Abstract
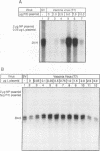
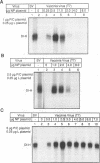
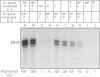
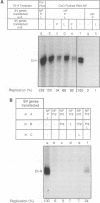
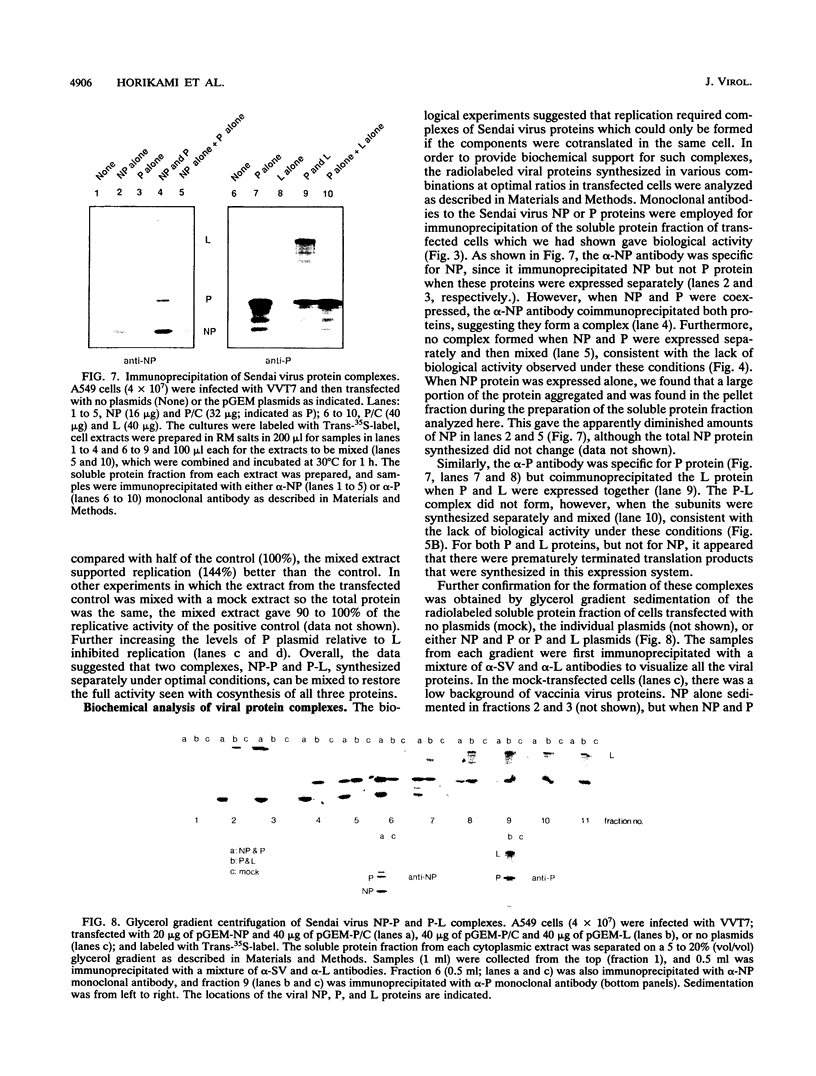
We present evidence that the formation of NP-P and P-L protein complexes is essential for replication of the genome of Sendai defective interfering (DI-H) virus in vitro, using extracts of cells expressing these viral proteins from plasmids. Optimal replication of DI-H nucleocapsid RNA required extracts of cells transfected with critical amounts and ratios of each of the plasmids and was three- to fivefold better than replication with a control extract prepared from a natural virus infection. Extracts in which NP and P proteins were coexpressed supported replication of the genome of purified DI-H virus which contained endogenous polymerase proteins, but extracts in which NP and P were expressed separately and then mixed were inactive. Similarly, the P and L proteins must be coexpressed for biological activity. The replication data thus suggest that two protein complexes, NP-P and P-L, are required for nucleocapsid RNA replication and that these complexes must form during or soon after synthesis of the proteins. Biochemical evidence in support of the formation of each complex includes coimmunoprecipitation of both proteins of each complex with an antibody specific for one component and cosedimentation of the subunits of each complex. We propose that the P-L complex serves as the RNA polymerase and NP-P is required for encapsidation of newly synthesized RNA.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baker S. C., Moyer S. A. Encapsidation of Sendai virus genome RNAs by purified NP protein during in vitro replication. J Virol. 1988 Mar;62(3):834–838. doi: 10.1128/jvi.62.3.834-838.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carlsen S. R., Peluso R. W., Moyer S. A. In vitro replication of Sendai virus wild-type and defective interfering particle genome RNAs. J Virol. 1985 May;54(2):493–500. doi: 10.1128/jvi.54.2.493-500.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curran J., Boeck R., Kolakofsky D. The Sendai virus P gene expresses both an essential protein and an inhibitor of RNA synthesis by shuffling modules via mRNA editing. EMBO J. 1991 Oct;10(10):3079–3085. doi: 10.1002/j.1460-2075.1991.tb07860.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuerst T. R., Niles E. G., Studier F. W., Moss B. Eukaryotic transient-expression system based on recombinant vaccinia virus that synthesizes bacteriophage T7 RNA polymerase. Proc Natl Acad Sci U S A. 1986 Nov;83(21):8122–8126. doi: 10.1073/pnas.83.21.8122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glazier K., Raghow R., Kingsbury D. W. Regulation of Sendai virus transcription: evidence for a single promoter in vivo. J Virol. 1977 Mar;21(3):863–871. doi: 10.1128/jvi.21.3.863-871.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gotoh H., Shioda T., Sakai Y., Mizumoto K., Shibuta H. Rescue of Sendai virus from viral ribonucleoprotein-transfected cells by infection with recombinant vaccinia viruses carrying Sendai virus L and P/C genes. Virology. 1989 Aug;171(2):434–443. doi: 10.1016/0042-6822(89)90612-0. [DOI] [PubMed] [Google Scholar]
- Hamaguchi M., Yoshida T., Nishikawa K., Naruse H., Nagai Y. Transcriptive complex of Newcastle disease virus. I. Both L and P proteins are required to constitute an active complex. Virology. 1983 Jul 15;128(1):105–117. doi: 10.1016/0042-6822(83)90322-7. [DOI] [PubMed] [Google Scholar]
- Hardy W. R., Strauss J. H. Processing the nonstructural polyproteins of Sindbis virus: study of the kinetics in vivo by using monospecific antibodies. J Virol. 1988 Mar;62(3):998–1007. doi: 10.1128/jvi.62.3.998-1007.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Homann H. E., Willenbrink W., Buchholz C. J., Neubert W. J. Sendai virus protein-protein interactions studied by a protein-blotting protein-overlay technique: mapping of domains on NP protein required for binding to P protein. J Virol. 1991 Mar;65(3):1304–1309. doi: 10.1128/jvi.65.3.1304-1309.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard M., Wertz G. Vesicular stomatitis virus RNA replication: a role for the NS protein. J Gen Virol. 1989 Oct;70(Pt 10):2683–2694. doi: 10.1099/0022-1317-70-10-2683. [DOI] [PubMed] [Google Scholar]
- Huber M., Cattaneo R., Spielhofer P., Orvell C., Norrby E., Messerli M., Perriard J. C., Billeter M. A. Measles virus phosphoprotein retains the nucleocapsid protein in the cytoplasm. Virology. 1991 Nov;185(1):299–308. doi: 10.1016/0042-6822(91)90777-9. [DOI] [PubMed] [Google Scholar]
- Jenison S. A., Firzlaff J. M., Langenberg A., Galloway D. A. Identification of immunoreactive antigens of human papillomavirus type 6b by using Escherichia coli-expressed fusion proteins. J Virol. 1988 Jun;62(6):2115–2123. doi: 10.1128/jvi.62.6.2115-2123.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- La Ferla F. M., Peluso R. W. The 1:1 N-NS protein complex of vesicular stomatitis virus is essential for efficient genome replication. J Virol. 1989 Sep;63(9):3852–3857. doi: 10.1128/jvi.63.9.3852-3857.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leppert M., Kort L., Kolakofsky D. Further characterization of Sendai virus DI-RNAs: a model for their generation. Cell. 1977 Oct;12(2):539–552. doi: 10.1016/0092-8674(77)90130-1. [DOI] [PubMed] [Google Scholar]
- Leppert M., Rittenhouse L., Perrault J., Summers D. F., Kolakofsky D. Plus and minus strand leader RNAs in negative strand virus-infected cells. Cell. 1979 Nov;18(3):735–747. doi: 10.1016/0092-8674(79)90127-2. [DOI] [PubMed] [Google Scholar]
- Meier E., Harmison G. G., Schubert M. Homotypic and heterotypic exclusion of vesicular stomatitis virus replication by high levels of recombinant polymerase protein L. J Virol. 1987 Oct;61(10):3133–3142. doi: 10.1128/jvi.61.10.3133-3142.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pattnaik A. K., Wertz G. W. Replication and amplification of defective interfering particle RNAs of vesicular stomatitis virus in cells expressing viral proteins from vectors containing cloned cDNAs. J Virol. 1990 Jun;64(6):2948–2957. doi: 10.1128/jvi.64.6.2948-2957.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peluso R. W., Moyer S. A. Viral proteins required for the in vitro replication of vesicular stomatitis virus defective interfering particle genome RNA. Virology. 1988 Feb;162(2):369–376. doi: 10.1016/0042-6822(88)90477-1. [DOI] [PubMed] [Google Scholar]
- Portner A., Murti K. G., Morgan E. M., Kingsbury D. W. Antibodies against Sendai virus L protein: distribution of the protein in nucleocapsids revealed by immunoelectron microscopy. Virology. 1988 Mar;163(1):236–239. doi: 10.1016/0042-6822(88)90257-7. [DOI] [PubMed] [Google Scholar]
- Ryan K. W., Kingsbury D. W. Carboxyl-terminal region of Sendai virus P protein is required for binding to viral nucleocapsids. Virology. 1988 Nov;167(1):106–112. doi: 10.1016/0042-6822(88)90059-1. [DOI] [PubMed] [Google Scholar]
- Vidal S., Kolakofsky D. Modified model for the switch from Sendai virus transcription to replication. J Virol. 1989 May;63(5):1951–1958. doi: 10.1128/jvi.63.5.1951-1958.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]