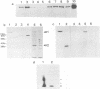
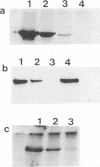
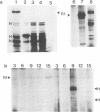
Abstract
We report on a protein kinase function encoded by the unique N terminus of the herpes simplex virus type 1 (HSV-1) ribonucleotide reductase large subunit (R1). R1 expressed in Escherichia coli exhibited autophosphorylation activity in a reaction which depended on the presence of the unique N terminus. When the N terminus was separately expressed in E. coli and partially purified, a similar autophosphorylation reaction was observed. Importantly, transphosphorylation of histones and of proteins in HSV-1-infected cell extracts was also observed with purified R1 and with truncated R1 mutants in which most of the N terminus was deleted. Ion-exchange chromatography was used to separate the autophosphorylating activity of the N terminus from the transphosphorylating activity of an E. coli contaminant protein kinase. We propose a putative function for this activity of the HSV-1 R1 N terminus during the immediate-early phase of virus replication.
Full text
PDF
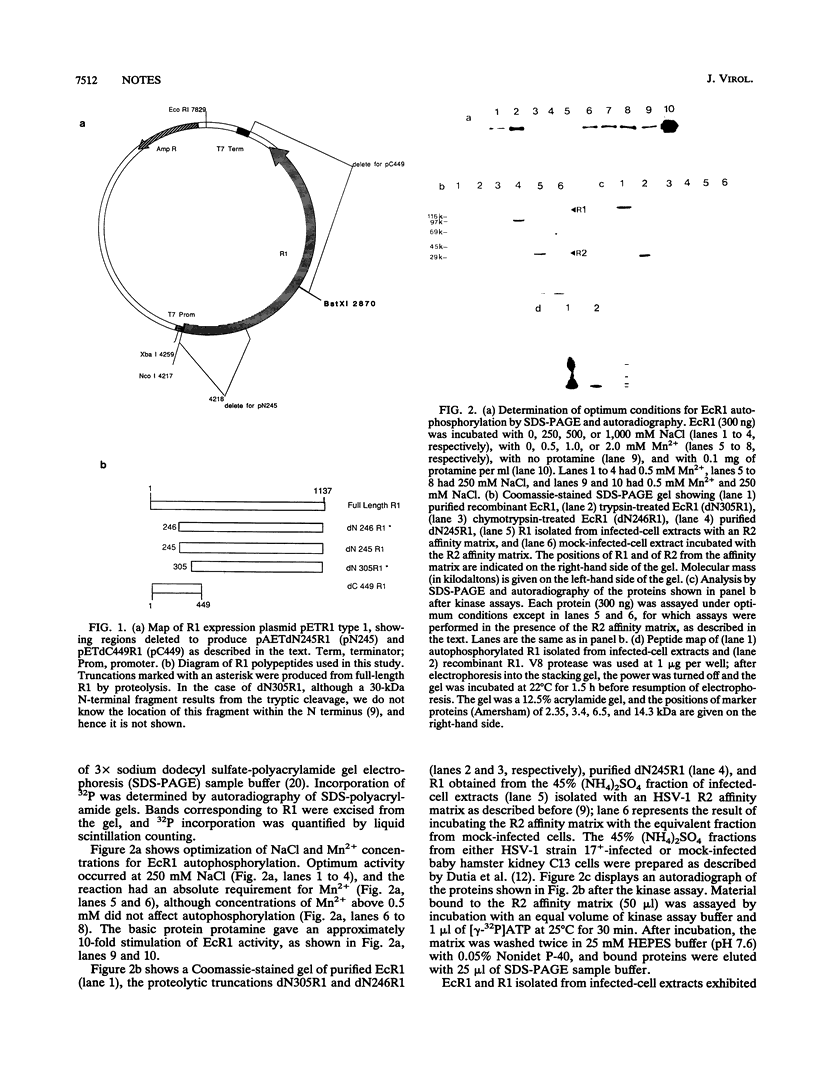




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ali M. A., McWeeney D., Milosavljevic A., Jurka J., Jariwalla R. J. Enhanced malignant transformation induced by expression of a distinct protein domain of ribonucleotide reductase large subunit from herpes simplex virus type 2. Proc Natl Acad Sci U S A. 1991 Sep 15;88(18):8257–8261. doi: 10.1073/pnas.88.18.8257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bacchetti S., Evelegh M. J., Muirhead B. Identification and separation of the two subunits of the herpes simplex virus ribonucleotide reductase. J Virol. 1986 Mar;57(3):1177–1181. doi: 10.1128/jvi.57.3.1177-1181.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung T. D., Wymer J. P., Kulka M., Smith C. C., Aurelian L. Myristylation and polylysine-mediated activation of the protein kinase domain of the large subunit of herpes simplex virus type 2 ribonucleotide reductase (ICP10). Virology. 1990 Nov;179(1):168–178. doi: 10.1016/0042-6822(90)90286-z. [DOI] [PubMed] [Google Scholar]
- Chung T. D., Wymer J. P., Smith C. C., Kulka M., Aurelian L. Protein kinase activity associated with the large subunit of herpes simplex virus type 2 ribonucleotide reductase (ICP10). J Virol. 1989 Aug;63(8):3389–3398. doi: 10.1128/jvi.63.8.3389-3398.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clements J. B., Watson R. J., Wilkie N. M. Temporal regulation of herpes simplex virus type 1 transcription: location of transcripts on the viral genome. Cell. 1977 Sep;12(1):275–285. doi: 10.1016/0092-8674(77)90205-7. [DOI] [PubMed] [Google Scholar]
- Cleveland D. W., Fischer S. G., Kirschner M. W., Laemmli U. K. Peptide mapping by limited proteolysis in sodium dodecyl sulfate and analysis by gel electrophoresis. J Biol Chem. 1977 Feb 10;252(3):1102–1106. [PubMed] [Google Scholar]
- Cohen G. H., Factor M. N., Ponce de Leon M. Inhibition of herpes simplex virus type 2 replication by thymidine. J Virol. 1974 Jul;14(1):20–25. doi: 10.1128/jvi.14.1.20-25.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen G. H. Ribonucleotide reductase activity of synchronized KB cells infected with herpes simplex virus. J Virol. 1972 Mar;9(3):408–418. doi: 10.1128/jvi.9.3.408-418.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conner J., Macfarlane J., Lankinen H., Marsden H. The unique N terminus of the herpes simplex virus type 1 large subunit is not required for ribonucleotide reductase activity. J Gen Virol. 1992 Jan;73(Pt 1):103–112. doi: 10.1099/0022-1317-73-1-103. [DOI] [PubMed] [Google Scholar]
- Dutia B. M., Frame M. C., Subak-Sharpe J. H., Clark W. N., Marsden H. S. Specific inhibition of herpesvirus ribonucleotide reductase by synthetic peptides. Nature. 1986 May 22;321(6068):439–441. doi: 10.1038/321439a0. [DOI] [PubMed] [Google Scholar]
- Dutia B. M. Ribonucleotide reductase induced by herpes simplex virus has a virus-specified constituent. J Gen Virol. 1983 Mar;64(Pt 3):513–521. doi: 10.1099/0022-1317-64-3-513. [DOI] [PubMed] [Google Scholar]
- Frame M. C., Marsden H. S., Dutia B. M. The ribonucleotide reductase induced by herpes simplex virus type 1 involves minimally a complex of two polypeptides (136K and 38K). J Gen Virol. 1985 Jul;66(Pt 7):1581–1587. doi: 10.1099/0022-1317-66-7-1581. [DOI] [PubMed] [Google Scholar]
- Furlong J., Conner J., McLauchlan J., Lankinen H., Galt C., Marsden H. S., Clements J. B. The large subunit of herpes simplex virus type 1 ribonucleotide reductase: expression in Escherichia coli and purification. Virology. 1991 Jun;182(2):846–851. doi: 10.1016/0042-6822(91)90627-n. [DOI] [PubMed] [Google Scholar]
- Hanks S. K., Quinn A. M., Hunter T. The protein kinase family: conserved features and deduced phylogeny of the catalytic domains. Science. 1988 Jul 1;241(4861):42–52. doi: 10.1126/science.3291115. [DOI] [PubMed] [Google Scholar]
- Hattori M., Sakaki Y. Dideoxy sequencing method using denatured plasmid templates. Anal Biochem. 1986 Feb 1;152(2):232–238. doi: 10.1016/0003-2697(86)90403-3. [DOI] [PubMed] [Google Scholar]
- Ingemarson R., Lankinen H. The herpes simplex virus type 1 ribonucleotide reductase is a tight complex of the type alpha 2 beta 2 composed of 40K and 140K proteins, of which the latter shows multiple forms due to proteolysis. Virology. 1987 Feb;156(2):417–422. doi: 10.1016/0042-6822(87)90422-3. [DOI] [PubMed] [Google Scholar]
- Kamps M. P., Taylor S. S., Sefton B. M. Direct evidence that oncogenic tyrosine kinases and cyclic AMP-dependent protein kinase have homologous ATP-binding sites. Nature. 1984 Aug 16;310(5978):589–592. doi: 10.1038/310589a0. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lankinen H., McLauchlan J., Weir M., Furlong J., Conner J., McGarrity A., Mistry A., Clements J. B., Marsden H. S. Purification and characterization of the herpes simplex virus type 1 ribonucleotide reductase small subunit following expression in Escherichia coli. J Gen Virol. 1991 Jun;72(Pt 6):1383–1392. doi: 10.1099/0022-1317-72-6-1383. [DOI] [PubMed] [Google Scholar]
- Luo J. H., Aurelian L. The transmembrane helical segment but not the invariant lysine is required for the kinase activity of the large subunit of herpes simplex virus type 2 ribonucleotide reductase (ICP10). J Biol Chem. 1992 May 15;267(14):9645–9653. [PubMed] [Google Scholar]
- Luo J. H., Smith C. C., Kulka M., Aurelian L. A truncated protein kinase domain of the large subunit of herpes simplex virus type 2 ribonucleotide reductase (ICP10) expressed in Escherichia coli. J Biol Chem. 1991 Nov 5;266(31):20976–20983. [PubMed] [Google Scholar]
- Maru Y., Witte O. N. The BCR gene encodes a novel serine/threonine kinase activity within a single exon. Cell. 1991 Nov 1;67(3):459–468. doi: 10.1016/0092-8674(91)90521-y. [DOI] [PubMed] [Google Scholar]
- McGeoch D. J., Dalrymple M. A., Davison A. J., Dolan A., Frame M. C., McNab D., Perry L. J., Scott J. E., Taylor P. The complete DNA sequence of the long unique region in the genome of herpes simplex virus type 1. J Gen Virol. 1988 Jul;69(Pt 7):1531–1574. doi: 10.1099/0022-1317-69-7-1531. [DOI] [PubMed] [Google Scholar]
- Nikas I., Darling A. J., Lankinen H. M., Cross A. M., Marsden H. S., Clements J. B. A single amino acid substitution in the large subunit of herpes simplex virus type 1 ribonucleotide reductase which prevents subunit association. J Gen Virol. 1990 Oct;71(Pt 10):2369–2376. doi: 10.1099/0022-1317-71-10-2369. [DOI] [PubMed] [Google Scholar]
- Nikas I., McLauchlan J., Davison A. J., Taylor W. R., Clements J. B. Structural features of ribonucleotide reductase. Proteins. 1986 Dec;1(4):376–384. doi: 10.1002/prot.340010411. [DOI] [PubMed] [Google Scholar]
- Nordlund P., Sjöberg B. M., Eklund H. Three-dimensional structure of the free radical protein of ribonucleotide reductase. Nature. 1990 Jun 14;345(6276):593–598. doi: 10.1038/345593a0. [DOI] [PubMed] [Google Scholar]
- Paradis H., Gaudreau P., Massie B., Lamarche N., Guilbault C., Gravel S., Langelier Y. Affinity purification of active subunit 1 of herpes simplex virus type 1 ribonucleotide reductase exhibiting a protein kinase activity. J Biol Chem. 1991 May 25;266(15):9647–9651. [PubMed] [Google Scholar]
- Preston V. G., Palfreyman J. W., Dutia B. M. Identification of a herpes simplex virus type 1 polypeptide which is a component of the virus-induced ribonucleotide reductase. J Gen Virol. 1984 Sep;65(Pt 9):1457–1466. doi: 10.1099/0022-1317-65-9-1457. [DOI] [PubMed] [Google Scholar]
- Russo M. W., Lukas T. J., Cohen S., Staros J. V. Identification of residues in the nucleotide binding site of the epidermal growth factor receptor/kinase. J Biol Chem. 1985 May 10;260(9):5205–5208. [PubMed] [Google Scholar]
- Seto D. An improved method for sequencing double stranded plasmid DNA from minipreps using DMSO and modified template preparation. Nucleic Acids Res. 1990 Oct 11;18(19):5905–5906. doi: 10.1093/nar/18.19.5905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stubbe J. A. Protein radical involvement in biological catalysis? Annu Rev Biochem. 1989;58:257–285. doi: 10.1146/annurev.bi.58.070189.001353. [DOI] [PubMed] [Google Scholar]
- Stubbe J. Ribonucleotide reductases: amazing and confusing. J Biol Chem. 1990 Apr 5;265(10):5329–5332. [PubMed] [Google Scholar]
- Studier F. W., Rosenberg A. H., Dunn J. J., Dubendorff J. W. Use of T7 RNA polymerase to direct expression of cloned genes. Methods Enzymol. 1990;185:60–89. doi: 10.1016/0076-6879(90)85008-c. [DOI] [PubMed] [Google Scholar]
- Swain M. A., Galloway D. A. Herpes simplex virus specifies two subunits of ribonucleotide reductase encoded by 3'-coterminal transcripts. J Virol. 1986 Mar;57(3):802–808. doi: 10.1128/jvi.57.3.802-808.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thelander L., Reichard P. Reduction of ribonucleotides. Annu Rev Biochem. 1979;48:133–158. doi: 10.1146/annurev.bi.48.070179.001025. [DOI] [PubMed] [Google Scholar]
- Watson R. J., Preston C. M., Clements J. B. Separation and characterization of herpes simplex virus type 1 immediate-early mRNA's. J Virol. 1979 Jul;31(1):42–52. doi: 10.1128/jvi.31.1.42-52.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu J. Y., Zhou Z. Y., Judd A., Cartwright C. A., Robinson W. S. The hepatitis B virus-encoded transcriptional trans-activator hbx appears to be a novel protein serine/threonine kinase. Cell. 1990 Nov 16;63(4):687–695. doi: 10.1016/0092-8674(90)90135-2. [DOI] [PubMed] [Google Scholar]
- Wymer J. P., Chung T. D., Chang Y. N., Hayward G. S., Aurelian L. Identification of immediate-early-type cis-response elements in the promoter for the ribonucleotide reductase large subunit from herpes simplex virus type 2. J Virol. 1989 Jun;63(6):2773–2784. doi: 10.1128/jvi.63.6.2773-2784.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]