hr and 12 hr after introduction into sporulation medium. The ratio hot/cold represents the total percent of DSBs on the hot chromosome divided by the total percent on the cold chromosome. A hot/cold ratio of 1.0 indicates that DSBs were formed equally on both homologs.
DISCUSSION
In S. cerevisiae, several loci have been studied in detail both for gene conversion and for DSBs; the ARG4 locus has, perhaps, been examined in the most detail. At ARG4, several hundred base pairs of the promoter region are necessary, but not sufficient, for the normal levels of gene conversion observed at the gene (6, 20). The DSB observed in meiosis at this locus occurs in the same region (5). A number of laboratories have mapped meiotic DSBs in detail along whole chromosomes in S. cerevisiae and have found that breaks occur in regions between open reading frames (10–12). Given the close packing of yeast genes, these regions often contain promoters. Meiotic DSBs also tend to occur in regions that are more susceptible to DNase I cleavage; this observation is consistent with the hypothesis that DSBs occur in “open” chromatin (12). Since active promoters are often found in a more open configuration than other types of DNA, this also explains the association between DSB sites and promoter regions (12).
The recombination hot spot at the HIS2 gene displays gene conversion frequencies as high as 20.7% at the his2-hpa2 allele located ≈50 bp from the end of the coding region. The DSB model of Szostak et al. (3) predicts that the HIS2 locus should exhibit a high level of DSBs. The data presented in this paper are consistent with that prediction. In the normal HIS2 locus (with no alterations present), ≈4.3% of the DNA exhibits a DSB. The breaks appear early in meiosis and are dependent upon known recombination functions, and the amount of DSBs is correlated with the amount of gene conversion observed. If every DSB resulted in a detectable gene conversion event and if breaks occurred on only one of the four chromatids, one would expect a ratio of conversion/DSBs equal to 4. For the -hpa2 allele located near the high end of the polarity gradient at HIS2, the ratio for the wild-type diploid containing no alterations is 20.7%/4.3% = 4.8. We note that the correlation between the amount of DSBs and the amount of gene conversion is not linear, unlike the correlation at HIS4 (9). For the his2-xho allele measured (Table 1), the ratios of conversion to DSBs are >4.4, 3.3, and 1.95 for the TUBA66, wild-type, and ΔAha diploids, respectively. The efficiency of turning a DSB into a conversion event at HIS2 appears to decrease as there are more breaks.
One of the things that is novel about DSBs and the HIS2 hot spot is the locations of the breaks compared with the other natural yeast loci that have been studied both genetically and physically. DSB C actually occurs in the coding region of HIS2. This argues that meiotic DSBs can occur in the middle of a transcribed region. [We have demonstrated that the HIS2 gene is transcribed in meiosis at approximately the same level as mitosis (data not shown).] Although most meiotic DSBs appear to be associated with intergenic regions, DSB C at HIS2 indicates that there can be other possibilities. The data indicating a relationship between open chromatin and DSB sites (12) predict that the internal DSB C in the HIS2 gene is a DNase I sensitive site. We are in the process of testing that prediction.
The downstream DSB B at HIS2 occurs ≈200 bp 3′ of the coding region (Fig. 2; Fig. 6). The next open reading frame (ORF; YFR024C) downstream of HIS2 starts at position +1671 (relative to the +1 of the HIS2 ORF). [We have demonstrated that this ORF is not essential; neither deletions of it nor insertions in it have any effect on cell viability, growth rate, or meiosis (data not shown). We have therefore referred to this ORF as NEG1, for nonessential gene (Fig. 1).] NEG1 is transcribed in both mitosis and meiosis (data not shown), and the direction of transcription is the same as HIS2. Thus, the NEG1 promoter is located somewhere in the 665 bp of DNA between the last base of the HIS2 ORF (+1005) and the first base of the NEG1 ORF (+1671). There are possible TATA boxes at +1374 and +1238. We conclude that the downstream DSB B (at position +1200) could be located in the NEG1 promoter. We have demonstrated previously (14) that alterations located >500 bp downstream of the putative NEG1 promoter region can affect gene conversion at HIS2. This suggests that, if the NEG1 promoter is necessary for the high frequency of conversion observed at HIS2, it is not sufficient. Consistent with this, moving the 5.2-kb BglII fragment (Fig. 1) to chromosome II did not retain the recombination hot spot (14), even though the NEG1 promoter and ORF were present. To the extent that chromatin structure is important in determining DSB sites (12), these data suggest that such structure can be affected by factors other than local sequence. Finally, we note that the locations of the DSBs in strains with the Aha deletion (Fig. 4) are not detectably different from the locations in normal strains, even though the number of DSBs is increased 2.8-fold. This suggests that the locations of the breaks have some sequence specificity. If true, this indicates that the recombination hot spot at HIS2 has at least two separable components: the sites where the DSBs actually occur and the downstream sequences we have demonstrated are necessary for both the DSBs and conversion (14).
Figure 6.
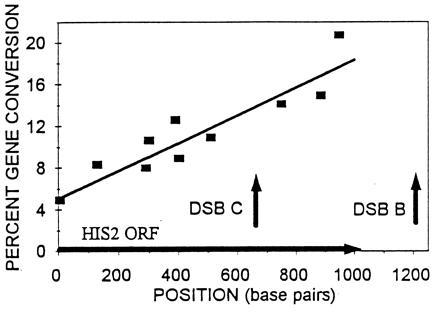
Map of DSBs relative to conversion gradient. Data for conversion comes from ref. 14, except for the his2-hpa2 allele. The arrow on the x-axis represents the HIS2 coding region. The least squares line has a correlation coefficient of 0.85. Vertical arrows represent the position of the DSBs.
The second novel property observed at HIS2 is that the DSBs occur equally on both homologs, even when only one of the two chromosomes contains the ΔAha alteration. DSB bands from both parents were examined simultaneously in one experiment, thereby allowing the breaks on the two chromosomes to be easily compared (Table 3). Since the hot chromosome stimulates breaks on the cold chromosome, ΔAha formally can be defined as semidominant; said another way, the deletion acts to stimulate recombination in trans. In the original version of the DSB model (3), and in the various modifications of it that have been proposed since, it was proposed that recombination is initiated by the broken ends of one homolog invading the other. For example, it has been suggested that the interaction of the single-stranded tails (from the DSB) with an intact homologous duplex helps to initiate recombination between homologs (21). More recently, Weiner and Kleckner (22) found evidence for interactions between homologs early during meiosis. These meiotic “interstitial” interactions were independent of the synaptonemal complex and did not require meiosis-specific DSBs. From these data, they proposed a model in which early meiotic pairing occurs by closely related DNA–DNA interactions between intact duplexes, possibly by paranemic interactions (22). They further hypothesized that meiotic DSBs converted these initial interactions from paranemic to plectonemic recombination intermediates. The Weiner and Kleckner model clearly proposes that DSBs occur after homologs are already interacting.
The features of the DSBs found at HIS2 are absolutely consistent with the hypothesis that DSBs are involved in, and required for, recombination at HIS2, as has been found for other loci in yeast. The data from analysis of the heterozygous ΔAha strains (Fig. 5; Table 3) are consistent with the view that meiotic DSBs are formed after homologs are already interacting, perhaps in the manner proposed by Weiner and Kleckner (22). If, in the diploid heterozygous for ΔAha, DSBs occurred independently on each chromosome at frequencies similar to those found in the homozygotes, the hot chromosome containing ΔAha should have 2.8 times as many breaks as the cold one. In contrast, the DSBs occur equally on both homologs. One could imagine that the hot chromosome is better at attracting the proteins involved in making the DSBs, but that once such proteins were present, they could make the breaks on either homolog with equal probability because the two chromosomes are in contact.
In an examination of DSBs occurring at the HIS4::LEU2 hot spot (a hot spot created when 77 bp of Escherichia coli DNA were coincidentally inserted at the junction), Xu and Kleckner (23) found that inserting an additional 32-bp BamHI linker increased the frequency of DSBs from 18.4% to 24.3% when homozygous; recombination frequencies were not reported. There are two breaks at the HIS4::LEU2 hot spot, but only the one near the location of the inserted linker DNA was stimulated. The frequency of the other DSB was diminished by the insertion from 4.4% to 2.2%. Xu and Kleckner suggested that there might be competition between the two break sites (some 2 kb apart). We note that both HIS2 DSBs reacted similarly to alterations that affected recombination; both breaks were increased by the ΔAha, and both were decreased by the TUBA66 insertion. We do not see evidence for competition between the two DSBs (located ≈600 bp apart) at HIS2. This might reflect differences between natural and artificial hot spots, the closer distance between the breaks at HIS2, or our alterations not being located at the sites where the breaks occur. Xu and Kleckner (23) also examined DSBs in crosses heterozygous for the stimulating BamHI linker. In such crosses, the total amount of DSBs increased to 20.7%, a value intermediate to those found in the homozygotes (see above). As in the crosses with the homozygous linker, the entire increase was in the site close to the linker. Although the differences were slight, the authors speculated that there was a trans effect of the BamHI linker; they argued that its presence on the hot homolog decreased DSBs at both sites on the other cold homolog. If their suggestion proves to be true, then the trans effect of alterations at the HIS2 locus and the HIS4::LEU2 locus are in opposite directions. However, both observations are consistent with the view that homologs interact before DSBs are formed.
Finally, we note that at least 10 genes are required for the initiation of meiotic recombination in S. cerevisiae (16). Mutations in these “early exchange” genes [e.g., REC104 (18)] reduce meiotic recombination to background mitotic levels; in those that have been tested, DSBs are not detectable (e.g., see Fig. 2). One can posit at least three types of roles for these early exchange genes. First, some of the functions encoded by these genes may act directly in the formation of DSBs. Second, some of them may be required both for the homolog interactions proposed by Weiner and Kleckner (22) and for formation of DSBs. Third, some of the early exchange functions might play a role only in pre-DSB homolog interaction. Weiner and Kleckner demonstrated that a spo11 mutation (which prevents DSBs) reduced interstitial interactions to 10% of normal, consistent with either the second or the third idea. If the only role of one of the early exchange genes were to make interstitial interactions between homologs, this would imply that such interactions are required for recombination in diploids since mutations in the early exchange genes abolish interchromosomal meiotic recombination. However, two laboratories have found meiotic DSBs in haploids; they occur at the appropriate places and with nearly normal kinetics (24, 25). If the role of any of the early exchange genes were limited to the formation of interstitial interactions, one would predict that mutations in such genes would have little effect upon DSBs occurring in a haploid meiosis, since there are no homologs present. Of the early exchange genes, only the effect of a rad50 mutation in haploid meiosis has been reported, and the rad50 null mutation reduces the amount of DSBs that occur in a haploid meiosis (25).
Acknowledgments
We thank Maureen Cantwell, Yang Mao-Draayer, Jack Gardiner, George Halley, Laura Salem, and John Nau for helpful comments on the manuscript. This work was supported by National Institutes of Health Grant R01-GM36846 to R.E.M.
Footnotes
Abbreviations: DSB, double strand break; ORF, open reading frame.
References
- 1.Holliday R. Genet Res. 1964;5:282–304. [Google Scholar]
- 2.Meselson M S, Radding C M. Proc Natl Acad Sci USA. 1975;72:358–363. doi: 10.1073/pnas.72.1.358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Szostak J W, Orr-Weaver T L, Rothstein R J, Stahl F W. Cell. 1983;33:24–35. doi: 10.1016/0092-8674(83)90331-8. [DOI] [PubMed] [Google Scholar]
- 4.West S C, Cassuto E, Howard-Flanders P. Nature (London) 1981;294:659–662. doi: 10.1038/294659a0. [DOI] [PubMed] [Google Scholar]
- 5.Sun H, Treco D, Schultes N P, Szostak J W. Nature (London) 1989;338:87–90. doi: 10.1038/338087a0. [DOI] [PubMed] [Google Scholar]
- 6.Nicholas A, Treco D, Schultes N P, Szostak J W. Nature (London) 1989;338:35–39. doi: 10.1038/338035a0. [DOI] [PubMed] [Google Scholar]
- 7.Alani E, Padmore R, Kleckner N. Cell. 1990;61:419–436. doi: 10.1016/0092-8674(90)90524-i. [DOI] [PubMed] [Google Scholar]
- 8.Cao L, Alani E, Kleckner N. Cell. 1990;61:1089–1101. doi: 10.1016/0092-8674(90)90072-m. [DOI] [PubMed] [Google Scholar]
- 9.Fan Q, Xu F, Petes T D. Mol Cell Biol. 1995;15:1679–1688. doi: 10.1128/mcb.15.3.1679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Game J. Dev Genet. 1992;13:485–497. doi: 10.1002/dvg.1020130610. [DOI] [PubMed] [Google Scholar]
- 11.Zenvirth D, Arbel T, Sherman A, Goldway M, Klein S, Simchen G. EMBO J. 1992;11:3341–3347. doi: 10.1002/j.1460-2075.1992.tb05423.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wu T C, Lichten M. Science. 1994;263:525–528. doi: 10.1126/science.8290959. [DOI] [PubMed] [Google Scholar]
- 13.Malone R E, Bullard S A, Lundquist S, Kim S, Tarkowski T. Nature (London) 1992;359:154–155. doi: 10.1038/359154a0. [DOI] [PubMed] [Google Scholar]
- 14.Malone R E, Kim S, Bullard S A, Lundquist S, Hutchings-Crow L, Cramton S, Lutfiyya L, Lee J. Genetics. 1994;137:5–18. doi: 10.1093/genetics/137.1.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rothstein R J. Methods Enzymol. 1991;194:281–301. doi: 10.1016/0076-6879(91)94022-5. [DOI] [PubMed] [Google Scholar]
- 16.Mao-Draayer Y, Galbraith A M, Pittman D L, Cool M, Malone R E. Genetics. 1996;144:71–86. doi: 10.1093/genetics/144.1.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sambrook J, Fritsch E F, Maniatis T. Molecular Cloning: A Laboratory Manual. 2nd Ed. Plainview, NY: Cold Spring Harbor Lab. Press; 1989. [Google Scholar]
- 18.Galbraith A, Malone R E. Dev Genet. 1992;13:392–402. doi: 10.1002/dvg.1020130603. [DOI] [PubMed] [Google Scholar]
- 19.Thaler D, Stahl F W. Annu Rev Genet. 1988;22:169–197. doi: 10.1146/annurev.ge.22.120188.001125. [DOI] [PubMed] [Google Scholar]
- 20.Schultes N P, Szostak J W. Mol Cell Biol. 1991;11:322–328. doi: 10.1128/mcb.11.1.322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sun H, Treco D, Szostak J W. Cell. 1991;64:1155–1161. doi: 10.1016/0092-8674(91)90270-9. [DOI] [PubMed] [Google Scholar]
- 22.Weiner B, Kleckner N. Cell. 1994;77:977–991. doi: 10.1016/0092-8674(94)90438-3. [DOI] [PubMed] [Google Scholar]
- 23.Xu L, Kleckner N. EMBO J. 1995;14:5115–5128. doi: 10.1002/j.1460-2075.1995.tb00194.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gilbertson L A, Stahl F W. Proc Natl Acad Sci USA. 1994;91:11934–11937. doi: 10.1073/pnas.91.25.11934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.de Massy B, Baudat F, Nicolas A. Proc Natl Acad Sci USA. 1994;91:11929–11933. doi: 10.1073/pnas.91.25.11929. [DOI] [PMC free article] [PubMed] [Google Scholar]