Abstract
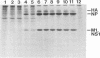
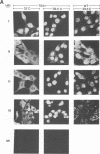
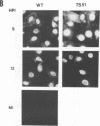
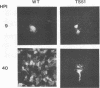
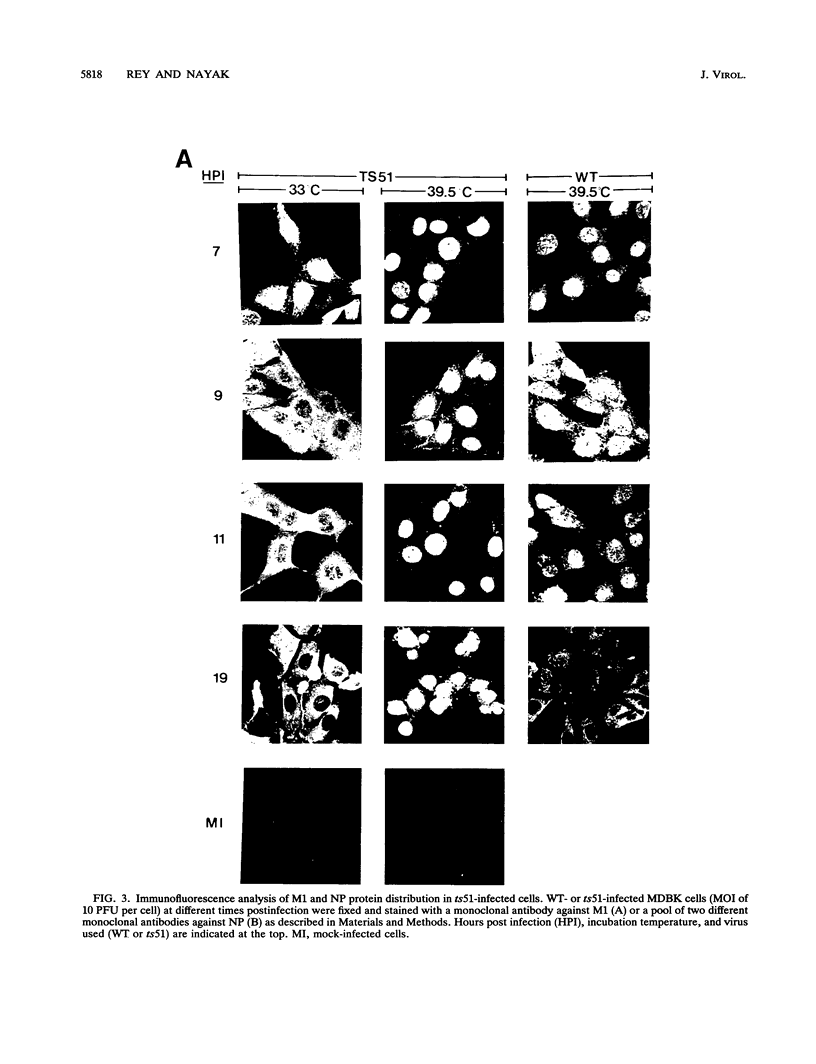
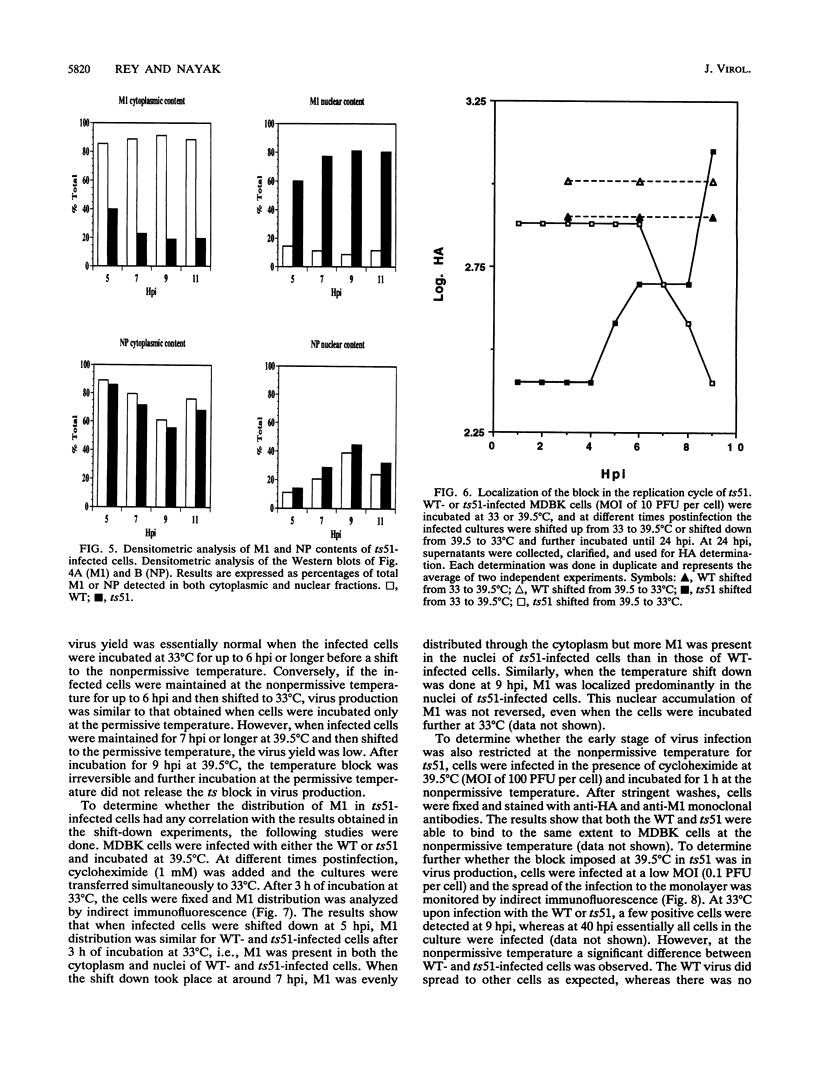
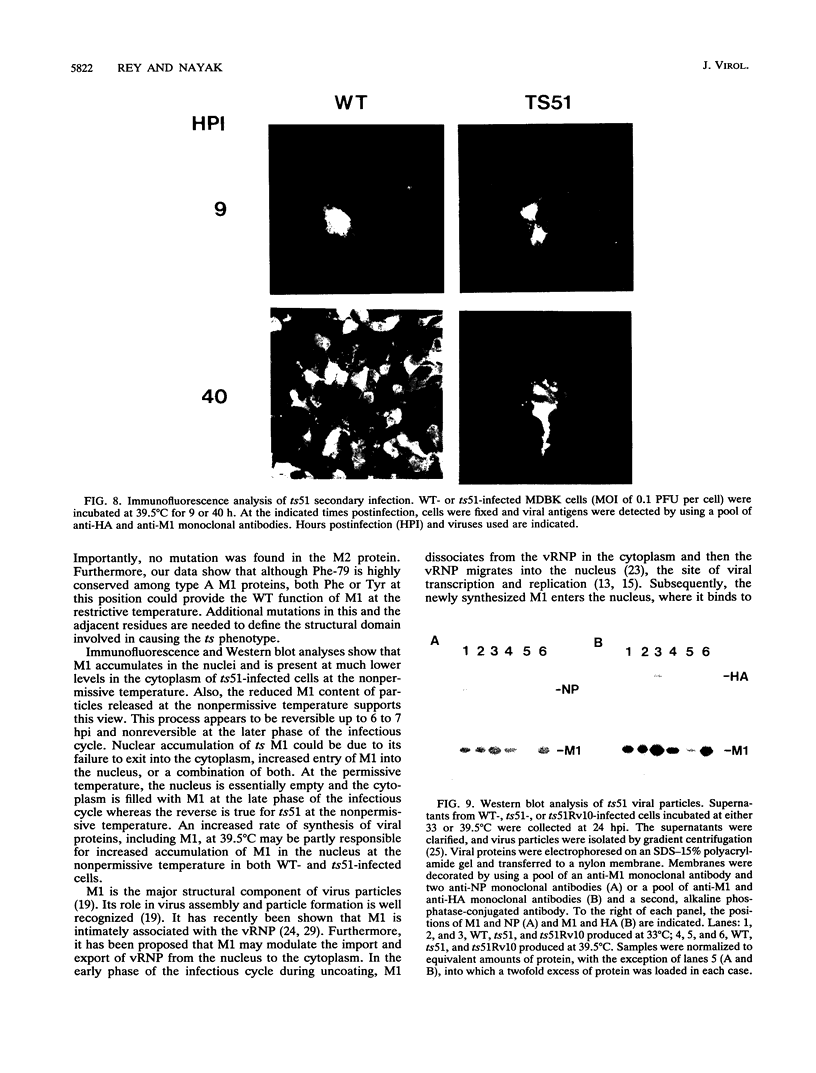
We investigated the properties of ts51, an influenza virus (A/WSN/33) temperature-sensitive RNA segment 7 mutant. Nucleotide sequence analysis revealed that ts51 possesses a single nucleotide mutation, T-261----C, in RNA segment 7, resulting in a single amino acid change. Phenylalanine (position 79) in the wild-type M1 protein was substituted by serine in ts51. This mutation was phenotypically characterized by dramatic nuclear accumulation of the M1 protein and interfered with some steps at the late stage of virus replication, possibly affecting the assembly and/or budding of viral particles. However, although M1 protein was retained within the nucleus, export of the newly synthesized viral ribonucleoprotein containing the minus-strand RNA into the cytoplasm was essentially the same at both permissive and nonpermissive temperatures. The roles of M1 in the export of viral ribonucleoproteins from the nucleus into the cytoplasm and in the virus particle assembly process are discussed.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- APOSTOLOV K., FLEWETT T. H. INTERNAL STRUCTURE OF INFLUENZA VIRUS. Virology. 1965 Jul;26:506–508. doi: 10.1016/0042-6822(65)90014-0. [DOI] [PubMed] [Google Scholar]
- Briedis D. J., Conti G., Munn E. A., Mahy B. W. Migration of influenza virus-specific polypeptides from cytoplasm to nucleus of infected cells. Virology. 1981 May;111(1):154–164. doi: 10.1016/0042-6822(81)90661-9. [DOI] [PubMed] [Google Scholar]
- Bächi T., Gerhard W., Lindenmann J., Mühlethaler K. Morphogenesis of influenza A virus in Ehrlich ascites tumor cells as revealed by thin-sectioning and freeze-etching. J Virol. 1969 Nov;4(5):769–776. doi: 10.1128/jvi.4.5.769-776.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chamberlain J. P. Fluorographic detection of radioactivity in polyacrylamide gels with the water-soluble fluor, sodium salicylate. Anal Biochem. 1979 Sep 15;98(1):132–135. doi: 10.1016/0003-2697(79)90716-4. [DOI] [PubMed] [Google Scholar]
- Compans R. W., Dimmock N. J. An electron microscopic study of single-cycle infection of chick embryo fibroblasts by influenza virus. Virology. 1969 Nov;39(3):499–515. doi: 10.1016/0042-6822(69)90098-1. [DOI] [PubMed] [Google Scholar]
- Giesendorf B., Bosch F. X., Orlich M., Scholtissek C., Rott R. Studies on the temperature sensitivity of influenza A virus reassortants nonpathogenic for chicken. Virus Res. 1986 Jul;5(1):27–42. doi: 10.1016/0168-1702(86)90063-8. [DOI] [PubMed] [Google Scholar]
- Gregoriades A., Frangione B. Insertion of influenza M protein into the viral lipid bilayer and localization of site of insertion. J Virol. 1981 Oct;40(1):323–328. doi: 10.1128/jvi.40.1.323-328.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hankins R. W., Nagata K., Kato A., Ishihama A. Mechanism of influenza virus transcription inhibition by matrix (M1) protein. Res Virol. 1990 May-Jun;141(3):305–314. doi: 10.1016/0923-2516(90)90002-z. [DOI] [PubMed] [Google Scholar]
- Herz C., Stavnezer E., Krug R., Gurney T., Jr Influenza virus, an RNA virus, synthesizes its messenger RNA in the nucleus of infected cells. Cell. 1981 Nov;26(3 Pt 1):391–400. doi: 10.1016/0092-8674(81)90208-7. [DOI] [PubMed] [Google Scholar]
- Hiti A. L., Nayak D. P. Complete nucleotide sequence of the neuraminidase gene of human influenza virus A/WSN/33. J Virol. 1982 Feb;41(2):730–734. doi: 10.1128/jvi.41.2.730-734.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janda J. M., Davis A. R., Nayak D. P., De B. K. Diversity and generation of defective interfering influenza virus particles. Virology. 1979 May;95(1):48–58. doi: 10.1016/0042-6822(79)90400-8. [DOI] [PubMed] [Google Scholar]
- Klenk H. D., Compans R. W., Choppin W. P. An electron microscopic study of the presence or absence of neuraminic acid in enveloped viruses. Virology. 1970 Dec;42(4):1158–1162. doi: 10.1016/0042-6822(70)90368-5. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Markushin S., Ghiasi H., Sokolov N., Shilov A., Sinitsin B., Brown D., Klimov A., Nayak D. Nucleotide sequence of RNA segment 7 and the predicted amino sequence of M1 and M2 proteins of FPV/Weybridge (H7N7) and WSN (H1N1) influenza viruses. Virus Res. 1988 May;10(2-3):263–271. doi: 10.1016/0168-1702(88)90021-4. [DOI] [PubMed] [Google Scholar]
- Martin K., Helenius A. Nuclear transport of influenza virus ribonucleoproteins: the viral matrix protein (M1) promotes export and inhibits import. Cell. 1991 Oct 4;67(1):117–130. doi: 10.1016/0092-8674(91)90576-k. [DOI] [PubMed] [Google Scholar]
- Martin K., Helenius A. Transport of incoming influenza virus nucleocapsids into the nucleus. J Virol. 1991 Jan;65(1):232–244. doi: 10.1128/jvi.65.1.232-244.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murti K. G., Brown P. S., Bean W. J., Jr, Webster R. G. Composition of the helical internal components of influenza virus as revealed by immunogold labeling/electron microscopy. Virology. 1992 Jan;186(1):294–299. doi: 10.1016/0042-6822(92)90084-3. [DOI] [PubMed] [Google Scholar]
- Palese P., Schulman J. L. Differences in RNA patterns of influenza A viruses. J Virol. 1976 Mar;17(3):876–884. doi: 10.1128/jvi.17.3.876-884.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pattnaik A. K., Brown D. J., Nayak D. P. Formation of influenza virus particles lacking hemagglutinin on the viral envelope. J Virol. 1986 Dec;60(3):994–1001. doi: 10.1128/jvi.60.3.994-1001.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penman S. RNA metabolism in the HeLa cell nucleus. J Mol Biol. 1966 May;17(1):117–130. doi: 10.1016/s0022-2836(66)80098-0. [DOI] [PubMed] [Google Scholar]
- Ritchey M. B., Palese P. Identification of the defective genes in three mutant groups of influenza virus. J Virol. 1977 Mar;21(3):1196–1204. doi: 10.1128/jvi.21.3.1196-1204.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruigrok R. W., Calder L. J., Wharton S. A. Electron microscopy of the influenza virus submembranal structure. Virology. 1989 Nov;173(1):311–316. doi: 10.1016/0042-6822(89)90248-1. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugiura A., Tobita K., Kilbourne E. D. Isolation and preliminary characterization of temperature-sensitive mutants of influenza virus. J Virol. 1972 Oct;10(4):639–647. doi: 10.1128/jvi.10.4.639-647.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugiura A., Ueda M., Tobita K., Enomoto C. Further isolation and characterization of temperature-sensitive mutants of influenza virus. Virology. 1975 Jun;65(2):363–373. doi: 10.1016/0042-6822(75)90042-2. [DOI] [PubMed] [Google Scholar]
- Wakefield L., Brownlee G. G. RNA-binding properties of influenza A virus matrix protein M1. Nucleic Acids Res. 1989 Nov 11;17(21):8569–8580. doi: 10.1093/nar/17.21.8569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winter G., Fields S. The structure of the gene encoding the nucleoprotein of human influenza virus A/PR/8/34. Virology. 1981 Oct 30;114(2):423–428. doi: 10.1016/0042-6822(81)90223-3. [DOI] [PubMed] [Google Scholar]
- Ye Z. P., Baylor N. W., Wagner R. R. Transcription-inhibition and RNA-binding domains of influenza A virus matrix protein mapped with anti-idiotypic antibodies and synthetic peptides. J Virol. 1989 Sep;63(9):3586–3594. doi: 10.1128/jvi.63.9.3586-3594.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ye Z. P., Pal R., Fox J. W., Wagner R. R. Functional and antigenic domains of the matrix (M1) protein of influenza A virus. J Virol. 1987 Feb;61(2):239–246. doi: 10.1128/jvi.61.2.239-246.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zakarian S., Smyth D. G. beta-Endorphin is processed differently in specific regions of rat pituitary and brain. Nature. 1982 Mar 18;296(5854):250–252. doi: 10.1038/296250a0. [DOI] [PubMed] [Google Scholar]
- Zvonarjev A. Y., Ghendon Y. Z. Influence of membrane (M) protein on influenza A virus virion transcriptase activity in vitro and its susceptibility to rimantadine. J Virol. 1980 Feb;33(2):583–586. doi: 10.1128/jvi.33.2.583-586.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Wyke K. L., Yewdell J. W., Reck L. J., Murphy B. R. Antigenic characterization of influenza A virus matrix protein with monoclonal antibodies. J Virol. 1984 Jan;49(1):248–252. doi: 10.1128/jvi.49.1.248-252.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]