Abstract
Purpose
In this study the chondrocytic differentiation and cartilage matrix accumulation of human mesenchymal stem cells (hMSCs) were investigated after encapsulation in a genetically engineered silk-elastinlike protein polymer SELP-47 K as an injectable matrix for delivery of cell-based therapeutics.
Materials and Methods
hMSCs were encapsulated in SELP-47 K and cultured for 4 weeks in chondrogenic medium with or without transforming growth factor-β3 (TGF). Chondrogenic differentiation was evaluated by histological, RNA and biochemical analyses for the expression of cartilage extracellular matrix components.
Results
Histological and immunohistochemical staining revealed that the cells acquired a rounded morphology and were embedded in significant amounts of chondrogenic extracellular matrix. Reverse transcriptase (RT)-PCR showed an up-regulation in aggrecan, type II and type X collagen and SOX9 in presence of TGF-β3. By day 28, constructs cultured in the presence of TGF-β3 exhibited significant increase in sulfated glycosaminoglycan and total collagen content up to 65 and 300%, respectively.
Conclusions
This study demonstrates that SELP-47 K hydrogel can be used as a scaffold for encapsulation and chondrogenesis of hMSCs. The ability to use recombinant techniques to precisely control SELP structure enables the investigation of injectable protein polymer scaffolds for soft-tissue engineering with varied physicochemical properties.
Keywords: chondrogenesis, genetically engineered polymers, hydrogels, silk-elastinelike polymers, tissue engineering
INTRODUCTION
Trauma to the articular cartilage surface of the joint represents a challenging clinical problem due to the poor ability of this tissue to self-repair (1). Current repair techniques such as microfracture, which stimulates cellular infiltration into the joint, have unpredictable clinical outcomes as they may produce a fibrocartilage tissue that degenerates with time (2). Alternative treatments include tissue reconstruction with autograft and allograft tissues (3). However, these procedures are restricted by the availability of suitable donor tissue. Through an integrated application of cells, biomaterial scaffolds, and biochemical and physical regulatory factors, tissue engineering emerges as a potential strategy for repair or augmentation of injured cartilaginous tissues.
The vast majority of polymers investigated so far for cartilage tissue engineering have been synthetic, natural, or composites thereof (4–6). Synthetic polymers by and large are statistically diverse in molecular size and composition, and lack intrinsic biofunctionality. Potential contamination with residual organic solvent and catalyst may also complicate biocompatibility. On the other hand, natural polymers such as collagen matrices used in tissue engineering (7,8) suffer from batch-to-batch variation, and the difficulty of introducing new functional motifs (e.g., stimuli-sensitive, biodegradable, and biorecognizable) to allow control over mechanical and biological properties. Biomaterials compatible with living organisms and tissues, and whose physicochemical, mechanical and biological characteristics and fate can be precisely controlled at the molecular level would be attractive.
Advances in recombinant DNA technology have allowed the genetic engineering of large molecular weight polymers containing repeating blocks of amino acids with precise composition, sequence and length (6,9–11). Control over polymeric architecture at the molecular level results in fine control over physicochemical and mechanical properties of the polymeric matrix (12–18). Protein-based polymers are generally biocompatible when used as implants, do not contain toxic monomer residues and solvents, and depending on structure can breakdown into nontoxic amino acids either reutilized or readily eliminated from the body (11).
One class of genetically engineered biomaterials is the family of silk-elastinlike protein polymers (SELPs), composed of amino acid sequence motifs from Bombyx mori (silkworm) silk (Gly-Ala-Gly-Ala-Gly-Ser) and mammalian elastin (Gly-Val-Gly-Val-Pro). Solubility, gelation, stimuli-sensitivity, drug release, material strength, biorecognition, and biodegradation profiles of SELPs can precisely be controlled by varying the composition and sequence of the polymers (19). SELP copolymers, depending on the number and sequence of silk and elastin blocks, undergo an irreversible sol-to-gel transition as a function of temperature, presumably through the formation of hydrogen bonds between the silk blocks (12,13,20–22). Fluid at room temperature, a SELP-47 aqueous solution would gel within minutes after injection into the body. These systems are biocompatible, have been investigated for matrix-mediated gene and drug delivery (13,21–23) and are in clinical testing as urethral bulking agents for the treatment of female stress urinary incontinence as well as dermal augmentation. However, their potential in tissue engineering applications and more specifically cartilage-like tissue is unexplored. In this study we investigated the in vitro chondrocytic differentiation and cartilage matrix accumulation of human mesenchymal stem cells (hMSCs) derived from bone marrow after encapsulation in SELP-47 K as an injectable matrix for delivery of cell-based therapeutics.
MATERIALS AND METHODS
Materials and Methods
The silk-elastinlike protein copolymer SELP-47 K, composed of four silk-like blocks, seven elastin-like blocks, and one modified elastin block containing a lysine (K) substitution (amino acid sequence in Fig. 1), was produced by Protein Polymer Technologies, Inc. (San Diego, CA) as a 12 wt% solution in 3 ml syringes and stored at −80°C until use. Poietics™ hMSC human mesenchymal stem cells harvested and cultured from normal human bone marrow, MSCGM and Chondrogenic Differentiation media and growth factor TGF-β3 were obtained from Cambrex Corporation(East Rutherford, NJ). Cell proliferation reagent WST-1 was obtained from Roche Applied Science (Indianapolis, IN). Live/Dead Viability/Cytotoxicity assay kit was purchased from Molecular Probes (Eugene, OR). RNA primers were from Integrated DNA Technologies (Coralville, IA). RNALater RNA stabilization reagent and Qiagen One Step RT-PCR kit were obtained from Qiagen (Valencia, CA). Blyscan sulfated glycosaminoglycan assay was acquired from Biocolor Ltd. (Newtownabbey, Northern Ireland).
Fig. 1.

The amino acid sequence of SELP-47 K. The 884 amino acid residues have a total molecular weight of 69,814 D. The polymer is composed of head and tail portions (underlined), and a series of silk-like (GAGAGS) and elastin-like (GVGVP) repeats (primary repetitive sequence in bold, number of repeats highlighted in gray). For amino acid designations, see abbreviations.
General Cell Culture
Human mesenchymal stem cells were cultured following the manufacturer’s recommended protocol. Briefly, cells were cultured with MSCGM media containing 10% FBS, 1% Penicillin/streptomycin and 1% glutamine. The cells were maintained at 37°C, 5% CO2 environment. Cells were subcultured 1:5 to 85–90% confluence after dissociation in trypsin/EDTA (0.05 ml/cm2).
Preparation of Hydrogel Cell Scaffolds
Syringes containing frozen 12 wt% SELP-47 K solution were thawed in water for 5 min at room temperature. hMSC were removed from culture plates with 0.05% trypsin Cambrex Corporation (East Rutherford, NJ) and centrifuged (600 ×g for 5 min) to form a pellet (106 cells/pellet). The cell pellets were resuspended in the SELP47 K polymer solution and the mixture volume was adjusted by addition of culture media to yield an 11.5% polymer solution containing 8×106 cells/ml. The mixture was transferred to disposable 1-ml syringes, incubated at 37°C for 30 min and cut into cylindrical discs of 50 mm3 (r=2.3 mm, h=2.8 mm). The encapsulated hMSCs were cultured in chondrogenic medium in the presence or absence of TGF-β3 for further studies (n=3). Cells were washed and provided with fresh mEdia every 2 days.
Cell Viability and Proliferation
After 1, 2 or 4 weeks of culture, the metabolic activity and proliferation of cells cultured in the hydrogel-composite were evaluated by WST-1 assay and Live/Dead assay reagent. WST-1 assay was performed by transferring constructs to 96-well plates containing 200 μl of fresh media per well. Forty microliter of WST-1 reagent were then added to each well and incubated for 4 h at 37°C, 5% CO2 environment. From each well, 100 μl was transferred to another 96-well plate and absorbance was examined at 450 nm using a microplate reader. The results from the WST-1 assay were normalized against scaffolds prepared from SELP47 K only. The LIVE/DEAD assay dye mixture was prepared according to manufacturer protocol. Briefly, 4 μl of 20 mM ethidium homodimer-1 (EthD-1) were mixed with 2 ml PBS and vortexed then 1 μl of 4 mM calcein AM was added and thoroughly mixed by vortexing. The resulting solution contained 4 μM EthD-1 (for staining of dead cells) and 2 μM calcein AM (for staining of live cells). Hydrogel/cell scaffolds were washed three times with PBS and treated with 250 μl of the dye mixture after 1, 14 and 28 days of culture. The cell-culture plates were wrapped in aluminum foil and incubated at room temperature for 30 min. The labeled cells were then examined under confocal microscope. A control group of dead cells was obtained by treating the hydrogel/cell scaffolds with 250 μl of 0.1% saponin in PBS for 10 min. The cytotoxic saponin solution was then removed and the scaffolds were washed with PBS and treated with 250 μl of the dye mixture.
RNA Extraction and Reverse Transcription Polymerase Chain Reaction
Each MSC/SELP-47 K gel construct was placed in 10-fold excess of gel volume of RNALater RNA stabilization reagent and stored at −80°C until RNA extraction. Total RNA was isolated from three constructs per group and from MSC cell pellets of the same passage cultured in similar condition using an RNeasy minikit (Qiagen, Valencia, CA). Samples were prepared for RNA extraction by mechanical homogenization in 2 ml RLT lysis buffer RNeasy minikit (Qiagen, Valencia, CA). After complete homogenization, 400 ml more of the RLT buffer was added to the microcentrifuge tubes and the suspension was further homogenized with a QIAshredder (Qiagen) column. The homogenates were transferred to columns from the RNeasy minikit after an equal volume of 70% ethanol was added. RNA was isolated according to the manufacturer’s protocol. The purified RNA samples were analyzed via reverse transcription polymerase chain reaction (RT-PCR) using OneStep RT-PCR kit (Qiagen, Valencia, CA) and run on the Biorad iCycler thermal cycler (Biorad, Hercules, CA) for 35 PCR cycles. Polymerase chain reaction (PCR) primers (forward and backward, 5′ to 3′) were as follows: β-actin, 5′-ATCTGGCACACCTTCTACAAT GAGCTGCG-3′ and 5′-CGTCATACTCCTGCTTGCT GATCCACATCTGC-3′; Aggrecan, 5′-CACGCTA CACCCTGGACTTG-3′ and 5′-CCATCTCTCAGGAAG CAGT-3′; collagen I, 5′-TGACGAGACCAAGAACTG-3′ and 5′-CAATCCAAACCACTGAAACC-3′; collagen II, 5′-AGGAGGCTGGCAGCTGTGTGC-3′ and 5′-CACTGGC AGTGGCGAGGTCAG-3′; collagen X, 5′-CCCTTT T T G C T G C T A G T A T C C - 3 ′ and 5 ′-C T G T T G T CCAGGTTTTCCTGGCAC-3′; sox 9, 5′-ACGTCATCTC CAACATCGAGACC-3′ and 5′-CTGTAGTGTGG GAGGTTGAAGGG-3′ Each PCR product was analyzed by separating 12 μl of the amplicon and 3 μl of loading buffer in a 1.5% agarose gel in TAE buffer. The relative levels of band intensity of the gene of interest were compared with those of the internal control of a housekeeping gene.
Histology and Immunohistochemistry
To assess cell morphology and the presence of cartilage specific matrix proteins, representative constructs from days 1, 14 and 28 of culture were formalin-fixed, paraffin-embedded, and sectioned (5 μm) for routine histology. Sections were stained with H&E for determination of cell morphology, Toluidine blue for the presence of negatively charged proteoglycans and Sirius red for presence and accumulation of collagen fibers. Immunohistochemical analysis was carried out using monoclonal mouse anti-human type I collagen or type II collagen antibodies (Research Diagnostics, Flanders, NJ) as the primary antibodies, and goat Anti-mouse HRP conjugated as the secondary antibody. The secondary antibody was detected by DAB metal-enhanced reagent. Immunohistochemical analysis of gel constructs was carried out by Histosery Inc. (Gaithburg, MD) using the Histostain-Plus Kit for AEC (Zymed Laboratories Inc., San Francisco, CA).
Biochemical Analyses
MSC/SELP-47 K constructs were lyophilized overnight and digested for 18 h at 65°C with 125 mg/ml papain (Worthington Biomedical, Lakewood, NJ) in buffer (100 mM phosphate, 10 mM EDTA, 2 mM cysteine, pH 6.5, using 1 ml enzyme per 5–10 mg dry sample. DNA content was determined spectrofluorometrically using PicoGreen DNA quantitation reagent (Molecular Probes, Eugene, OR). Sulphated-Glycosaminoglycan content in the polymer scaffolds was measured after hydrolysis in 20 U/ml papain at 60°C overnight. Samples were mixed with DMMB reagent (1,9-dimethylmethylene blue) and the absorbance was measured at 570 nm against a standard curve of chondroitin-4-sulfate (24). Total collagen content was determined from the reaction of hydroxyproline residues in collagen with p-dimethylaminobenzaldehyde (EM Science, Gibbstown, NJ) and chloramine-T (Mallinckrodt, Paris, KY) after acid hydrolysis of scaffold digests (6 N HCl at 115°C for 16–20 h) using a hydroproline: collagen conversion factor of 1:10 (25).
Data for the biochemical composition of the MSC/SELP-47 K constructs were statistically analyzed by one-way analysis of variance ANOVA (p<0.05) using SPSS (version 10.0; SPSS, Chicago, IL) software package. Statistical significance was determined at a value of p<0.05.
RESULTS
Viability of hMSC Encapsulated in SELP-47 K Hydrogels
To detect any possible adverse effects of SELP-47 K on cultured hMSCs the constructs were treated with WST-1 assay reagent, which measures cell viability relative to cell metabolic activity. There was an initial insignificant decrease in cell metabolic activity during culture in the chondrogenic medium with or without TGF-β3. This was in accordance with previous reports (26) which indicated loss of a portion of the marrow stromal cell population during culture, apparently through apoptosis. However, hMSCs remained metabolically active after 28 days of culture in chondrogenic medium in the presence or absence of TGF-β3 growth factor (Fig. 2). Observation of cells encapsulated in SELP-47 K gels after treatment with LIVE/DEAD reagent showed that the cells remained viable after 1, 14 and 28 days of culture compared to the control group treated with 0.1% saponin, a cytotoxic agent that resulted in 100% of cell death (Fig. 3).
Fig. 2.
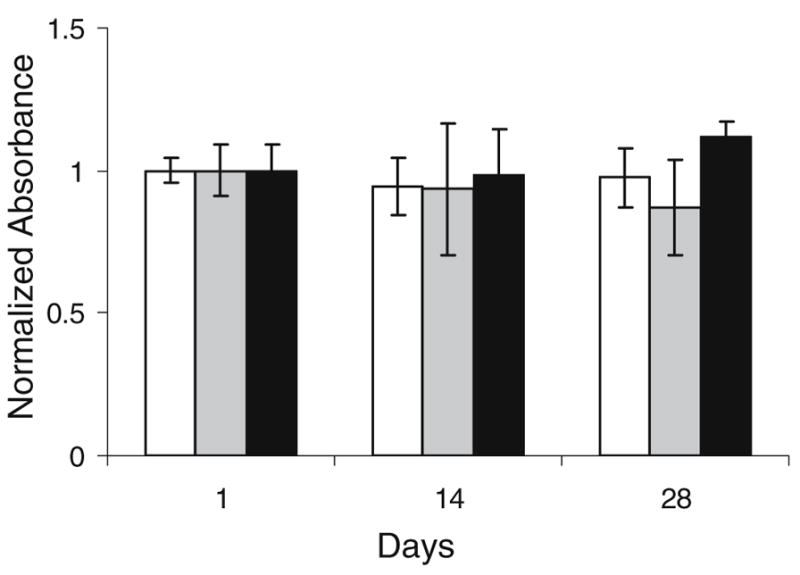
Determination of cell metabolic activity using WST-1 reagent for hMSC encapsulated in SELP-47 K hydrogel scaffolds and cultured in chondrogenic medium in the absence (white bar) and presence (gray bar) of TGF-β3 and (black bar) cell pellets cultured in the presence of TGF-β3 over 28 days. Values are calculated as absorbance per wet weight day×/absorbance per wet weight at day 1. Bars represent the mean ± standard deviation for n=3 samples.
Fig. 3.
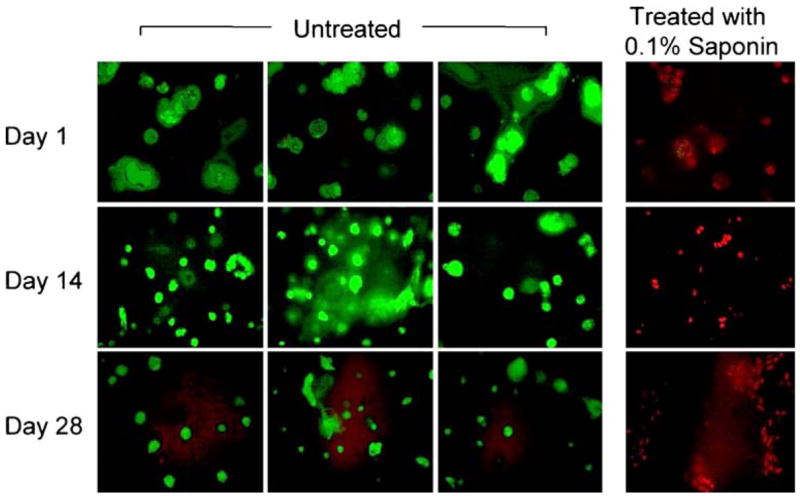
Confocal images of live (green) and dead (red) hMSC encapsulated in SELP-47 K hydrogel scaffolds cultured in chondrogenic medium in presence of TGF-β3 over 28 days. Control group was treated with 0.1% saponin for 10 min (cytotoxic agent). Bars represent the mean ± standard deviation for n=3 samples.
Histological and Immunohistochemical Analyses
The accumulation of chondrocytic extracellular matrix in hMSC/SELP-47 K constructs was examined by histology and immunohistochemistry. Histological H&E staining of fixed slides revealed that the cells adopted a rounded morphology and were embedded in significant amounts of extracellular matrix, regardless of the culture medium (Fig. 4). Histology sections of constructs treated with Sirius red, which stains collagen fibers red, showed increased accumulation of collagen in the vicinity of the cells by day 28, especially in samples cultured with TGF-β3. Additionally, Toluidine blue staining of the formed constructs showed S-GAG deposition in the intercellular regions at day 28, indicating the S-GAG was able to diffuse throughout the SELP-47 K gel (Fig. 4).
Fig. 4.

Histological staining of paraffin-embedded sections of hMSC/SELP-47 K constructs cultured in the absence (a–c) and presence (d–f) of TGF-β3 at day 28, stained with H and E (a and d), Sirius Red (b and e) and Toluidine Blue (c and f). Images were originally acquired at 100X magnification. Scale bar 20 μm.
Immunohistochemical labeling for collagen type I and II showed type I collagen on both the positive and negative TGF-β3 sections (Fig. 3). Interestingly, type II collagen staining was also observed in these samples, although more intense staining was clearly visible in the TGF-β3 sections (Fig. 5). This observation suggests the formation of newly synthesized cartilage-specific collagen, i.e. type II, in the SELP 47 K constructs.
Fig. 5.
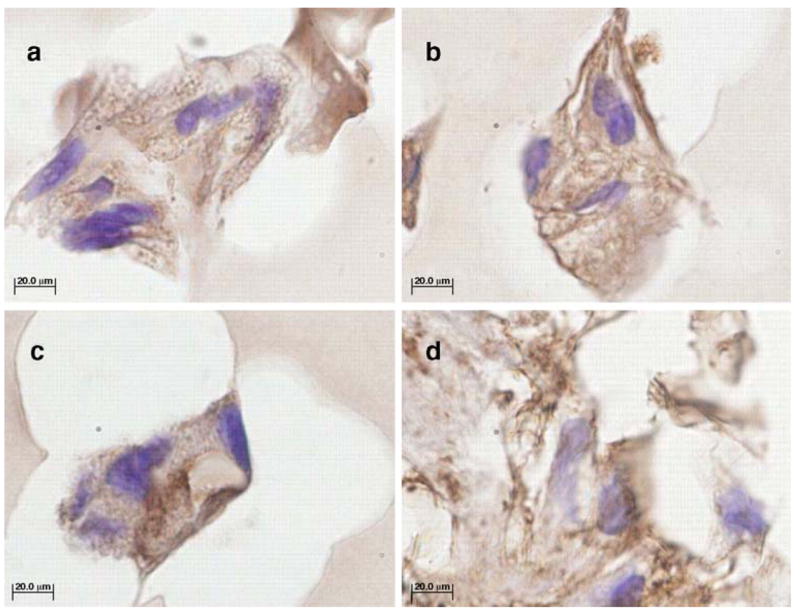
Immunohistochemical staining of paraffin-embedded sections -TGF-β3 (a and c) and + TGF-β3 (b and d) hMSC/SELP-47 K constructs with antibodies for type I collagen (a and b) and type II collagen (c and d) after 28 days of culture. Controls showed no staining for antigen (not shown). Images were originally acquired at 100X magnification. Scale bars=20 μm. Dark brown staining is positive for the collagen.
RT-PCR and Biochemical Analysis
The production of matrix proteins by the cells embedded in SELP-47 K gels was qualitatively detected with RT-PCR analysis (Fig. 6). In correlation with histological findings, agarose gel electrophoresis of the amplified RNA showed that the previously undifferentiated hMSCs shifted their genetic expression during the culture period. After 28 days, aggrecan, type II and X collagen and SOX9 transcription levels were significantly increased for +TGF-β3 constructs but was almost absent in constructs cultured in the absence of TGF-β3.
Fig. 6.
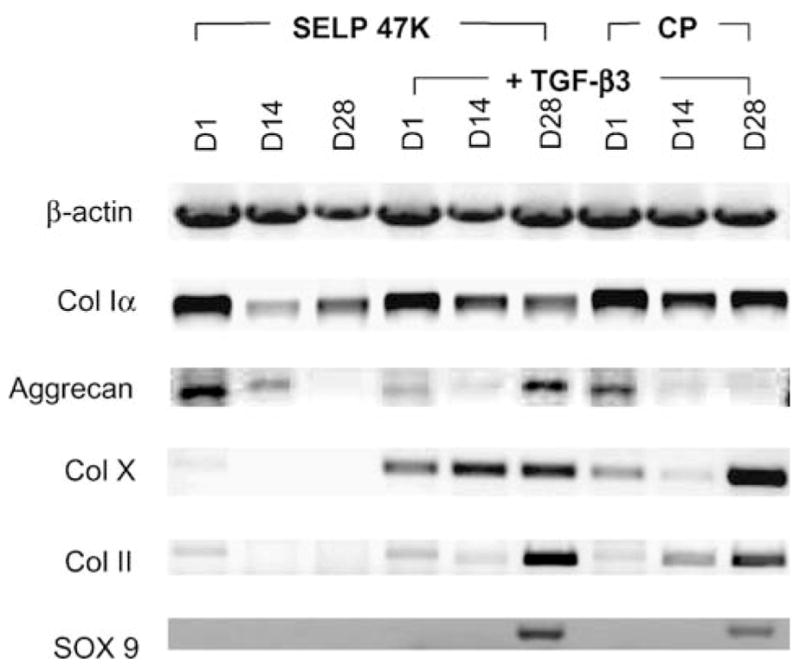
RT-PCR products for hMSC/SELP-47 K in the absence (lanes 1–3) and presence (lanes 4–6) of TGF-β3 and cell pellets (lanes 7–9) cultured in the presence of TGF-β3 after 1, 14 and 28 days (D1, D14 and D28, respectively). Primers used include β-actin (housekeeping), type I collagen, aggrecan, type X collagen, type II collagen and SOX 9 (from top to bottom of the gel).
In correlation with the RT-PCR and histologic findings, all constructs cultured in the presence of TGF-β3 exhibited a steady increase in S-GAG and total collagen accumulation. By day 28, S-GAG accumulation normalized to total DNA content was highest in hMSC/SELP-47 K constructs cultured in the presence of chondrogenic factors, whereas no increase was observed in the absence of TGF-β3 (Fig. 7a). The total collagen content normalized to DNA increased in hMSC/SELP-47 K constructs cultured under both conditions (Fig. 7b), with a more significant increase observed in the TGF-β3 samples.
Fig. 7.
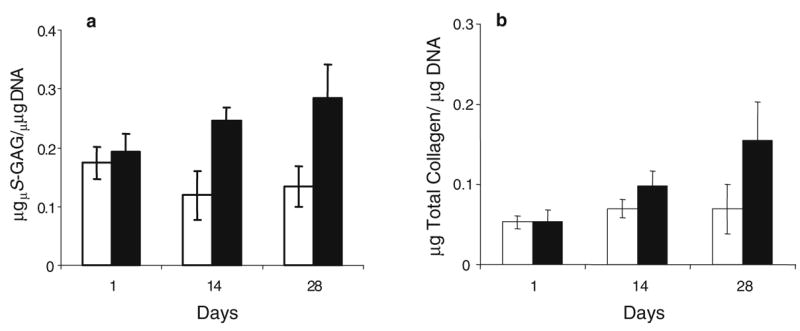
Results of biochemical assay for a S-GAG and b total collagen content per total DNA for day 1, 14 and 28 -TGF-β3 (white bar) and +TGF-β3 (black bar) constructs are depicted per total DNA content. Bars represent the mean ± standard deviation for n=3 samples.
DISCUSSION
Adult bone marrow derived mesenchymal stem cells (MSCs) are undifferentiated, multi-potential cells that can be easily isolated from adult bone marrow aspirates. They proliferate and differentiate in vitro to produce tissues such as bone, cartilage, fat, and tendon (27), making them an attractive cell source for autologous bone and cartilage tissue engineering. Thus far, few polymer scaffolds can provide the physicochemical and biological properties optimal for the repair or augmentation of cartilage defects using hMSCs. Hypothesizing that a biodegradable silk-elastinlike protein polymer hydrogel might be attractive for this application, we evaluated the in vitro chondrogenesis of hMSCs in this scaffold. When cultured in SELP scaffolds in chondrogenic medium, hMSCs expressed chondrogenic markers (aggrecan, type II and X collagen and SOX9) and accumulated cartilage-like matrix that contained sulfated glycosaminoglycans and collagen. The progression and extent of chondrogenesis were significantly increased, according to all measured parameters, in the presence of chondrogenic factor TGF-β3. Taken together, these data suggest that SELP hydrogel provides a suitable microenvironment for the chondrogenic differentiation of hMSCs in the presence of TGF-β3 in vitro.
SELPs are adaptable polymers where various combinations of silk-like and elastin-like blocks can produce customized gel formulations specific to a given application (11,19). So far SELP hydrogels have been considered for matrix-mediated drug and gene delivery (13,22,23). The ability to use recombinant genetic engineering techniques to customize and precisely control the polymer structural parameters resulted in successful manipulation of the physicochemical parameters of these hydrogels as well as release profiles of laden drugs or genetic materials (12).
Other investigators have shown that elastin like polymers (ELPs) exhibit mechanical behavior comparable to the intact protein (28). These polymers have been used as scaffolds for human adipose derived adult stem cells (hADAS) and showed the ability to promote their differentiation into a chondrogenic phenotype in three dimensional cultures (29). To achieve the mechanical strength required for functional cartilage repair, crosslinking of ELPs can be accomplished via radiative (30) or chemical (31) means or by in situ enzyme-initiated gelation via tissue transglutaminase (32). These methods may limit the use of ELPs as tissue engineering scaffolds due to possible harmful effects of radiation or chemical crosslinking on the cultured cells or the difficulty in precisely controlling enzymatic crosslinking to obtain the required mechanical strength.
Accordingly, the irreversible sol to gel transformation that characterizes members of the SELP family is an attractive property because it allows for injectable delivery of the cells at the diseased site as a fluid, transitioning peri-operatively to a solid. The gel-encapsulated cells remain physically in the tissue defect in which they are delivered, viable and capable of differentiation. Finally the ability to incorporate biorecognizable motifs with precise sequence in the polymer backbone can provide a suitable platform for cell recognition and differentiation in response to the biomaterial. Previous in vivo studies have shown that polymer SELP-47 K is biocompatible and exhibits very low immunogenic responses in test animals (13).
To our knowledge this is the first report examining the possibility of using SELP-47 K hydrogels as scaffolds for cell-based therapy. Our results demonstrate that hMSC isolated and encapsulated in SELP scaffolds survived and differentiated in this environment. Results of WST-1 cell viability assay and confocal microscopic observations after treatment of constructs with LIVE/DEAD reagent demonstrated that the hMSCs maintained their metabolic activity after 4 weeks of culture regardless of the presence of chondrogenic factor TGF-β3 in the culture medium. This is consistent with previous studies with other types of cells where minimal cytotoxicity was observed in cell cultures with SELP-47 K (13).
Histology and immunohistochemistry revealed that cartilage-specific matrix including glycosaminoglycans and type II collagen were produced in the hydrogels by previously undifferentiated MSCs in the presence of TGF-β3. Matrix production was also detected by RT-PCR after 14 days of culture in the presence of TGF-β3 where chondrocyte-specific genes such as aggrecan, type II and X collagen were observed, while SOX9 was detected after 28 days of culture. The mechanism causing the delay in SOX9 production is unclear. It may be due to partial differentiation of the cells that is usually associated with rounded cell morphology acquired in three-dimensional culture compared to flat-shape cells growing in two-dimensional culture (33–35). The inability to produce chondrocyte-specific genes in the absence of TGF-β3 demonstrates that SELP-47 K is an inert scaffold with regard to chondrogenesis. Therefore, the potential exists to enhance the bioactivity of the scaffold by the addition to the SELP material of chemical or biological regulators such as chondrogenic factors, collagen or chondroitin sulphate.
In correlation with the histological and RT-PCR findings, biochemical analysis showed that growing hMSC cells in SELP-47 K scaffolds in chondrogenic media in the presence of TGF-β3 resulted in greater content of S-GAG and total collagen compared to cultures without TGF-β3. However, values of S-GAG/DNA and collagen/DNA after 28 days of culture were low compared with chondrocytes cultured in other polymeric scaffolds such as poly(ethylene glycol)-dimethacrylate (PEGDM) and poly(lactide-co-glycolide) (PLGA) scaffolds (36,37), or other types of stem cells such as human adipose derived adult stem cells cultured in agarose, alginate and gelatin scaffolds (38–40). This may relate to the lower starting cell density in the constructs cultured in SELP-47 K (0.4×106 hMSC/scaffold). Further investigations will be required to determine the proliferation and apoptotic states of the cells in SELP-47 K to fully understand the effect of the biomaterial.
The potential of SELP-47 K in stem cell-based therapy remains to be further optimized and evaluated. First, a culture period extended over the 28 days of our study would heighten the differentiation and ECM production. Second, in SELP-47 K each eight elasin-like units are separated by four silk unit blocks, which creates a strong mechanical barrier that might limit the flexibility and amount of free space into which extracellular proteins could be deposited. It was previously noted that an increase in the mesh size of the hydrogel results in an increase of total collagen production (37). Accordingly it is possible that the small mesh size of SELP-47 K hydrogels is unfavorable for the diffusion of the produced extracellular matrix protein. A SELP with longer elastin blocks, and hence fewer silk blocks, per monomer unit could increase the mesh size of the hydrogel. This may have a positive influence on the differentiation of hMSC in these scaffolds and production of chondrogenic extracellular matrix that mimics native articular cartilage.
In summary, silk-elastinlike hydrogels are viable scaffolds for the in vitro chondrogenesis of hMSCs stimulated by TGF-β3. The uniquely well-controlled properties of genetically engineered polymers are well positioned to satisfy the diverse biomaterials requirements. This study adds to the selection of promising scaffold materials from which tissue engineers can choose for specific soft-tissue engineering applications.
CONCLUSION
In this study we demonstrated that the recombinant protein polymer SELP-47 K hydrogel can be used as a promising scaffold for the encapsulation and chondrogenesis of hMSCs. Evidence was provided that in vitro chondro-genesis can be enhanced by the addition of chondrogenic growth factor TGF-β3. The ability to precisely control the structure and physicochemical properties of SELPs by recombinant techniques is attractive. Moreover, in allowing controlled delivery of bioactive agents in a minimally invasive manner, these injectable recombinant scaffolds would be appealing for cell-based therapeutics.
Acknowledgments
Support by NIH (EB003447) is acknowledged.
ABBREVIATIONS
- Ala
alanine
- ANOVA
analysis of variance
- DNA
deoxyribonucleic acid
- ECM
extracellular matrix
- EDTA
ethylenediaminetetraacetic acid
- FBS
fetal bovine serum
- Gly
glycine
- H & E
haematoxylin and eosin
- hadAS
human adipose derived adult stem
- hMSCs
human mesenchymal stem cells
- MSCGM
mesenchymal stem cells general media
- PEGDM
poly(ethylene glycol)-dimethacrylate
- PLGA
poly(lactide-co-glycolide)
- Pro
proline
- RNA
ribonucleic acid
- RT-PCR
reverse transcriptase polymerase chain reaction
- Ser
serine
- SELP
silk-elastinlike protein polymer
- S-GAG
sulfated glycosaminoglycans
- TGF
transforming growth factor
- Val
valine
References
- 1.Kim HK, Moran ME, Salter RB. The potential for regeneration of articular cartilage in defects created by chondral shaving and subchondral abrasion. An experimental investigation in rabbits. J Bone Jt Surg, Am. 1991;73:1301–1315. [PubMed] [Google Scholar]
- 2.Brittberg M, Lindahl A, Nilsson A, Ohlsson C, Isaksson O, Peterson L. Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N Engl J Med. 1994;331:889–895. doi: 10.1056/NEJM199410063311401. [DOI] [PubMed] [Google Scholar]
- 3.Wakitani S, Goto T, Pineda SJ, Young RG, Mansour JM, Caplan AI, Goldberg VM. Mesenchymal cell-based repair of large, full-thickness defects of articular cartilage. J Bone Jt Surg, Am. 1994;76:579–592. doi: 10.2106/00004623-199404000-00013. [DOI] [PubMed] [Google Scholar]
- 4.Chaikof EL, Matthew H, Kohn J, Mikos AG, Prestwich GD, Yip CM. Biomaterials and scaffolds in reparative medicine. Ann NY Acad Sci. 2002;961:96–105. doi: 10.1111/j.1749-6632.2002.tb03057.x. [DOI] [PubMed] [Google Scholar]
- 5.Griffith LG, Naughton G. Tissue engineering-current challenges and expanding opportunities. Science. 2002;295:1009–1014. doi: 10.1126/science.1069210. [DOI] [PubMed] [Google Scholar]
- 6.Langer R, Tirrell DA. Designing materials for biology and medicine. Nature. 2004;428:487–492. doi: 10.1038/nature02388. [DOI] [PubMed] [Google Scholar]
- 7.Doukas J, Chandler LA, Gonzalez AM, Gu D, Hoganson DK, Ma C, Nguyen T, Printz MA, Nesbit M, Herlyn M, Crombleholme TM, Aukerman SL, Sosnowski BA, Pierce GF. Matrix immobilization enhances the tissue repair activity of growth factor gene therapy vectors. Hum Gene Ther. 2001;12:783–798. doi: 10.1089/104303401750148720. [DOI] [PubMed] [Google Scholar]
- 8.Chandler LA, Doukas J, Gonzalez AM, Hoganson DK, Gu DL, Ma C, Nesbit M, Crombleholme TM, Herlyn M, Sosnowski BA, Pierce GF. FGF2-Targeted adenovirus encoding platelet-derived growth factor-B enhances de novo tissue formation. Molec Ther. 2000;2:153–160. doi: 10.1006/mthe.2000.0102. [DOI] [PubMed] [Google Scholar]
- 9.Hubbell JA, Massia SP, Desai NP, Drumheller PD. Endothelial cell-selective materials for tissue engineering in the vascular graft via a new receptor. Biotechnology (NY) 1991;9:568–572. doi: 10.1038/nbt0691-568. [DOI] [PubMed] [Google Scholar]
- 10.Cappello J, Ghandehari H. Engineered protein polymers for drug delivery and biomedical applications. Adv Drug Deliv Rev. 2002;54:1053–1159. doi: 10.1016/s0169-409x(02)00063-7. [DOI] [PubMed] [Google Scholar]
- 11.Haider M, Megeed Z, Ghandehari H. Genetically engineered polymers: status and prospects for controlled release. J Control Release. 2004;95:1–26. doi: 10.1016/j.jconrel.2003.11.011. [DOI] [PubMed] [Google Scholar]
- 12.Haider M, Leung V, Ferrari F, Crissman J, Powell J, Cappello J, Ghandehari H. Molecular engineering of silk-elastinlike polymers for matrix-mediated gene delivery: biosynthesis and characterization. Mol Pharmacol. 2005;2:139–150. doi: 10.1021/mp049906s. [DOI] [PubMed] [Google Scholar]
- 13.Cappello J, Crissman JW, Crissman M, Ferrari FA, Textor G, Wallis O, Whitledge JR, Zhou X, Burman D, Aukerman L, Stedronsky ER. In-situ self-assembling protein polymer gel systems for administration, delivery, and release of drugs. J Control Release. 1998;53:105–117. doi: 10.1016/s0168-3659(97)00243-5. [DOI] [PubMed] [Google Scholar]
- 14.Manno M, Emanuele A, Martorana V, San Biagio PL, Bulone D, Palma-Vittorelli MB, Mcpherson DT, Xu J, Parker TM, Urry DW. Interaction of processes on different length scales in a bioelastomer capable of performing energy conversion. Biopolymers. 2001;59:51–64. doi: 10.1002/1097-0282(200107)59:1<51::AID-BIP1005>3.0.CO;2-8. [DOI] [PubMed] [Google Scholar]
- 15.Panitch A, Yamaoka T, Fournier MJ, Mason TL, Tirrell DA. Design and biosynthesis of elastin-like artificial extracellular matrix proteins containing periodically spaced fibronectin CS5 domains. Macromolecules. 1999;32:1701–1703. [Google Scholar]
- 16.Petka WA, Harden JL, McGrath KP, Wirtz D, Tirrell DA. Reversible hydrogels from self-assembling artificial proteins. Science. 1998;281:389–392. doi: 10.1126/science.281.5375.389. [DOI] [PubMed] [Google Scholar]
- 17.Urry DW, Harris CM, Luan CX, Luan C-H, Gowda DC, Parker TM, Peng SQ, Xu J. Transductional protein-based polymers as new controlled release vehicles. In: Park K, editor. Controlled Drug Delivery: Challenges and Strategies. American Chemical Society; Washington, DC: 1997. pp. 405–436. [Google Scholar]
- 18.Urry DW, Pattanaik A, Accavitti MA, Luan CX, Mcpherson DT, Xu J, Gowda DC, Parker TM, Harris CM, Jing N. Transductional elastic and plastic protein-based polymers as potential medical devices. In: Domb AJ, Kost J, Wiseman DM, editors. Handbook of Biodegradable Polymers. Harwood Academic; Amsterdam: 1997. pp. 367–386. [Google Scholar]
- 19.Megeed Z, Cappello J, Ghandehari H. Genetically engineered silk-elastinlike protein polymers for controlled drug delivery. Adv Drug Deliv Rev. 2002;54:1075–1091. doi: 10.1016/s0169-409x(02)00063-7. [DOI] [PubMed] [Google Scholar]
- 20.Dinerman AA, Cappello J, Ghandehari H, Hoag SW. Swelling behavior of a genetically engineered silk-elastinlike protein polymer hydrogel. Biomaterials. 2002;23:4203–4210. doi: 10.1016/s0142-9612(02)00164-3. [DOI] [PubMed] [Google Scholar]
- 21.Megeed Z, Haider M, Li D, O’Malley BW, Jr, Cappello J, Ghandehari H. In vitro and in vivo evaluation of recombinant silk-elastinlike hydrogels for cancer gene therapy. J Control Release. 2004;94:433–445. doi: 10.1016/j.jconrel.2003.10.027. [DOI] [PubMed] [Google Scholar]
- 22.Megeed Z, Cappello J, Ghandehari H. Controlled release of plasmid DNA from a genetically engineered silk-elastinlike hydrogel. Pharm Res. 2002;19:954–959. doi: 10.1023/a:1016406120288. [DOI] [PubMed] [Google Scholar]
- 23.Dinerman AA, Cappello J, Ghandehari H, Hoag SW. Solute diffusion in genetically engineered silk-elastinlike protein polymer hydrogels. J Control Release. 2002;82:277–287. doi: 10.1016/s0168-3659(02)00134-7. [DOI] [PubMed] [Google Scholar]
- 24.Farndale RW, Buttle DJ, Barrett AJ. Improved quantitation and discrimination of sulphated glycosaminoglycans by use of dimethylmethylene blue. Biochim Biophys Acta. 1986;883:173–177. doi: 10.1016/0304-4165(86)90306-5. [DOI] [PubMed] [Google Scholar]
- 25.Woessner JF., Jr The determination of hydroxyproline in tissue and protein samples containing small proportions of this imino acid. Arch Biochem Biophys. 1961;93:440–447. doi: 10.1016/0003-9861(61)90291-0. [DOI] [PubMed] [Google Scholar]
- 26.Sekiya I, Vuoristo JT, Larson BL, Prockop DJ. In vitro cartilage formation by human adult stem cells from bone marrow stroma defines the sequence of cellular and molecular events during chondrogenesis. Proc Natl Acad Sci USA. 2002;99:4397–4402. doi: 10.1073/pnas.052716199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–147. doi: 10.1126/science.284.5411.143. [DOI] [PubMed] [Google Scholar]
- 28.Lee J, Macosko CW, Urry DW. Elastomeric polypentapeptides cross-linked into matrixes and fibers. Biomacromolecules. 2001;2:170–179. doi: 10.1021/bm0000900. [DOI] [PubMed] [Google Scholar]
- 29.Betre H, Ong SR, Guilak F, Chilkoti A, Fermor B, Setton LA. Chondrocytic differentiation of human adipose-derived adult stem cells in elastin-like polypeptide. Biomaterials. 2006;27:91–99. doi: 10.1016/j.biomaterials.2005.05.071. [DOI] [PubMed] [Google Scholar]
- 30.Nagapudi K, Brinkman WT, Leisen JE, Huang L, Mcmillan RA, Apkarian RP, Conticello VP, Chaikof EL. Photomediated solid-state cross-linking of an elastin-mimetic recombinantprotein polymer. Macromolecules. 2002;35:1730–1737. [Google Scholar]
- 31.Mcmillan RA, Conticello VP. Synthesis and characterization of elastin-mimetic protein gels derived from a well-defined polypeptide precursor. Macromolecules. 2000;33:4809–4821. [Google Scholar]
- 32.Mchale MK, Setton LA, Chilkoti A. Synthesis and in vitro evaluation of enzymatically cross-linked elastin-like polypeptide gels for cartilaginous tissue repair. Tissue Eng. 2005;11:1768–1779. doi: 10.1089/ten.2005.11.1768. [DOI] [PubMed] [Google Scholar]
- 33.Johnstone B, Hering TM, Caplan AI, Goldberg VM, Yoo JU. In vitro chondrogenesis of bone marrow-derived mesenchymal progenitor cells. Exp Cell Res. 1998;238:265–272. doi: 10.1006/excr.1997.3858. [DOI] [PubMed] [Google Scholar]
- 34.Diduch DR, Jordan LC, Mierisch CM, Balian G. Marrow stromal cells embedded in alginate for repair of osteochondral defects. Arthroscopy. 2000;16:571–577. doi: 10.1053/jars.2000.4827. [DOI] [PubMed] [Google Scholar]
- 35.Williams CG, Kim TK, Taboas A, Malik A, Manson P, Elisseeff J. In vitro chondrogenesis of bone marrow-derived mesenchymal stem cells in a photopolymerizing hydrogel. Tissue Eng. 2003;9:679–688. doi: 10.1089/107632703768247377. [DOI] [PubMed] [Google Scholar]
- 36.Freed L, Vunjak-Novakovic G. Tissue engineering of cartilage. In: BJ, editor. The Biomedical Engineering Handbook. CRC; Boca Raton, FL: 1995. pp. 1778–1796. [Google Scholar]
- 37.Bryant SJ, Anseth KS. Hydrogel properties influence ECM production by chondrocytes photoencapsulated in poly (ethylene glycol) hydrogels. J Biomed Mater Res. 2002;59:63–72. doi: 10.1002/jbm.1217. [DOI] [PubMed] [Google Scholar]
- 38.Barry F, Boynton RE, Liu B, Murphy JM. Chondrogenic differentiation of mesenchymal stem cells from bone marrow: differentiation-dependent gene expression of matrix components. Exp Cell Res. 2001;268:189–200. doi: 10.1006/excr.2001.5278. [DOI] [PubMed] [Google Scholar]
- 39.Kavalkovich KW, Boynton RE, Murphy JM, Barry F. Chondrogenic differentiation of human mesenchymal stem cells within an alginate layer culture system. In Vitro Cell Dev Biol, Anim. 2002;38:457–466. doi: 10.1290/1071-2690(2002)038<0457:cdohms>2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 40.Ponticiello MS, Schinagl RM, Kadiyala S, Barry FP. Gelatin-based resorbable sponge as a carrier matrix for human mesenchymal stem cells in cartilage regeneration therapy. J Biomed Mater Res. 2000;52:246–255. doi: 10.1002/1097-4636(200011)52:2<246::aid-jbm2>3.0.co;2-w. [DOI] [PubMed] [Google Scholar]


