Abstract
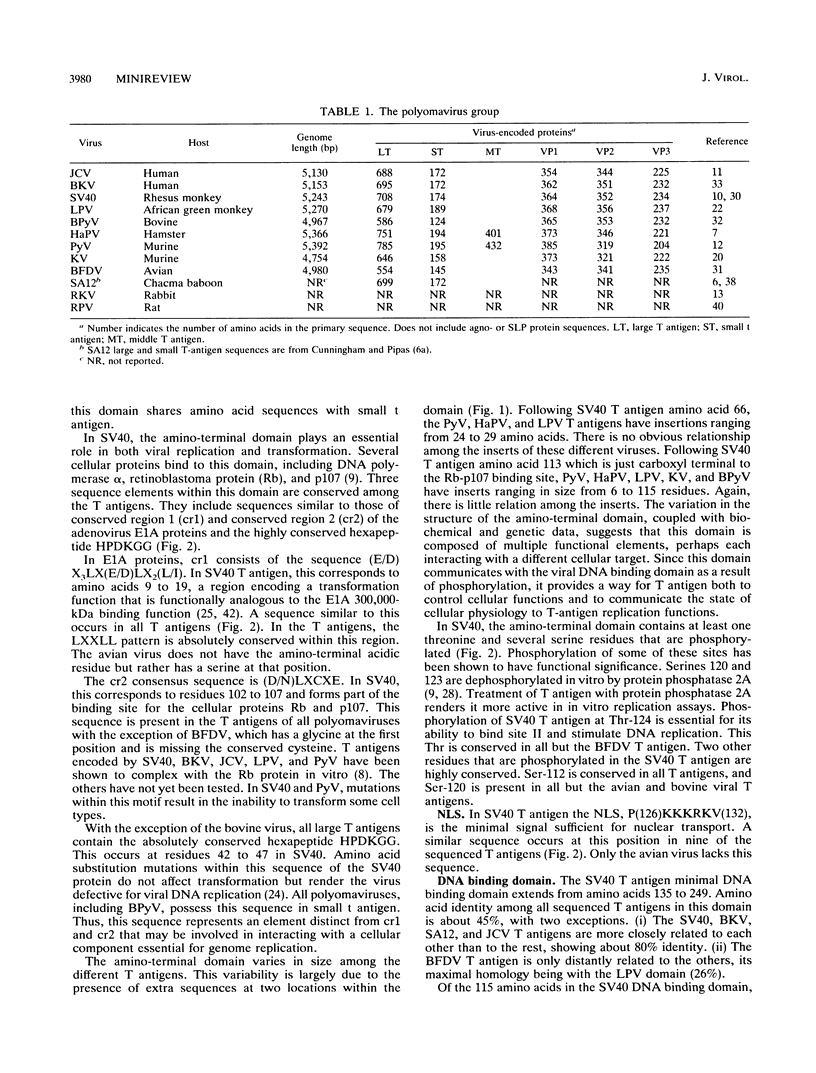
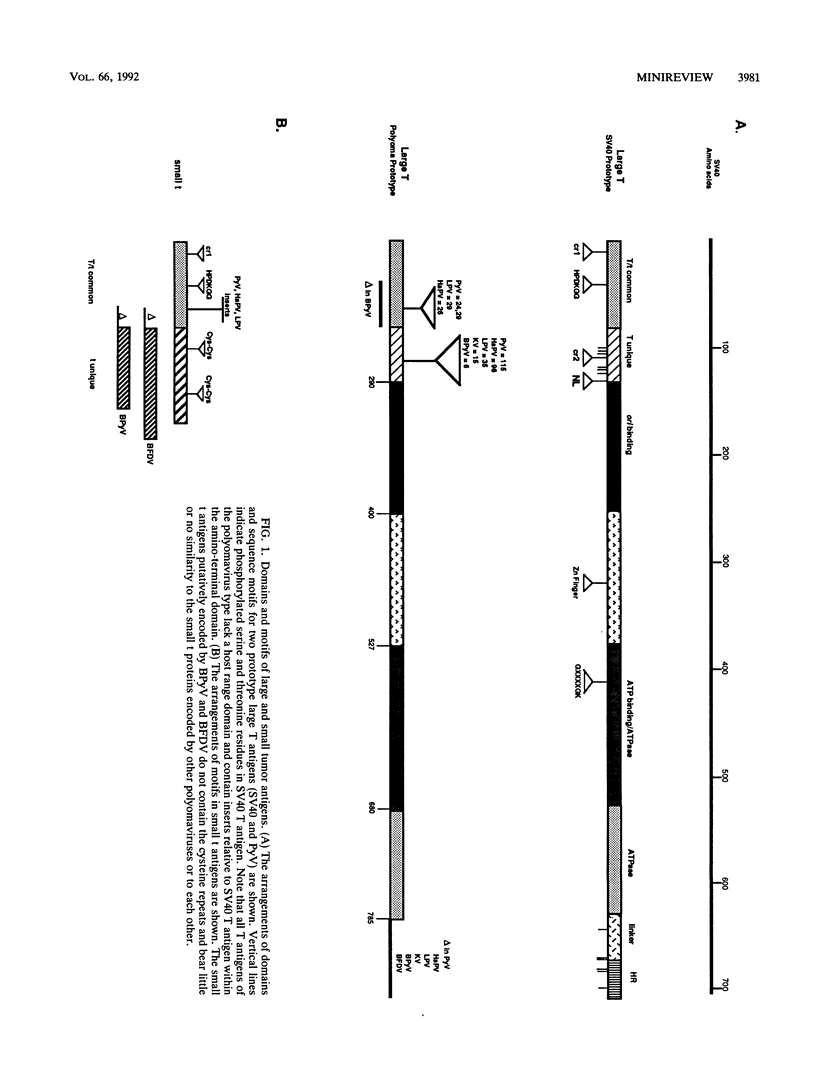
Although 12 different members of the polyomavirus group have now been identified, only SV40 and PyV have been studied extensively. Whereas each member of the group shows a restricted host range, viruses infecting species from birds to humans have been reported. Although little is known concerning the biology of natural infections in the wild, it is apparent that these viruses exhibit various cell-type tropisms. Some viruses, such as LPV (B lymphocytes) or KV (pulmonary endothelium), are tightly restricted to specific cell types, while others, such as PyV, infect a variety of tissues in the animal. Despite these differences, all polyomaviruses share a common strategy of productive infection, expressing T antigens which act both on cellular targets, preparing cellular metabolism for supporting optimal viral replication, and then on targets within the viral genome, to regulate viral DNA replication, transcription, and assembly. Presumably, this common replication strategy restricts the degree to which the sequences of these viruses can diverge. Thus, sequence motifs conserved among these different viruses may indicate key structural elements essential for biochemical function. In this article I have compared the sequences of all polyomavirus-encoded large and small T antigens sequenced to date. This has led to the following conclusions and speculations. (i) Comparison of the domain organization of different large T antigens reveals that these proteins fall into two structural classes. Members of the SV40 class, which include SV40, JCV, BKV, and SA12, possess a carboxyl-terminal domain, which in SV40 has been shown to be dispensable for viral DNA replication but essential for virion assembly. The PyV class lacks the carboxyl-terminal domain and carries additional amino acids within the amino-terminal domain. When total amino acid identity is examined, members of the SV40 class show the highest degree of conservation (65 to 85%), while sequence identity among the remaining viruses varies from 18 to 55%. (ii) The DNA binding domains of most large T antigens are closely related, with amino acid identities ranging from 35 to 86%. Several residues within this domain are invariant among all T antigens. All of these viruses have multiple copies of the consensus T-antigen-binding pentanucleotide (GAGGC) in their ori region, suggesting that all T antigens recognize this sequence. The single exception is the large T antigen encoded by the avian virus BFDV. The putative DNA binding domain of this protein shows little or no sequence relation to that of other T antigens. Furthermore, the GAGGC motif is not found in the ori region of this virus.(ABSTRACT TRUNCATED AT 400 WORDS)
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bradley M. K., Smith T. F., Lathrop R. H., Livingston D. M., Webster T. A. Consensus topography in the ATP binding site of the simian virus 40 and polyomavirus large tumor antigens. Proc Natl Acad Sci U S A. 1987 Jun;84(12):4026–4030. doi: 10.1073/pnas.84.12.4026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark R., Peden K., Pipas J. M., Nathans D., Tjian R. Biochemical activities of T-antigen proteins encoded by simian virus 40 A gene deletion mutants. Mol Cell Biol. 1983 Feb;3(2):220–228. doi: 10.1128/mcb.3.2.220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cole C. N., Crawford L. V., Berg P. Simian virus 40 mutants with deletions at the 3' end of the early region are defective in adenovirus helper function. J Virol. 1979 Jun;30(3):683–691. doi: 10.1128/jvi.30.3.683-691.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cole C. N., Tornow J., Clark R., Tjian R. Properties of the simian virus 40 (SV40) large T antigens encoded by SV40 mutants with deletions in gene A. J Virol. 1986 Feb;57(2):539–546. doi: 10.1128/jvi.57.2.539-546.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunningham T. P., Pipas J. M. Simian agent 12 is a BK virus-like papovavirus which replicates in monkey cells. J Virol. 1985 May;54(2):483–492. doi: 10.1128/jvi.54.2.483-492.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delmas V., Bastien C., Scherneck S., Feunteun J. A new member of the polyomavirus family: the hamster papovavirus. Complete nucleotide sequence and transformation properties. EMBO J. 1985 May;4(5):1279–1286. doi: 10.1002/j.1460-2075.1985.tb03773.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dyson N., Bernards R., Friend S. H., Gooding L. R., Hassell J. A., Major E. O., Pipas J. M., Vandyke T., Harlow E. Large T antigens of many polyomaviruses are able to form complexes with the retinoblastoma protein. J Virol. 1990 Mar;64(3):1353–1356. doi: 10.1128/jvi.64.3.1353-1356.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fanning E. Simian virus 40 large T antigen: the puzzle, the pieces, and the emerging picture. J Virol. 1992 Mar;66(3):1289–1293. doi: 10.1128/jvi.66.3.1289-1293.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiers W., Contreras R., Haegemann G., Rogiers R., Van de Voorde A., Van Heuverswyn H., Van Herreweghe J., Volckaert G., Ysebaert M. Complete nucleotide sequence of SV40 DNA. Nature. 1978 May 11;273(5658):113–120. doi: 10.1038/273113a0. [DOI] [PubMed] [Google Scholar]
- Frisque R. J., Bream G. L., Cannella M. T. Human polyomavirus JC virus genome. J Virol. 1984 Aug;51(2):458–469. doi: 10.1128/jvi.51.2.458-469.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HARTLEY J. W., ROWE W. P. NEW PAPOVAVIRUS CONTAMINATING SHOPE PAPILLOMATA. Science. 1964 Jan 17;143(3603):258–260. doi: 10.1126/science.143.3603.258. [DOI] [PubMed] [Google Scholar]
- Kelly T. J. SV40 DNA replication. J Biol Chem. 1988 Dec 5;263(34):17889–17892. [PubMed] [Google Scholar]
- Khalili K., Brady J., Pipas J. M., Spence S. L., Sadofsky M., Khoury G. Carboxyl-terminal mutants of the large tumor antigen of simian virus 40: a role for the early protein late in the lytic cycle. Proc Natl Acad Sci U S A. 1988 Jan;85(2):354–358. doi: 10.1073/pnas.85.2.354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loeber G., Parsons R., Tegtmeyer P. The zinc finger region of simian virus 40 large T antigen. J Virol. 1989 Jan;63(1):94–100. doi: 10.1128/jvi.63.1.94-100.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loeken M., Bikel I., Livingston D. M., Brady J. trans-activation of RNA polymerase II and III promoters by SV40 small t antigen. Cell. 1988 Dec 23;55(6):1171–1177. doi: 10.1016/0092-8674(88)90261-9. [DOI] [PubMed] [Google Scholar]
- Manos M. M., Gluzman Y. Genetic and biochemical analysis of transformation-competent, replication-defective simian virus 40 large T antigen mutants. J Virol. 1985 Jan;53(1):120–127. doi: 10.1128/jvi.53.1.120-127.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Margolskee R. F., Nathans D. Simian virus 40 mutant T antigens with relaxed specificity for the nucleotide sequence at the viral DNA origin of replication. J Virol. 1984 Feb;49(2):386–393. doi: 10.1128/jvi.49.2.386-393.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayer M., Dörries K. Nucleotide sequence and genome organization of the murine polyomavirus, Kilham strain. Virology. 1991 Apr;181(2):469–480. doi: 10.1016/0042-6822(91)90879-g. [DOI] [PubMed] [Google Scholar]
- Pallas D. C., Shahrik L. K., Martin B. L., Jaspers S., Miller T. B., Brautigan D. L., Roberts T. M. Polyoma small and middle T antigens and SV40 small t antigen form stable complexes with protein phosphatase 2A. Cell. 1990 Jan 12;60(1):167–176. doi: 10.1016/0092-8674(90)90726-u. [DOI] [PubMed] [Google Scholar]
- Pawlita M., Clad A., zur Hausen H. Complete DNA sequence of lymphotropic papovavirus: prototype of a new species of the polyomavirus genus. Virology. 1985 May;143(1):196–211. doi: 10.1016/0042-6822(85)90108-4. [DOI] [PubMed] [Google Scholar]
- Peden K. W., Pipas J. M. Simian virus 40 mutants with amino-acid substitutions near the amino terminus of large T antigen. Virus Genes. 1992 Apr;6(2):107–118. doi: 10.1007/BF01703060. [DOI] [PubMed] [Google Scholar]
- Peden K. W., Spence S. L., Tack L. C., Cartwright C. A., Srinivasan A., Pipas J. M. A DNA replication-positive mutant of simian virus 40 that is defective for transformation and the production of infectious virions. J Virol. 1990 Jun;64(6):2912–2921. doi: 10.1128/jvi.64.6.2912-2921.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peden K. W., Srinivasan A., Farber J. M., Pipas J. M. Mutants with changes within or near a hydrophobic region of simian virus 40 large tumor antigen are defective for binding cellular protein p53. Virology. 1989 Jan;168(1):13–21. doi: 10.1016/0042-6822(89)90398-x. [DOI] [PubMed] [Google Scholar]
- Pipas J. M. Mutations near the carboxyl terminus of the simian virus 40 large tumor antigen alter viral host range. J Virol. 1985 May;54(2):569–575. doi: 10.1128/jvi.54.2.569-575.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prives C. The replication functions of SV40 T antigen are regulated by phosphorylation. Cell. 1990 Jun 1;61(5):735–738. doi: 10.1016/0092-8674(90)90179-i. [DOI] [PubMed] [Google Scholar]
- Rajan P., Dhamankar V., Rundell K., Thimmapaya B. Simian virus 40 small-t does not transactivate RNA polymerase II promoters in virus infections. J Virol. 1991 Dec;65(12):6553–6561. doi: 10.1128/jvi.65.12.6553-6561.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reddy V. B., Thimmappaya B., Dhar R., Subramanian K. N., Zain B. S., Pan J., Ghosh P. K., Celma M. L., Weissman S. M. The genome of simian virus 40. Science. 1978 May 5;200(4341):494–502. doi: 10.1126/science.205947. [DOI] [PubMed] [Google Scholar]
- Rott O., Kröger M., Müller H., Hobom G. The genome of budgerigar fledgling disease virus, an avian polyomavirus. Virology. 1988 Jul;165(1):74–86. doi: 10.1016/0042-6822(88)90660-5. [DOI] [PubMed] [Google Scholar]
- Schuurman R., Sol C., van der Noordaa J. The complete nucleotide sequence of bovine polyomavirus. J Gen Virol. 1990 Aug;71(Pt 8):1723–1735. doi: 10.1099/0022-1317-71-8-1723. [DOI] [PubMed] [Google Scholar]
- Seif I., Khoury G., Dhar R. The genome of human papovavirus BKV. Cell. 1979 Dec;18(4):963–977. doi: 10.1016/0092-8674(79)90209-5. [DOI] [PubMed] [Google Scholar]
- Srinivasan A., Peden K. W., Pipas J. M. The large tumor antigen of simian virus 40 encodes at least two distinct transforming functions. J Virol. 1989 Dec;63(12):5459–5463. doi: 10.1128/jvi.63.12.5459-5463.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stacy T., Chamberlain M., Cole C. N. Simian virus 40 host range/helper function mutations cause multiple defects in viral late gene expression. J Virol. 1989 Dec;63(12):5208–5215. doi: 10.1128/jvi.63.12.5208-5215.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stillman B. Initiation of eukaryotic DNA replication in vitro. Annu Rev Cell Biol. 1989;5:197–245. doi: 10.1146/annurev.cb.05.110189.001213. [DOI] [PubMed] [Google Scholar]
- Tack L. C., Cartwright C. A., Wright J. H., Eckhart W., Peden K. W., Srinivasan A., Pipas J. M. Properties of a simian virus 40 mutant T antigen substituted in the hydrophobic region: defective ATPase and oligomerization activities and altered phosphorylation accompany an inability to complex with cellular p53. J Virol. 1989 Aug;63(8):3362–3367. doi: 10.1128/jvi.63.8.3362-3367.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valis J. D., Newell N., Reissig M., Malherbe H., Kaschula V. R., Shah K. V. Characterization of SA12 as a simian virus 40-related papovavirus of chacma baboons. Infect Immun. 1977 Oct;18(1):247–252. doi: 10.1128/iai.18.1.247-252.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker J. E., Saraste M., Runswick M. J., Gay N. J. Distantly related sequences in the alpha- and beta-subunits of ATP synthase, myosin, kinases and other ATP-requiring enzymes and a common nucleotide binding fold. EMBO J. 1982;1(8):945–951. doi: 10.1002/j.1460-2075.1982.tb01276.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward J. M., Lock A., Collins M. J., Jr, Gonda M. A., Reynolds C. W. Papovaviral sialoadenitis in athymic nude rats. Lab Anim. 1984 Jan;18(1):84–89. doi: 10.1258/002367784780864884. [DOI] [PubMed] [Google Scholar]
- Wun-Kim K., Simmons D. T. Mapping of helicase and helicase substrate-binding domains on simian virus 40 large T antigen. J Virol. 1990 May;64(5):2014–2020. doi: 10.1128/jvi.64.5.2014-2020.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yaciuk P., Carter M. C., Pipas J. M., Moran E. Simian virus 40 large-T antigen expresses a biological activity complementary to the p300-associated transforming function of the adenovirus E1A gene products. Mol Cell Biol. 1991 Apr;11(4):2116–2124. doi: 10.1128/mcb.11.4.2116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang S. I., Lickteig R. L., Estes R., Rundell K., Walter G., Mumby M. C. Control of protein phosphatase 2A by simian virus 40 small-t antigen. Mol Cell Biol. 1991 Apr;11(4):1988–1995. doi: 10.1128/mcb.11.4.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]


