Abstract
Disseminated breast tumour cells in sentinel lymph nodes (SNs) were evaluated by quantitative real-time PCR and the sensitivity of this assay was compared to the routine histological analysis. First, several candidate marker genes were tested for their specificity in axillary lymph nodes (ALN) of 50 breast cancer patients and 43 women without breast cancer. The marker gene panel selected, designed to detect the mRNA of CK19, p1B, EGP2 and SBEM, was subsequently applied to detect metastases in 70 SNs that were free of metastases as determined by standard histological evaluation. Remarkably, seven negative SNs showed increased marker gene expression, suggesting the presence of (micro) metastases. Four of these seven SNs positive by real-time PCR proved to contain tumour deposits after careful review of the slides or further sectioning of the paraffin-embedded material. In three PCR positive SNs, however, no tumour cells were found by haematoxylin and eosin staining (H&E) and immunohistologically analysis. The quantitative real-time PCR assay with multiple mRNA markers for the detection of disseminated breast cancer cells in SNs thus resulted in an upstaging of SNs containing metastastic disease of 10% compared to the routine histological analysis. The application of this technique may be of clinical relevance, as it is suggested that micrometastatic disease in SNs are associated with further nodal non-SN metastases in breast cancer.
Keywords: sentinel lymph nodes, metastases, breast cancer, quantitative real-time PCR
In breast cancer to date, the axillary lymph node (ALN) status remains the most valuable individual prognostic factor for disease course and recurrence (McGuire, 1987; Foster, 1996). Involvement of lymph nodes and the number of lymph nodes harbouring metastases at primary diagnosis have an inverse relationship with the disease prognosis, meaning that patients with lymph nodes free of metastases have a better outcome (Fisher et al, 1983; Carter et al, 1989; Hellman, 1994; Quiet et al, 1995; Saimura et al, 1999). However, 20–30% of node-negative patients will develop a relapse in 5–10 years after diagnosis (Kamby et al, 1991; Hellman, 1994). In addition, it is known that 20–30% of node-positive patients are long-term survivors (Rosen et al, 1989; Joensuu et al, 1998). Based on these obvious shortcomings of the lymph node status, new procedures and markers are continuously investigated with regard to their prognostic value. Recently, it was shown that primary tumours themselves already contain a gene expression profile that is strongly predictive of metastasis and poor survival (van de Vijver et al, 2002; van't Veer et al, 2002), thereby challenging treatment choice based on routine prognostic markers in the future. However, until gene-expression profiling will increasingly be used for clinical decision-making, the nodal status remains a main prognostic factor.
Until recently, the standard treatment for patients with operable breast cancer included the complete dissection of 10–30 ALN. More than half of these patients were found to have metastases-free lymph nodes and thus had been subjected to unnecessary surgical risks and complications (Giuliano et al, 1997). A less invasive method for the assessment of lymph node status is the sentinel lymph node biopsy (SLNB). Thereby, the lymphatic route of tumour cells to the lymph node(s) that primarily drains the tumour and most likely harbours metastatic disease are mapped (Giuliano et al, 1994). The sentinel lymph node (SN) is highly predictive of the histo-pathology of the remaining lymphatic basin and can accurately predict axillary nodal status in at least 98% of cases (Giuliano et al, 1995, 1997). Furthermore, the SN biopsy allows a more extensive and focused search for metastases in one or two nodes, in contrast to the present limited analysis of the multiple lymph nodes from a complete dissection. Multiple step sectioning and immunohistochemistry staining of the SN increases the accuracy of axillary staging in breast cancer patients compared with axillary lymph node dissection (ALND) plus routine histo-pathologic examination of lymph nodes (Giuliano et al, 1995). In this way additional micrometastases are identified, which may relate to prognosis (Bourez and Rutgers, 2001; Tjan-Heijnen et al, 2001). The SN procedure therewith provides an accurate and feasible alternative for complete ALND as a staging tool in breast cancer and can prevent lymph node negative women from unnecessary surgery (Bourez et al, 2002).
In the study presented, a sensitive real-time PCR approach is used for the detection of metastases in SNs. We have examined several candidate marker genes, with particular emphasis on sensitivity and specificity of these genes in ALN of patients with and without metastases. Finally, a marker panel of four genes comprising cytokeratin 19 (CK19), trefoil factor-3 (p1B), epithelial glycoprotein-2 (EGP-2) and small breast epithelial mucin (SBEM) was used to compare the quantitative real-time PCR detection method to the routine analysis of SNs, including multiple step sectioning and immunohistochemical staining.
MATERIAL AND METHODS
Axillary and SN specimens from breast cancer patients
Axillary (n=50) and SNs (n=89) of breast cancer patients were selected from the fresh-frozen tissue bank of the Netherlands Cancer Institute/Antoni van Leeuwenhoek hospital (NKI/AvL). As negative controls 43 ALNs were obtained from patients without breast cancer undergoing a preventive breast ablation or a lymph node biopsy for analysis of lymphoma. The latter group was classified to have benign enlarged lymph nodes. The SN procedure was introduced in 1996 at the NKI/AvL (Rutgers et al, 1998) and since then performed on more than 700 patients. The SN was removed after localisation of the node by combination of radioactivity measurement by a gamma detection probe (Neoprobe 100/1500, Neoprobe Corporation, Dublin, UK) exploiting the remaining activity after preoperative lymphoscintigraphy, and visually by peroperative injection of Patent Blue V (Blue Patente V; Laboratoire Guerbet, Aulnay-sous-Bois, France) (Doting et al, 2000).
The ALNs obtained at preventive breast ablation and the lymph nodes retrieved from analysis of lymphoma were bisected, snap frozen in liquid nitrogen and stored at −80°C. SNs were subjected to frozen section evaluation. Sentinel lymph nodes up to 0.5 cm were completely embedded in Tissue Tek (Sakura Finetek, Zoeterwoude, The Netherlands), larger SNs were bisected or lamellated in slices of 0.2 cm and separately sectioned. Frozen section cutting was aimed at obtaining a complete cross section at a single level and preventing tissue loss. Before the frozen section procedure, the microtome was carefully cleaned to avoid contamination; all lost tissue was collected and stored at −80°C. The amount of lost tissue varied, and was estimated at least 20 sections of 10 μm. Remaining SN tissue was examined after formalin fixation and paraffin embedding by haematoxylin and eosin (H&E) staining and immunohistochemistry at three levels (150 μm distance). CAM 5.2 (Becton Dickinson, San Jose, USA) was used as antibody.
RNA isolation and quantitative real-time PCR
RNA was isolated from 30 tissue sections of 30 μm thickness made from each axillary node, and from the lost frozen tissue of the SNs, using RNAzol B (Campro Scientific, Amersfoort, The Netherlands). In total, 1 μg total RNA was used for cDNA synthesis (20 μl), as described previously (Lambrechts et al, 1999).
Based on the published genomic sequences of CK19, p1B, EGP-2, PS2, mammaglobin and SBEM, the sequences of the real-time quantitative PCR primers (Sigma Genosys, Cambridge, UK) and of the 5′-fluorescently FAM labelled probes (Applied Biosystems, Nieuwerkerk a/d IJssel, The Netherlands) were selected using the Perkin-Elmer Primer Express® software (PE, Foster City, USA) (Table 1). All primers were designed to be intron-spanning to preclude amplification of genomic DNA. To normalise relative levels of expression, commercially available primers and probes for the housekeeping genes, glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and β-actin were used (Applied Biosystems).
Table 1. Primer and probe sequences of each breast cancer marker gene for real-time PCR amplification. All sequences are written 5′ → 3′.
| Marker gene | Genbank accession no. | Primers | Probe (5′FAM-3′TAMRA) |
|---|---|---|---|
| p1B | (L15203) | Sense: CTGAGGAGTACGTGGGCCTG | CTGCAAACCAGTGTGCCGTGCC |
| Antisense: AGTCCACCCTGTCCTTGGC | |||
| PS2 | (X00474) | Sense: GAGGCCCAGACAGAGACGTG | CTGCTGTTTCGACGACACCGTTCG |
| Antisense: CCCTGCAGAAGTGTCTAAAATTCA | |||
| CK19 | (NM002276) | Sense: CTACAGCCACTACTACACGAC | CACCATTGAGAACTCCAGGATTGTCCTGC |
| Antisense: CAGAGCCTGTTCCGTCTCAAA | |||
| EGP2 | (M32306) | Sense: CAGTTGGTGCACAAAATACTGTCA | TTGCTCAAAGCTGGCTGCCAAATGTT |
| Antisense: CCATTCATTTCTGCCTTCATCA | |||
| SBEM | (AF414087) | Sense: CTCTTGGGGAGTTTTCCATCTTTCTG | CCCAGAATCCGACAACAGCTGCTCC |
| Antisense: CTTCATCATCAGCAGGACCAGTAG | |||
| MaGl | (AF015224) | Sense: TTCTTAACCAAACGGATGAAACTCT | TGCTGTCATATATTAATTGCATAAACACCTCAACATTG |
| Antisense: GGTCTTGCAGAAAGTTAAAATAAATCAC |
Serially diluted cDNA synthesised from RNA isolated and pooled from 6 × 106 cells of five breast cancer cell lines (MCF7, CAMA, T47D, MPL13, SkBr3 (American Tissue Culture Collection, Rockville, USA)), respectively, was used to generate standard curves for control and marker gene expression. For all cDNA dilutions, the fluorescence was detected from 0 to 50 PCR cycles for the control and marker gene in singleplex reactions and resulted in the CT-value for each cDNA dilution and each target (CT-value (threshold cycle): the PCR cycle at which a significant increase in fluorescence is detected, due to the exponential accumulation of PCR products, represented in arbitrary units (TaqMan Universal PCR Master Mix Protocol, Applied Biosystems) (Bieche et al, 1999). The quantities found for the GAPDH control and marker gene were used to calculate the relative quantity of control and marker gene expression in ALN and SNs. The second control gene, β-actin, was only used for the confirmation of GAPDH expression. Each experiment was performed in triplicate. The quality control of the PCR reactions was assessed by standardised PCR conditions, including in each experiment a genomic DNA control and a negative nontemplate control.
Statistics
The QDA score function, as defined earlier (Bosma et al, 2002), was calculated from the expression levels of the six marker genes CK19, p1B, EGP-2, SBEM, PS2 and mammaglobin tested, in different combinations. QDA is a statistical technique to find the combination of quadratic and linear functions of variables (e.g. marker genes), which leads to an optimal separation between groups (e.g. tumour cell positive and negative lymph nodes). It is a generalisation of the more familiar Fisher's Linear Discrimination Analysis (LDA), which allows only linear functions. QDA performs better than LDA if the groups differ not only with respect to the means of the variables but also with respect to standard deviations and/or correlations. A positive discriminant score of the four marker genes selected, CK19, p1B, EGP-2 and SBEM, indicates the presence of breast tumour cells, a negative discriminant score indicates the absence of tumour cells. The discriminant threshold score separating these two groups is zero.
Sensitivity
A cDNA pool of three tumour cell positive ALNs was diluted 1 : 10, 1 : 100, 1 : 1000, 1 : 10 000 and 1 : 100 000 in a cDNA pool of three control ALNs. For all cDNA dilutions the fluorescence was detected from 0 to 50 PCR cycles, for each individual marker gene in triplicates. The CT-values obtained were compared to serially diluted cDNA synthesised from RNA isolated from 5000 MCF7 cells. The dilution of MCF7 RNA of 1 : 10, 1 : 100, 1 : 1000, 1 : 10 000 and 1 : 100 000 corresponds to the total RNA amount obtained from 500, 50, 5, 0.5 and 0.05 MCF7 cells, respectively. All standard curves for these experiments were generated using the cell line mix as described for the marker and control gene real-time PCR reactions.
RESULTS
Marker gene selection for the detection of metastatic breast tumour cells in ALN
Several potential marker genes were tested to determine genes that are highly expressed in 50 ALNs of breast cancer patients with pathological verified tumour involvement, but that are at the same time expressed at low levels in 43 ALNs of women without evidence of breast cancer. Gene expression was quantitated for the four breast cancer marker genes CK19, PS2, EGP-2 and p1B, which we used earlier for the detection of circulating tumour cells in peripheral blood of breast cancer patients (Bosma et al, 2002). Furthermore, the genes mammaglobin, epidermal growth factor receptor (EGFR) and SBEM were tested, described in the literature as breast cancer marker genes (De Luca et al, 2000; Mitas et al, 2001; Miksicek et al, 2002; Zehentner et al, 2002). For each gene, CT-values for negative control lymph nodes and metastatic breast cancer lymph nodes were obtained from triplicate reactions. For all ALN samples, the expression level for each of the marker genes relative to the breast cancer cell line mix standard curve was calculated and corrected for the input of cDNA based on the GAPDH control (see Materials and Methods) (Table 2).
Table 2. Expression levels (quantities) for each housekeeping gene relative to the standard curve and relative quantities for each marker gene relative to the standard curve and corrected for the input of cDNA based on the GAPDH control in arbitrary units (au).
| Housekeeping gene | Tissue | Median quantity | Range |
|---|---|---|---|
| GAPDH | ALNneg | 0.30000 | 0.01800–1.85000 |
| ALNpos | 0.15500 | 0.00002–1.90000 | |
| β-Actin | ALNneg | 0.76250 | 0.09800–2.75000 |
| ALNpos | 0.28000 | 0.00001–1.35000 | |
| Marker gene | Tissue | Median relative quantity | Range |
| CK19 | ALNneg | 0.00001 | 0.000003–0.000103 |
| ALNpos | 0.04931 | 0.00022–8.23529 | |
| p1B | ALNneg | 0.01778 | 0.00009–0.23583 |
| ALNpos | 0.13400 | 0.00017–55.5556 | |
| EGP2 | ALNneg | 0.00035 | 0.00009–0.00270 |
| ALNpos | 0.07300 | 0.00112–0.69109 | |
| SBEM | ALNneg | 0.00047 | 0.00011–0.02112 |
| ALNpos | 0.07800 | 0.00073–84.7619 | |
| PS2 | ALNneg | 0.00001 | 0.00000–0.00026 |
| ALNpos | 0.01319 | 0.00000–10.0457 | |
| Mammaglobin | ALNneg | 0.000001 | 0.00000–0.00003 |
| ALNpos | 0.00014 | 0.00001–1.16994 | |
| EGFR | ALNneg | 3.06956 | 0.80620–10.1852 |
| ALNpos | 1.45317 | 0.05461–58.2897 |
ALNneg=tumour cell free control axillary lymph nodes; ALNpos=axillary lymph nodes harbouring breast tumour cells.
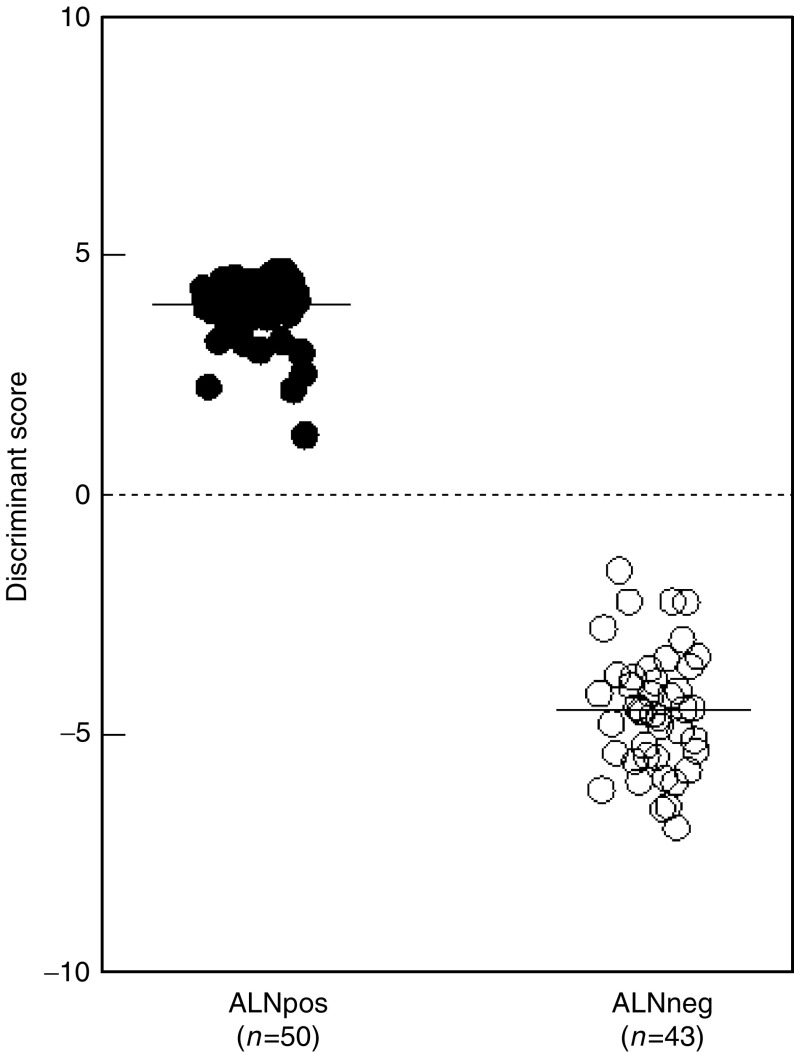
We observed that the median expression levels of EGFR were higher in the control ALNs than in the tumour cell positive lymph nodes (Table 2). Thus, this gene was not useful for the detection of metastatic breast cancer in lymph nodes, whereas the median expression levels for CK19, p1B, EGP-2, SBEM, PS2 and mammaglobin were significantly higher in the ALNs containing metastatic breast cancer (Table 2), determined by the Mann–Whitney test (data not shown). To optimally use the expression levels of these latter marker genes to separate lymph nodes with and without tumour cell involvement, the quadratic discriminant analysis (QDA) was employed (Bosma et al, 2002) (see Material and Methods). A combination of four marker genes was shown to have the highest specificity in separating tumour cell negative and positive lymph nodes (data not shown). In the following step, we tested different combinations of sets of four genes to determine the marker gene panel that gave the largest discriminant score between negative and tumour cell positive lymph nodes. The marker set including CK19, p1B, EGP2 and SBEM gave the largest separation between breast tumour cell negative and positive ALNs with zero misclassified normal control lymph nodes (Figure 1). The median for the positive and negative lymph nodes in the discriminant score is 4.05 (range: 1.25–467) and −4.51 (range: −6.97 to −2.79), respectively (P-value <0.0001). The threshold score discriminating the two groups is zero. A cross validation also showed a 100% separation between the tumour cell positive and negative ALNs (data not shown). These results indicate that the real-time PCR analysis using a marker panel of genes is specific and at least as sensitive as the standard histological analysis.
Figure 1.
Discriminant score of expression of the four marker genes CK19, p1B, EGP2 and SBEM in ALN of patients with and without breast cancer. The median expression levels for the markergene panel within a group are indicated by a horizontal line. Closed circles represent breast cancer patients, open circles women without breast cancer. The discriminant score separating the two groups is indicated by a dashed line.
Sensitivity
To define the value of the quantitative real-time PCR analysis for the detection of metastatic breast cancer cells, we determined the sensitivity of the marker panel. Working with a solid tissue such as lymph nodes makes it difficult to dilute breast tumour cells directly in the tissue like it is usually done for sensitivity assays in peripheral blood (Kufer et al, 2002). Instead, we pooled cDNA of three tumour cell positive ALNs and serially diluted this cDNA corresponding to five log steps in a cDNA pool of three control ALNs. A real-time PCR was performed for all cDNA dilutions, for each marker gene individually, and the detection limit of the assay was defined. To get an indication of how many breast tumour cells can be detected using the quantitative PCR approach, we compared the CT-values obtained to those of serially diluted cDNA synthesise from RNA isolated from 5000 MCF7 cells. The dilution of 10−1 up to 10−5 corresponds to 500 to 0.05 MCF7 cells, respectively. The standard curves for all experiments were generated from a pool of five breast cancer cell lines, which expressed all four marker genes tested at similar high levels (Figure 2A). The limit of detection of the cDNA pool of tumour cell positive lymph nodes diluted in a pool of negative lymph nodes is shown in Figure 2B for every of the four marker genes individually. The CT-value of p1B reaches a plateau when less than 50 MCF7 cells are present, indicating that a sensitivity of 50 breast tumour cells can be reliably detected by this marker. The detection limit for SBEM corresponds to the presence of five MCF7 cells and for CK19 and EGP2 to as low as 0.5 MCF7 cells (Figure 2B).
Figure 2.
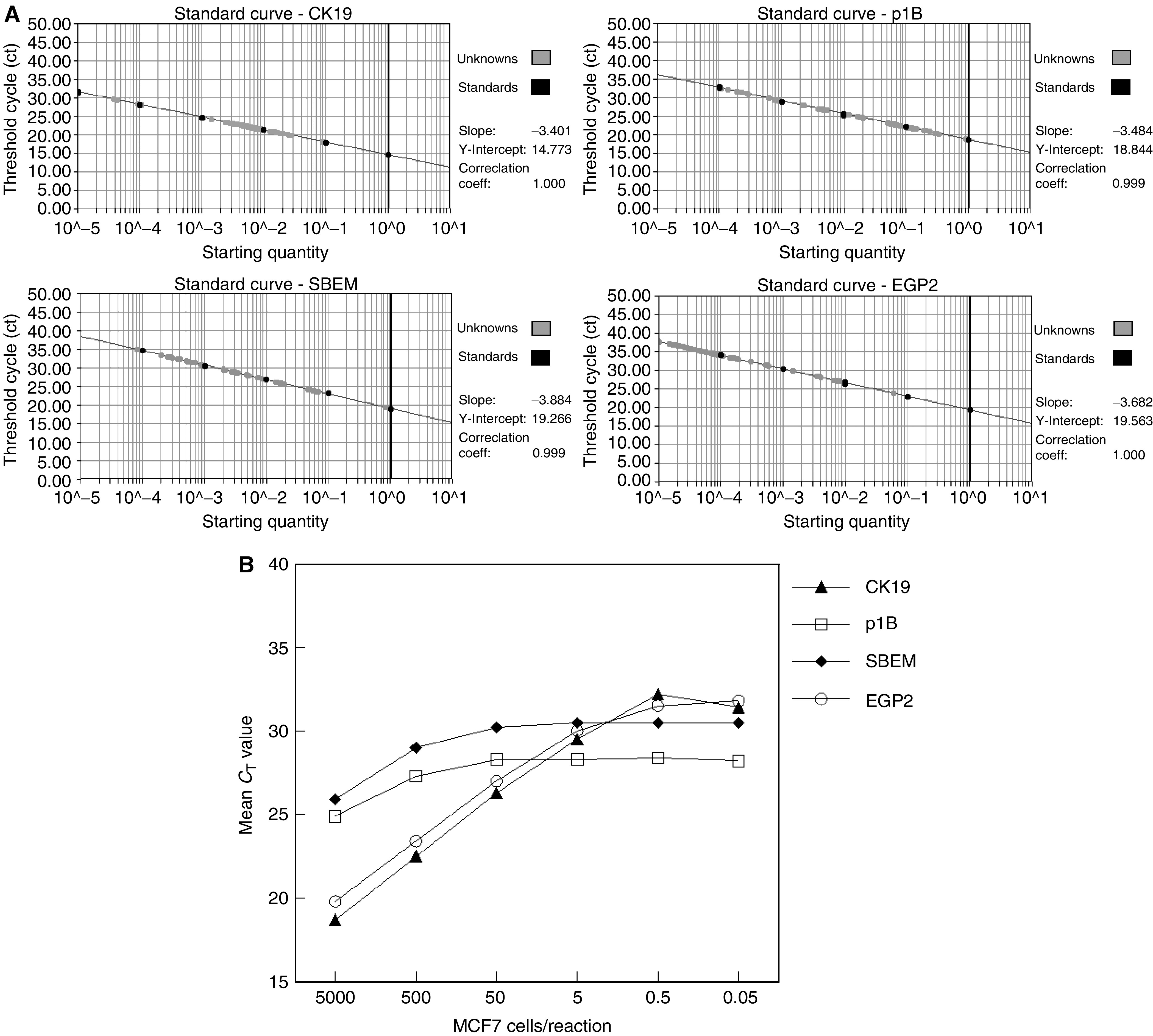
(A) Standard curves of CK19, p1B, EGP2 and SBEM, used for determining the sensitivity of the marker genes (black circles). Grey circles represent lymph node samples tested. The threshold cycle (CT-value) is plotted against the starting quantity of a pool of five breast cancer cells lines. (B) Sensitivity of the markerpanel CK19, p1B, EGP2 and SBEM real-time PCR defined by analysis of serial dilution of a cDNA pool of three tumour cell positive ALN in a cDNA pool of three control lymph nodes. Subsequently, CT-values obtained were compared to serially diluted MCF7 cDNA, for each marker gene individually. The detection limit of p1B with regard to the background expression of control lymph nodes corresponds to the amount of 50 MCF7 cells, for SBEM to five MCF7 cells and for CK19 and EGP2 to 0.5 MCF7 cells.
Marker gene expression in SNs
To determine whether real-time PCR analysis was also capable of detecting metastases in SNs, we analysed 70 SNs without evidence of metastatic breast tumour cells following standard histological analysis. The median discriminant score for the tumour cell negative SNs was, as for the control ALNs, smaller than zero, the threshold score that separates the tumour cell negative from the tumour cell positive ALN group. Additionally, 19 histologically proven positive SNs were analysed using the marker gene panel to stage the accuracy of the real-time approach. The median discriminant score is lower compared to the tumour cell positive ALNs. However, the set of marker genes is still over expressed in the positive SNs and the discriminant scores of all 19 SN are above the threshold of zero, indicating the presence of metastatic breast tumour cells. The marker panel containing CK19, EGP-2, p1B and SBEM can therefore also be applied for the evaluation of SNs.
Remarkably, the evaluation of the 70 histologically negative SNs on an individual basis showed for seven SNs a positive discriminant score of the four mRNA marker genes, indicating the presence of metastatic tumour cells (Figure 3). Subsequently, the frozen tissue and paraffin sections of these seven SNs, tumour cell positive by real-time PCR, were reviewed. The slides of two cases revealed tumour deposits at review, which were missed at the first evaluation. In one of these SNs, tumour was present in the frozen section slide, in the other one in the H&E stained slide of the first level. In both cases the deposits were cell clusters with a diameter smaller than 0.2 cm (micrometastases). No evidence of metastases was found in the slides of the standard evaluation of the SNs of the other five cases. The remaining paraffin-embedded material of these nodes was completely step sectioned at intervals of 50 μm, stained by H&E and immunohistochemistry and revealed micrometastases in one case, whereas the other four nodes remained negative. In one of these latter patients with a tumour cell negative SN, however, a non-SN removed during the SN procedure showed to harbour breast tumour cells verified by standard histological analysis. Therefore, the possibility that the histologically negative SN also harbours metastatic cells might be high, as the positive discriminant score of the marker panel obtained by real-time PCR indicates.
Figure 3.
Discriminant score of expression of the four marker genes CK19, p1B, EGP2 and SBEM in ALN of patients with and without breast cancer, and in histologically negative and positive SN of breast cancer patients. The median expression levels for the markergene panel within a group are indicated by a horizontal line. Seven of the 70 histological negative SN show a positive discriminant score, indicated by a surrounding black triangle. The discriminant score separating the tumour cell positive and negative lymph nodes is indicated by a dashed line.
DISCUSSION
In this study, we have established a highly sensitive and quantitative multimarker real-time PCR approach to detect disseminated breast cancer cells in ALNs and SNs. Based on experiences it is unlikely that false positive results can be avoided if only a single marker gene is used in a RT–PCR based test system (Lambrechts et al, 1999). Using more than a single marker gene, as applied in our experiments, is a potential way to overcome the problem of illegitimate expression (Chelly et al, 1989), assuming that there is a little chance of encountering significant illegitimate mRNA of more than one gene at a time.
The marker genes tested, CK19, p1B, EGP-2, SBEM, PS2 and mammaglobin, share the characteristics that they are all expressed at high levels in ALNs harbouring metastatic breast tumour cells, but only at very low levels in lymph node tissue itself (Table 2). However, EGFR, a mRNA marker gene described for circulating tumour cell detection in peripheral blood of breast cancer patients (De Luca et al, 2000), appeared not to be applicable as a marker gene in lymph nodes due to its high background expression in control lymph nodes. Applying a discriminant score (QDA) showed that the marker set including CK19, p1B, EGP2 and SBEM gave the most significant separation between tumour cell negative and positive ALNs with zero misclassified control lymph nodes (P<0.0001).
The expression level for each of the marker genes was calculated relative to the standard curve and corrected for the input of cDNA based on the GAPDH control. The standard curves were generated from a mix of five breast cancer cell lines to assure that every marker gene tested will be expressed at high levels, therewith creating reproducible and reliable experiments (Figure 2A). A single breast cancer cell line as standard shows variation in gene expression levels of individual marker genes, a noticeable feature when determining the sensitivity of our marker panel used. The expression levels of SBEM and p1B in MCF7 cells are distinctly lower than those of CK19 and EGP2. This might be the reason for the nonlinear kinetics of the PCR reaction in the sensitivity experiment (Figure 2B), compared to the linear kinetics in the real-time PCR assays (Figure 2A), and the establishment of the amplification plateau below 30 cycles observed for SBEM and p1B.
When applying the set of marker genes to detect (micro) metastases in SNs, seven of the 70 histologically breast tumour cell free SNs showed a positive discriminant score, predicting the presence of metastatic disease. Four of these seven SNs could be histologically confirmed by more intensive review of slides or further sectioning of the paraffin-embedded material, what shows that standard evaluation is false-negative in 4%, whereas in three SNs no tumour cells were found by H&E staining and immunohistochemical analysis.
Using a real-time PCR approach we achieve an upstaging of SNs containing breast cancer metastases of 10% compared to the standard histological analysis. Our findings are in contrast to the recent results published by Schroder et al (2003) who found that in SNs immunostaining appears to be more sensitive/specific than quantitative PCR for breast tumour cell detection. The apparent discrepancy between our results and that of Schroder et al are likely due to the increased sensitivity achieved by the use of a multimarker panel in combination with the QDA score.
The follow-up times of the seven patients, whose histologically negative SNs showed a discriminant score predicting the presence of tumour cells using the real-time PCR approach, are too short to give an indication whether the upstaging of SNs reached by quantitative PCR has a prognostic value. Furthermore, it remains unknown whether micrometastatic disease in pathology-negative SNs have clinical significance. Ongoing clinical trials that will address this important issue (Grube and Giuliano, 2001; Ross, 2001). However, recently it was suggested that approximately 18% of the SNs harbouring micrometastases might be associated with further nodal non-SN metastases (Cserni et al, 2003). Our results provide information that could lead to a better management of breast cancer patients by reducing the rate of false-negative SNs using a quantitative real-time PCR approach with multiple mRNA markers instead of standard histological analysis for the detection of metastases in SNs.
Acknowledgments
We thank G Hart for statistical support and D de Jong for helping to select adequate control material. This study was supported by a grant to P Verduijn from the EORTC breast group.
References
- Bosma AJ, Weigelt B, Lambrechts AC, Verhagen OJ, Pruntel R, Hart AAM, Rodenhuis S, van't Veer LJ (2002) Detection of circulating breast tumor cells by differential expression of marker genes. Clin Cancer Res 8: 1871–1877 [PubMed] [Google Scholar]
- Bourez RL, Rutgers EJ (2001) The European Organization for Research and Treatment of Cancer (EORTC) Breast Cancer Group: quality control of surgical trials. Surg Oncol Clin N Am 10: 807–819 ix [PubMed] [Google Scholar]
- Bourez RL, Rutgers EJ, Van De Velde CJ (2002) Will we need lymph node dissection at all in the future? Clin Breast Cancer 3: 315–322 [DOI] [PubMed] [Google Scholar]
- Bieche I, Laurendeau I, Tozlu S, Olivi M, Vidaud D, Lidereau R, Vidaud M (1999) Quantitation of MYC gene expression in sporadic breast tumors with a real-time reverse transcription-PCR assay. Cancer Res 59: 2759–2765 [PubMed] [Google Scholar]
- Carter CL, Allen C, Henson DE (1989) Relation of tumor size, lymph node status, and survival in 24,740 breast cancer cases. Cancer 63: 181–187 [DOI] [PubMed] [Google Scholar]
- Chelly J, Concordet JP, Kaplan JC, Kahn A (1989) Illegitimate transcription: transcription of any gene in any cell type. Proc Natl Acad Sci USA 86: 2617–2621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cserni G, Amendoeira I, Apostolikas N, Bellocq JP, Bianchi S, Bussolati G, Boecker W, Borisch B, Connolly CE, Decker T, Dervan P, Drijkoningen M, Ellis IO, Elston CW, Eusebi V, Faverly D, Heikkila P, Holland R, Kerner H, Kulka J, Jacquemier J, Lacerda M, Martinez-Penuela J, De Miguel C, Peterse JL, Rank F, Regitnig P, Reiner A, Sapino A, Sigal-Zafrani B, Tanous AM, Thorstenson S, Zozaya E, Wells CA, European Working Group for Breast Screening Pathology (2003) Pathological work-up of sentinel lymph nodes in breast cancer. Review of current data to be considered for the formulation of guidelines. Eur J Cancer 39: 1654–1667 [DOI] [PubMed] [Google Scholar]
- De Luca A, Pignata S, Casamassimi A, D'Antonio A, Gridelli C, Rossi A, Cremona F, Parisi V, De Matteis A, Normanno N (2000) Detection of circulating tumor cells in carcinoma patients by a novel epidermal growth factor receptor reverse transcription-PCR assay. Clin Cancer Res 6: 1439–1444 [PubMed] [Google Scholar]
- Doting MH, Jansen L, Nieweg OE, Piers DA, Tiebosch AT, Koops HS, Rutgers EJ, Kroon BB, Peterse JL, Olmos RA, de Vries J (2000) Lymphatic mapping with intralesional tracer administration in breast carcinoma patients. Cancer 88: 2546–2552 [PubMed] [Google Scholar]
- Fisher B, Bauer M, Wickerham DL, Redmond CK, Fisher ER (1983) Relation of number of axillary lymph nodes to the prognosis of patients with primary breast cancer. An NSABP update. Cancer 52: 1551–1557 [DOI] [PubMed] [Google Scholar]
- Foster Jr RS (1996) The biologic and clinical significance of lymphatic metastases in breast cancer. Surg Oncol Clin N Am 5: 79–104 [PubMed] [Google Scholar]
- Giuliano AE, Dale PS, Turner RR, Morton DL, Evans SW, Krasne DL (1995) Improved axillary staging of breast cancer with sentinel lymphadenectomy. Ann Surg 222: 394–401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giuliano AE, Jones RC, Brennan M, Statman R (1997) Sentinel lymphadenectomy in breast cancer. J Clin Oncol 15: 2345–2350 [DOI] [PubMed] [Google Scholar]
- Giuliano AE, Kirgan DM, Guenther JM, Morton DL (1994) Lymphatic mapping and sentinel lymphadenectomy for breast cancer. Ann Surg 220: 391–401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grube BJ, Giuliano AE (2001) Observation of the breast cancer patient with a tumor-positive sentinel node: implications of the ACOSOG Z0011 trial. Semin Surg Oncol 20: 230–237 [DOI] [PubMed] [Google Scholar]
- Hellman S (1994) Karnofsky Memorial Lecture. Natural history of small breast cancers. J Clin Oncol 12: 2229–2234 [DOI] [PubMed] [Google Scholar]
- Joensuu H, Pylkkanen L, Toikkanen S (1998) Long-term survival in node-positive breast cancer treated by locoregional therapy alone. Br J Cancer 78: 795–799 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamby C, Rasmussen BB, Kristensen B (1991) Prognostic indicators of metastatic bone disease in human breast cancer. Cancer 68: 2045–2050 [DOI] [PubMed] [Google Scholar]
- Kufer P, Zippelius A, Lutterbuse R, Mecklenburg I, Enzmann T, Montag A, Weckermann D, Passlick B, Prang N, Reichardt P, Dugas M, Kollermann MW, Pantel K, Riethmuller G (2002) Heterogeneous expression of MAGE-A genes in occult disseminated tumor cells: a novel multimarker reverse transcription-polymerase chain reaction for diagnosis of micrometastatic disease. Cancer Res 62: 251–261 [PubMed] [Google Scholar]
- Lambrechts AC, Bosma AJ, Klaver SG, Top B, Perebolte L, Van't Veer LJ, Rodenhuis S (1999) Comparison of immunocytochemistry, reverse transcriptase polymerase chain reaction, and nucleic acid sequence-based amplification for the detection of circulating breast cancer cells. Breast Cancer Res Treatm 56: 219–231 [DOI] [PubMed] [Google Scholar]
- McGuire WL (1987) Prognostic factors for recurrence and survival in human breast cancer. Breast Cancer Res Treat 10: 5–9 [DOI] [PubMed] [Google Scholar]
- Miksicek RJ, Myal Y, Watson PH, Walker C, Murphy LC, Leygue E (2002) Identification of a novel breast- and salivary gland-specific, mucin-like gene strongly expressed in normal and tumor human mammary epithelium. Cancer Res 62: 2736–2740 [PubMed] [Google Scholar]
- Mitas M, Mikhitarian K, Walters C, Baron PL, Elliott BM, Brothers TE, Robison JG, Metcalf JS, Palesch YY, Zhang Z, Gillanders WE, Cole DJ (2001) Quantitative real-time RT-PCR detection of breast cancer micrometastasis using a multigene marker panel. Int J Cancer 93: 162–171 [DOI] [PubMed] [Google Scholar]
- Quiet CA, Ferguson DJ, Weichselbaum RR, Hellman S (1995) Natural history of node-negative breast cancer: a study of 826 patients with long-term follow up. J Clin Oncol 13: 1144–1151 [DOI] [PubMed] [Google Scholar]
- Rosen PR, Groshen S, Saigo PE, Kinne DW, Hellman S (1989) A long-term follow-up study of survival in stage I (T1N0M0) and stage II (T1N1M0) breast carcinoma. J Clin Oncol 7: 355–366 [DOI] [PubMed] [Google Scholar]
- Ross MI (2001) Sentinel node dissection in early-stage breast cancer: ongoing prospective randomized trials in the USA. Ann Surg Oncol 8: 77S–81S [PubMed] [Google Scholar]
- Rutgers EJ, Jansen L, Nieweg OE, de Vries J, Schraffordt Koops H, Kroon BB (1998) Technique of sentinel node biopsy in breast cancer. Eur J Surg Oncol 24: 316–319 [DOI] [PubMed] [Google Scholar]
- Saimura M, Fukutomi T, Tsuda H, Sato H, Miyamoto K, Akashi-Tanaka S, Nanasawa T (1999) Prognosis of a series of 763 consecutive node-negative invasive breast cancer patients without adjuvant therapy: analysis of clinicopathological prognostic factor. J Surg Oncol 71: 101–105 [DOI] [PubMed] [Google Scholar]
- Schroder CP, Ruiters MH, de Jong S, Tiebosch AT, Wesseling J, Veenstra R, de Vries J, Hoekstra HJ, de Leij LF, de Vries EG (2003) Detection of micrometastatic breast cancer by means of real time quantitative RT-PCR and immunostaining in perioperative blood samples and sentinel nodes. Int J Cancer 106: 611–618 [DOI] [PubMed] [Google Scholar]
- Tjan-Heijnen VC, Buit P, de Widt-Evert LM, Ruers TJ, Beex LV (2001) Micro-metastases in axillary lymph nodes: an increasing classification and treatment dilemma in breast cancer due to the introduction of the sentinel lymph node procedure. Breast Cancer Res Treat 70: 81–88 [DOI] [PubMed] [Google Scholar]
- van de Vijver MJ, He YD, van't Veer LJ, Dai H, Hart AA, Voskuil DW, Schreiber GJ, Peterse JL, Roberts C, Marton MJ, Parrish M, Atsma D, Witteveen A, Glas A, Delahaye L, van der Velde T, Bartelink H, Rodenhuis S, Rutgers ET, Friend SH, Bernards R (2002) A gene-expression signature as a predictor of survival in breast cancer. N Engl J Med 347: 1999–2009 [DOI] [PubMed] [Google Scholar]
- van't Veer LJ, Dai H, van de Vijver MJ, He YD, Hart AA, Mao M, Peterse HL, van der Kooy K, Marton MJ, Witteveen AT, Schreiber GJ, Kerkhoven RM, Roberts C, Linsley PS, Bernards R, Friend SH (2002) Gene expression profiling predicts clinical outcome of breast cancer. Nature 415: 530–536 [DOI] [PubMed] [Google Scholar]
- Zehentner BK, Dillon DC, Jiang Y, Xu J, Bennington A, Molesh DA, Zhang X, Reed SG, Persing D, Houghton RL (2002) Application of a multigene reverse transcription-PCR assay for detection of mammaglobin and complementary transcribed genes in breast cancer lymph nodes. Clin Chem 48: 1225–1231 [PMC free article] [PubMed] [Google Scholar]