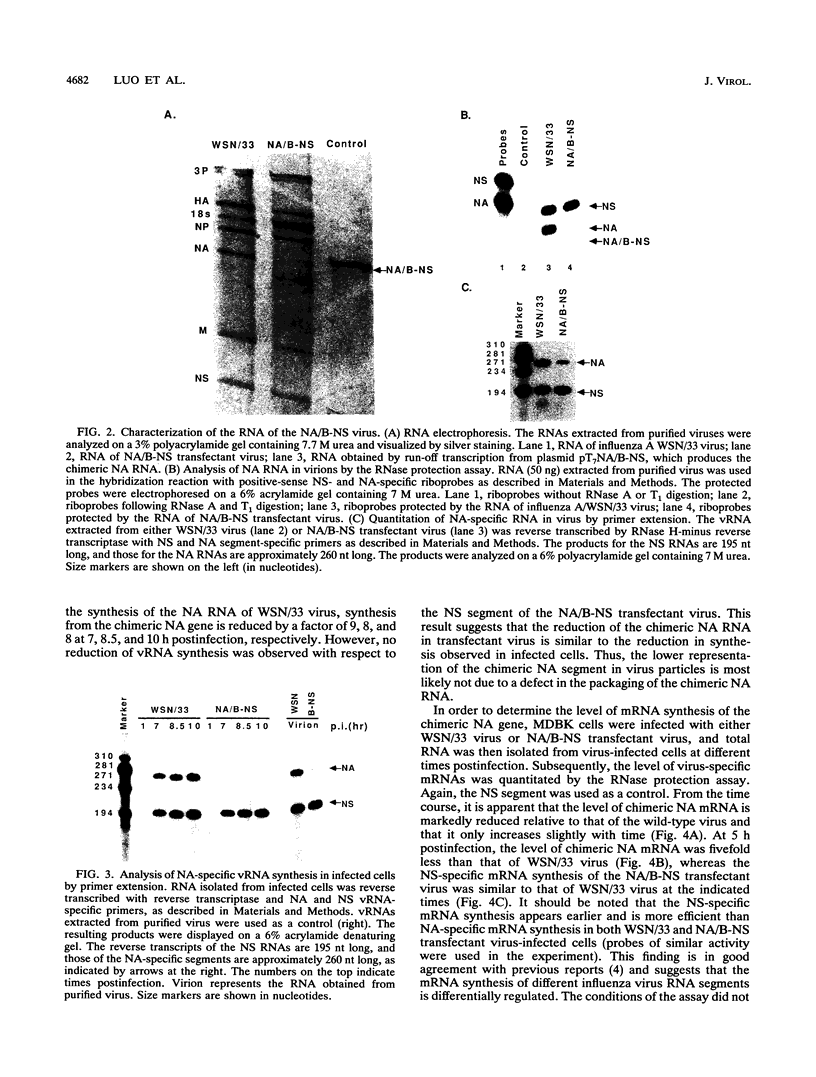
Abstract
The ribonucleoprotein transfection system for influenza virus allowed us to construct an influenza A virus containing a chimeric neuraminidase (NA) gene in which the noncoding sequence is derived from the NS gene of influenza B virus (T. Muster, E. K. Subbarao, M. Enami, B. P. Murphy, and P. Palese, Proc. Natl. Acad. Sci. USA 88:5177-5181, 1991). This transfectant virus is attenuated in mice and grows to lower titers in tissue culture than wild-type virus. Since such a virus has characteristics desirable for a live attenuated vaccine strain, attempts were made to characterize this virus at the molecular level. Our analysis suggests that the attenuation of the virus is due to changes in the cis signal sequences, which resulted in a reduction of transcription and replication of the chimeric NA gene. The major finding concerns a sixfold reduction in NA-specific viral RNA in the virion, causing a reduction in the ratio of infectious particles to physical particles compared with the ratio in wild-type virus. Although the NA-specific mRNA level is also reduced in transfectant virus-infected cells, it does not appear to contribute to the attenuation characteristics of the virus. The levels of the other RNAs and their expression appear to be unchanged for the transfectant virus. It is suggested that downregulation of the synthesis of one viral RNA segment leads to the generation of defective viruses during each replication cycle. We believe that this represents a general principle for attenuation which may be applied to other segmented viruses containing either single-stranded or double-stranded RNA.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Enami M., Fukuda R., Ishihama A. Transcription and replication of eight RNA segments of influenza virus. Virology. 1985 Apr 15;142(1):68–77. doi: 10.1016/0042-6822(85)90423-4. [DOI] [PubMed] [Google Scholar]
- Enami M., Luytjes W., Krystal M., Palese P. Introduction of site-specific mutations into the genome of influenza virus. Proc Natl Acad Sci U S A. 1990 May;87(10):3802–3805. doi: 10.1073/pnas.87.10.3802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enami M., Sharma G., Benham C., Palese P. An influenza virus containing nine different RNA segments. Virology. 1991 Nov;185(1):291–298. doi: 10.1016/0042-6822(91)90776-8. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lamb R. A., Choppin P. W. The gene structure and replication of influenza virus. Annu Rev Biochem. 1983;52:467–506. doi: 10.1146/annurev.bi.52.070183.002343. [DOI] [PubMed] [Google Scholar]
- Luo G. X., Luytjes W., Enami M., Palese P. The polyadenylation signal of influenza virus RNA involves a stretch of uridines followed by the RNA duplex of the panhandle structure. J Virol. 1991 Jun;65(6):2861–2867. doi: 10.1128/jvi.65.6.2861-2867.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo G. X., Taylor J. Template switching by reverse transcriptase during DNA synthesis. J Virol. 1990 Sep;64(9):4321–4328. doi: 10.1128/jvi.64.9.4321-4328.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo G., Palese P. Genetic analysis of influenza virus. Curr Opin Genet Dev. 1992 Feb;2(1):77–81. doi: 10.1016/s0959-437x(05)80326-0. [DOI] [PubMed] [Google Scholar]
- Luytjes W., Krystal M., Enami M., Parvin J. D., Palese P. Amplification, expression, and packaging of foreign gene by influenza virus. Cell. 1989 Dec 22;59(6):1107–1113. doi: 10.1016/0092-8674(89)90766-6. [DOI] [PubMed] [Google Scholar]
- Muster T., Subbarao E. K., Enami M., Murphy B. R., Palese P. An influenza A virus containing influenza B virus 5' and 3' noncoding regions on the neuraminidase gene is attenuated in mice. Proc Natl Acad Sci U S A. 1991 Jun 15;88(12):5177–5181. doi: 10.1073/pnas.88.12.5177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parvin J. D., Palese P., Honda A., Ishihama A., Krystal M. Promoter analysis of influenza virus RNA polymerase. J Virol. 1989 Dec;63(12):5142–5152. doi: 10.1128/jvi.63.12.5142-5152.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roner M. R., Sutphin L. A., Joklik W. K. Reovirus RNA is infectious. Virology. 1990 Dec;179(2):845–852. doi: 10.1016/0042-6822(90)90153-i. [DOI] [PubMed] [Google Scholar]
- Scholtissek C., Rohde W., Harms E., Rott R., Orlich M., Boschek C. B. A possible partial heterozygote of an influenza A virus. Virology. 1978 Sep;89(2):506–516. doi: 10.1016/0042-6822(78)90192-7. [DOI] [PubMed] [Google Scholar]
- Seong B. L., Brownlee G. G. A new method for reconstituting influenza polymerase and RNA in vitro: a study of the promoter elements for cRNA and vRNA synthesis in vitro and viral rescue in vivo. Virology. 1992 Jan;186(1):247–260. doi: 10.1016/0042-6822(92)90079-5. [DOI] [PubMed] [Google Scholar]
- Shapiro G. I., Gurney T., Jr, Krug R. M. Influenza virus gene expression: control mechanisms at early and late times of infection and nuclear-cytoplasmic transport of virus-specific RNAs. J Virol. 1987 Mar;61(3):764–773. doi: 10.1128/jvi.61.3.764-773.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith G. L., Hay A. J. Replication of the influenza virus genome. Virology. 1982 Apr 15;118(1):96–108. doi: 10.1016/0042-6822(82)90323-3. [DOI] [PubMed] [Google Scholar]