Abstract
The histone H4 acetylation status of the active X (Xa) and inactive X (Xi) chromosomes was investigated at the level of individual genes. A moderate level of acetylation was observed along the lengths of genes on both the Xi and Xa, regardless of their X inactivation status. However, this moderate level of acetylation was modified specifically in promoter regions. Transcriptionally active genes showed elevated levels of acetylation at their promoters on both the Xi and Xa. In contrast, promoters of X-inactivated genes were markedly hypoacetylated, which coincided with the methylation of adjacent CG dinucleotides. This promoter-specific hypoacetylation may be a key component of an X inactivation machinery that operates at the level of individual genes.
In mammals, X chromosome dosage is equalized between males and females by X inactivation, the transcriptional silencing of one of the two X chromosomes in female cells. The chromatin of the inactive X (Xi) is distinct from all other chromosomes, including the active X (Xa), in that it is late replicating (1, 2), methylated on CG dinucleotides (3, 4), enriched in the histone H2A variant macroH2A (5), and hypoacetylated on histones H2A, H3, and H4 (6–8). These unique features of the Xi chromatin are associated with, and may contribute to, the silencing of its several thousand resident genes.
Although most genes on the Xi are transcriptionally repressed, there are clear exceptions. First, the XIST gene, which initiates X inactivation, is transcribed only from the Xi (9). Second, in both humans and mice, a number of genes escape X inactivation—that is, they are transcribed from the Xi as well as the Xa (for review see refs. 10 and 11). Genes that escape X inactivation typically have homologs on the Y chromosome, which in theory obviates their need for dosage compensation (12).
Among the unique properties of the Xi chromatin, CG methylation and late replication have been studied at the level of individual genes and were found to correlate closely with gene expression. Where examined, these traits are associated with particular genes that are silenced, but not with those that escape X inactivation (2, 13, 14). In contrast, histone acetylation on the Xi has not been examined extensively at the level of individual genes. The prevailing view that the Xi is hypoacetylated comes from immunofluorescence microscopy analysis, which showed that all chromosomes except the Xi stained brightly with antisera against acetylated histones in female cells (6–8). However, these microscopy studies on metaphase chromosomes are very limited in resolution and thus cannot address the acetylation status of individual genes.
A chromatin immunoprecipitation (IP) technique has been developed recently that allows the acetylation status of individual nucleosomes to be examined (15–17). We used this method to investigate the acetylation status of histone H4 associated with numerous genes on both the Xi and Xa. Our results indicate that nucleosomes on the Xi are in fact acetylated along the lengths of genes at low levels that are comparable to their counterparts on the Xa. Notable exceptions to this general observation are found at promoter regions. On the Xi, promoters of genes that undergo X inactivation are enriched in hypoacetylated isoforms of H4, whereas promoters of genes that escape transcriptional silencing are acetylated on H4 at elevated levels. The hypoacetylation of nucleosomes at the promoters of silenced genes correlates with methylation of coincident CG dinucleotides.
Materials and Methods
Cell Culture.
The GMO6318 (NIGMS Human Genetic Mutant Cell Repository) human/hamster somatic cell hybrid containing a single human Xa was maintained under selection in MEM medium containing HAT (hypoxanthine/aminopterin/thymidine) supplement (GIBCO/BRL). The X8–6T2S1 human/hamster cell hybrid (18) containing a single human Xi was grown in RPMI medium 1640 containing 10% (vol/vol) FCS.
Chromatin IPs.
Confluent cells on 10-cm plates were incubated in PBS containing 1% formaldehyde for 10 min at 37°C. Chromatin IPs were then performed by using various polyclonal antisera as described (15, 16). Briefly, sonication of crosslinked nuclei was performed in a cup horn (Branson Sonifier 450) under conditions that gave a range in DNA fragments from 200–1,000 bp. Anti-AcH4 or anti-H4Ac12 antiserum (5 μl; Upstate Biotechnology, Lake Placid, NY), anti-H4Ac5 or anti-H4Ac8 antiserum (10 μl; Upstate Biotechnology), and anti-H4Ac16 (5 μl; Serotec) or anti-HA antiserum (3 μl; Roche Molecular Biochemicals) were incubated overnight with precleared nuclear lysates. Immune complexes were then recovered with Protein A Sepharose beads.
PCR.
A portion (1/10) of the chromatin immunoprecipitate was added to a 20-μl reaction mix containing 1.5 mM MgCl2, 50 mM KCl, 10 mM Tris (pH 9.0), 1% Triton X-100, 0.5 μM each primer, 0.1 mM dNTPs, and 1 unit of Taq polymerase. Competitive PCRs contained an additional 1 unit of Taq Extender (Stratagene). After denaturation at 95°C for 1.5 min, 30 cycles of PCR were performed; each cycle consisted of 1 min at 95°C, 45 s at 60°C, and 45 s at 72°C. Under these conditions, PCR product yield depended linearly on input genomic DNA up to a maximum of 50 ng of DNA. Typically, less than 10 ng of genomic DNA from the chromatin IPs was added to PCRs, which is well within the linear range of the assay. Products were then resolved on 4% (vol/vol) NuSieve agarose (FMC) gels. Quantitation of PCR products was performed with the Eagle Eye II imaging system and eaglesight software (Stratagene).
Methylation Assays.
Genomic DNA was prepared from human female (293) cells, human male lymphoblastoid cells, or human/hamster somatic cell hybrids that contained either the human Xi or the human Xa. The methylation status of promoter regions of all genes other than XIST was measured by PCR with genomic DNA that had been extensively digested with HpaII as template. The methylation status of the XIST promoter was assayed by PCR with genomic DNA that had been digested with HhaI, AvaI, and BstU I as described (19). Primers spanned several methylation-sensitive restriction sites in the 5′ end of tested genes. Template DNA (50 ng) was subjected to between 28 and 31 cycles of PCR under the conditions described above, supplemented with 2% (vol/vol) DMSO.
Primer Sequences.
The primer sets shown in Table 1 are specific for human genomic DNA.
Table 1.
Primers for PCR analysis
| Gene | Forward | Reverse |
|---|---|---|
| Promoter-specific primers | ||
| OCRL | Refer to OCRL (0) below | Refer to OCRL (0) below |
| PGK1 | ACGCGGCTGCTCTGGGC | TTAGGGGCGGAGCAGGAAG |
| SMCX | CTTGTTCCTCCGCCGTTGCA | CCATCTTGGTTTGTCAGCGT |
| SB1.8 | CAGGGTGCTGTGGAATCTATT | TTGTCATACTCCTGCGCCA |
| POLA | CTGGGGAAAACGATCCAACC | CTGAAAGCCAATCAGCGGC |
| SOX3 | CGAACCTGTCAATCACGGGT | CCTGATGAGTTCTCTCGAAC |
| IDS | GCGCAGTCTTCATGGGTTC | CGGGGTGGCGGCATTTCG |
| XPCT | CTGGCCCGGCTCCTGGC | GCTTTGTTTGCGCCAACCTG |
| NDP | TGGAAAAGTGCCATTGCAGT | CTCGCCCTTGTTCTGCTACG |
| XIST | xst31r (ref. 43) | xst 29r (ref. 43) |
| ZFX | Refer to ZFX (0) below | Refer to ZFX (0) below |
| ZFX-specific primers | ||
| ZFX (−8) | CACATCTCTCCTCTACTTCCT | TGCTTCCCAGGTTTTCACTA |
| ZFX (0) | GTGCTGTGTTAAAGGATAGC | AGGAGCCCAATTGGGTATGG |
| ZFX (20) | CTGGTTACCCTTGTGGGAAC | GGTCTATCAAGTGCACACAT |
| ZFX (50) | AAGACTGTACCGCCTTCACT | TGGGTACAGGTGGTTCTCC |
| ZFX (65) | GGTCACTGTAATGCCAGATC | GCATATAGGCATAGCATCTG |
| XIST-specific primers | ||
| XIST (−0.5) | TCCGTTCTCTTATGGTTGGG | TACCATCTTCAAGGGACTTC |
| XIST (0) | ACGTGTCAAGAAGACACTAG | xst 29r (ref. 43) |
| XIST (1.5) | AAGGTCTTGCCGCAGTGTAA | GTTGGGTTATGCAGCAATCC |
| XIST (3) | CTGTGTTGGCCACCTAAAAC | GCACAGCAAAAAGCGCAGTA |
| XIST (10) | CTTGTTAAGCAAGCGCCCCA | CATGACACCATGGCTACCTG |
| XIST (20) | TGAAGACCCATGTCTCTACA | TGGCTCACGTTCTGCTTTTC |
| XIST (25) | CTTTGCTCTCCTAGATGTGG | GAGAAGGGGAAGGGGTAACA |
| XIST (32) | CAGCATGGGTGACCACCAGA | GTTAGGGACAGTGAGTTAGAA |
| OCRL-specific primers | ||
| OCRL (0) | ACAAGTCTAGCTCCCAGCT | CCGATCCGACGACACTGGC |
| OCRL (1) | GAAAGACTTCCCAGTTTCTG | ATCACCATATTTGGCCTGAC |
| OCRL (2) | CCAACACCATGCGGAAGCTC | TATCAACAGGCCACTGTCTG |
| OCRL (5) | AGGCATTTAGCTACCAGAAG | CAGGGACCTATATGACAGGA |
| OCRL (25) | GCCTATCACTTCCTTGCTTA | AGGCACAGAGACAGTAAATC |
| OCRL (40) | GCAGCAAAACAGCTGGCTGT | AGGAGAAGGAGCATACAGAA |
Results
Silencing of X-Linked Genes Correlates with Hypoacetylation of Histone H4 in Promoter Regions.
The chromatin IP assay was used to examine the acetylation status of histone H4 at selected sites on the human X chromosome. By design, this assay examines the acetylation status of particular genomic DNA sequences in a population of cells. The fact that female cells contain both an Xi and an Xa makes it difficult to examine individually the acetylation status of a particular sequence on a single X chromosome. To circumvent this difficulty, experiments were performed with human/hamster somatic cell hybrids that contained either the human Xa or the human Xi. These cell lines allow individual X chromosomes to be examined in isolation from their homologs. Previous reports indicate that gene expression, methylation status, and replication timing of the human X in these hybrid cells closely resemble those observed in human cells (2, 20, 21). Reverse transcription–PCR analysis was used to confirm the expression status of all 11 X-linked genes used in subsequent acetylation assays (data not shown). The expression status of these genes in the hybrid cells is listed in Table 2 and is consistent, in all cases, with their previously described X inactivation status (for review see ref. 10).
Table 2.
H4 acetylation status of promoter regions of X-linked genes
| Gene | X inactivation status | H4 acetylation
|
|
|---|---|---|---|
| Xi | Xa | ||
| ZFX | Escape | + | + |
| SB1.8 | Escape | + | + |
| SMCX | Escape | + | + |
| OCRL | Subject | − | + |
| PGK1 | Subject | − | + |
| POLA | Subject | − | + |
| SOX3 | Subject | − | + |
| IDS | Subject | − | + |
| XPCT | Subject | − | + |
| NDP | Subject | − | + |
| XIST | Active only on Xi | + | − |
+ indicates that the promoter region of the gene is present in the H4 acetylated fraction. − indicates that the promoter region of the gene is not detectable in the H4 acetylated fraction.
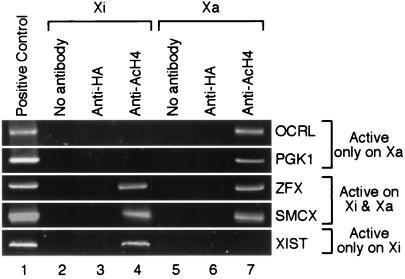
To perform chromatin IP assays, cells were fixed with formaldehyde followed by sonication. This procedure produced chromatin fragments containing 200–1,000 bp of DNA, which corresponded to one or several nucleosomes (data not shown). Chromatin IPs were performed with antisera that recognized histone H4 acetylated on any one of its four N-terminal lysine residues (anti-AcH4) but that did not recognize unacetylated H4 (22). Immunoprecipitates were subjected to PCR amplification to assay for the presence of particular sequences in the acetylated fraction. The promoter regions of OCRL and PGK1, two genes known to be silenced on the Xi (10), were examined first (Fig. 1). PCR products corresponding to these regions were not detected in immunoprecipitates recovered from the Xi-containing hybrid but were present in IPs recovered from the Xa-containing hybrid. In contrast, PCR products from the promoter regions of ZFX and SMCX, two genes known to be expressed from both Xi and Xa (23–25), were recovered by chromatin IPs from both hybrids. Finally, a PCR product corresponding to the promoter region of XIST, a gene expressed only from the Xi (9), was recovered by chromatin IPs only from the Xi-containing hybrid.
Figure 1.
Histone H4 acetylation at promoter regions of X-linked genes. Chromatin IPs were performed with antisera as designated on human/hamster somatic cell hybrids that contain either the human Xi or human Xa. PCRs were performed on immunoprecipitates with primers that amplified promoter-proximal regions of genes. The positive control is genomic DNA (lane 1). Negative controls for IPs were provided by experiments either with no antibody (lanes 2 and 5) or with anti-HA (hemagglutinin) antiserum, which does not recognize chromatin (lanes 3 and 6). The AcH4 antiserum recognizes histone H4 acetylated at any one of four positions in its N terminus (lanes 4 and 7).
The above results strongly suggest a correlation between the acetylation status of H4 in nucleosomes at the promoter and transcriptional activity of the adjacent gene. To substantiate this correlation further, additional genes that either escape or undergo X inactivation were assayed. As summarized in Table 2, hypoacetylation of histone H4 at the promoter indeed correlated with transcriptional silencing for all X-linked genes examined.
Analysis of Site-Specific Lysine Acetylation Confirms Acetylation Status of Promoter Regions.
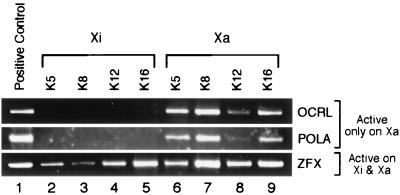
The amino terminus of histone H4 contains four highly conserved lysines (Lys-5, -8, -12, and -16) that can each serve as a substrate for acetylation by histone acetyltransferases. Because the antisera used above recognized acetylation at any of the four lysine acceptor sites, it was not clear whether the promoter-proximal acetylation was present on all acceptor sites or whether it was confined to only a subset of sites. To determine the acetylation status of individual lysine residues, chromatin IPs were performed with antisera that could specifically recognize H4 isoforms acetylated at particular lysine residues (26). The antiserum specific for H4 acetylated on Lys-5, for instance, would recognize the histone only if it were acetylated on Lys-5. Nucleosomes at the transcriptionally silent OCRL and POLA promoters on the Xi were indeed deficient in acetylation of all four lysine acceptor sites on the Xi (Fig. 2). In contrast, nucleosomes at the transcriptionally active OCRL and POLA promoters on the Xa or at the ZFX promoter on both the Xi and Xa were acetylated on all four lysine acceptor sites.
Figure 2.
Site-specific acetylation of histone H4 at promoter regions. Chromatin IPs were performed on hybrid cells by using antisera that distinguish among H4 isoforms acetylated on particular lysine residues (K5, K8, K12, or K16) as indicated. PCR primers were the same as those listed in Table 2 to amplify promoter regions of X-linked genes. Genomic DNA provided a positive control for the PCR assay (lane 1).
Genes on both Xi and Xa Are Acetylated at Similar Levels in Regions Downstream of the Promoter.
The histone H4 acetylation status along the entire lengths of several representative genes was determined. The ZFX gene was chosen to represent genes that escape X inactivation, which are expressed from both the Xi and Xa (23). To compare the degree of H4 acetylation at various sites along the ZFX locus, in both Xi- and Xa-containing hybrid cells, a competitive PCR assay was used (17). In this assay, chromatin IPs were subjected to PCR with two pairs of primers; one pair that amplified the ZFX promoter region was added to all reactions as an internal standard, whereas the second primer pair was varied from reaction to reaction to sample different positions along the ZFX locus. PCR conditions were optimized such that the total amount of product obtained depended linearly on the amount of input DNA (see Materials and Methods). The degree of H4 acetylation at a particular site was then determined by its relative enrichment compared with the ZFX promoter in the chromatin immunoprecipitate. Finally, to control for differences in amplification efficiency between each pair of primers, the ratio of the two products from immunoprecipitates was normalized to the ratio obtained from an input DNA control. This input DNA control was derived from cells that had been processed in parallel but that had not been subjected to chromatin IP.
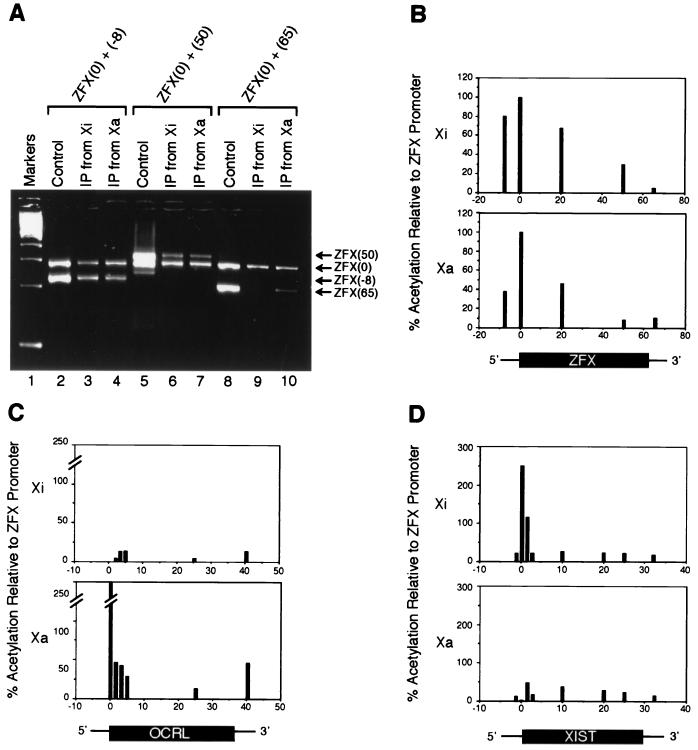
Fig. 3A shows a representative gel image of the competitive PCR analysis that sampled three sites in the ZFX genomic locus, with the ZFX promoter-proximal region ZFX(0) as the internal standard. The three sites are ZFX (−8) (8 kb upstream of the transcription start site), ZFX (50) (50 kb downstream from the transcription start site within the transcribed portion of the gene), and ZFX (65) [65 kb downstream from the promoter and 5 kb downstream from the poly(A) site]. Densitometry analysis of PCR products, after correcting for variations in amplification efficiency between different primer sets, was used to calculate the ratio of each site relative to the promoter site in the Xi- or Xa-containing hybrids. Fig. 3B is a graphical representation of H4 acetylation profiles at several sites along the ZFX gene on both Xi and Xa, obtained by averaging results from two independent experiments that varied by less than 10%. Because the same immunoprecipitate was used to assay acetylation levels of various sites along a single X chromosome, comparisons among these sites are internally controlled. Comparisons of the same site between the two chromosomes—Xi and Xa—are also valid, because when equal amounts of chromatin from the Xi- and Xa-containing cells were used in IPs, comparable amounts of PCR product were obtained for the internal standard, ZFX (0), from Xi and Xa (data not shown). Two features are evident in the profiles. First, the highest levels of H4 acetylation are found at the ZFX promoter on both Xi and Xa. The levels of acetylation decrease both upstream of the promoter and along the transcription unit in each case. Second, the degree and distribution of H4 acetylation along the ZFX gene are comparable between Xi and Xa.
Figure 3.
Quantitative PCR analysis of histone H4 acetylation along X-linked genes. (A) Sample gel of PCR with internal standard used to quantitate relative enrichment of various ZFX sites in chromatin immunoprecipitate. Two sets of primers were used in each PCR: one set, ZFX (0), amplified the ZFX promoter-proximal region and served as an internal standard of comparison, whereas the other set sampled positions along the ZFX genomic locus: ZFX (−8) is 8 kilobases (kb) upstream of the promoter; ZFX (20) is 20 kb downstream of the promoter (data not shown); ZFX (50) is 50 kb downstream of the promoter in the transcribed portion of the gene; and ZFX(65) is 5 kb downstream of the poly(A) site. The control is genomic DNA isolated from cells that were processed in parallel but that were not subjected to IPs. To eliminate differences that were caused by amplification efficiency between primer sets, the amplification ratio from immunoprecipitates was normalized to the ratio obtained with control genomic DNA. After normalization, the relative intensity of the sample PCR product was used to estimate its enrichment relative to ZFX (0) in the immunoprecipitate. (B–D) Graphical representations of relative H4 acetylation levels along X-linked genes as assayed by PCR with an internal standard. Three genes were assayed: ZFX, which escapes X inactivation (B); OCRL, which undergoes X inactivation (C); and XIST, which is transcribed exclusively from Xi (D). The ZFX (0) site was arbitrarily set as the unit of comparison (100%), and acetylation levels at all other sites were expressed as a percentage relative to ZFX (0).
Similar assays were also performed for the OCRL gene, which represents the many X-inactivated genes expressed only from the Xa (10). The degree of acetylation along the gene was once again normalized to the ZFX promoter on the same chromosome. This experiment was also duplicated, and the results, which varied by less than 10% between the two experiments, were averaged and are depicted in graphical form in Fig. 3C. The profile obtained for OCRL on the Xa was similar to that obtained for ZFX: the acetylation levels were greatest at the promoter region and declined to much lower levels in the downstream portion of the gene. However, on the Xi, there was an absence of acetylation of OCRL in the vicinity of the promoter, not only relative to the promoter on the Xa, but also relative to the remainder of the OCRL locus.
Finally, PCR was performed on XIST, which is the only gene known to be expressed exclusively from the Xi (9). By including an internal standard against the ZFX promoter, as was done before, the correlation between promoter-proximal acetylation and gene expression was again evident: the XIST promoter was highly acetylated on the Xi, from which it is transcribed, but was acetylated at a markedly lower level on the Xa, from which it is not transcribed (Fig. 3D). The differences between alleles on Xi and Xa were again confined to the promoter region, and the overall acetylation levels were comparable between the two alleles outside of the promoter. These results are in agreement with a recent analysis of the acetylation profile of the Xist locus in male and female mouse cells (27).
The above results suggest that there is a fairly constant level of histone H4 acetylation extending throughout most of the genetic loci on the Xi. This low-level acetylation does not correlate with transcriptional status. In contrast, the status of H4 acetylation in the vicinity of the promoter strongly correlates with transcriptional activity of the gene.
Methylation of Promoter Regions of X-linked Genes Correlates with H4 Hypoacetylation and Transcriptional Silencing.
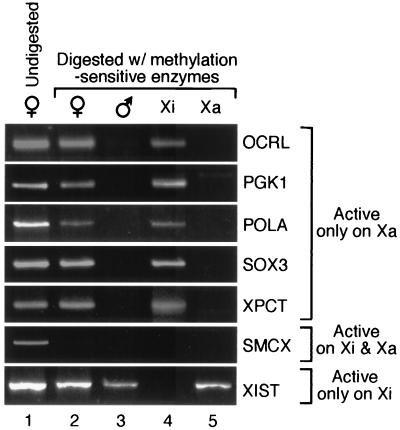
To explore whether the hypoacetylation observed at promoters of silenced genes coincided with methylation of CG dinucleotides, genomic DNA samples from female cells, male cells, and human/hamster cell hybrids were digested with methylation-sensitive enzymes. Digestion was monitored by PCR assays with primers that spanned several methylation-sensitive restriction sites at promoters of tested genes. If the CG sites were methylated, they would resist digestion, and the DNA would then be available to serve as template in the PCR. Fig. 4 shows the results of this analysis. Methylation was detected at promoter regions of X-inactivated genes, including OCRL, PGK1, POLA, SOX3, and XPCT, in all cell types that contained an Xi. In contrast, the promoter of the SMCX gene, which is acetylated and expressed from the Xi as well as the Xa, was not methylated detectably on either Xi or Xa. Finally, the promoter region of the XIST gene was methylated only in cells that contained an Xa chromosome. These results are consistent with previous methylation studies of several of these genes (12, 19, 28). The most significant aspect of these findings is that the primers used here were the same as those used to assess the H4 acetylation status of promoter regions shown in Table 1. The findings indicate that the lack of acetylation coincides with the presence of methylation at promoters of X-inactivated genes. In addition, these results confirm prior reports that human X chromosomes retain the same methylation status in hybrid cells as in intact human cells (29).
Figure 4.
Methylation analysis of promoter regions of X-linked genes. Genomic DNA prepared from various sources was digested with methylation-sensitive enzymes. Digestion was monitored by a PCR assay with primers that spanned several restriction sites. Methylated sequences escape digestion and can serve as template in the PCR. Undigested genomic DNA provided a positive control for the PCR assay (lane 1). PCR assays were performed on digested DNA isolated from female (293) cells (lane 2), male (lymphoblastoid) cells (lane 3), and human/hamster cell hybrids containing either the Xi (lane 4) or Xa (lane 5). PCR primers were the same as used to assess the H4 acetylation status shown in Table 2.
Discussion
Previous analyses of the histone acetylation status of the Xa and Xi chromatin have relied primarily on immunofluorescence microscopy of metaphase cells, which examines the acetylation status of large regions of chromatin (6–8). The chromatin IP studies described here provide extensive examination of individual nucleosomes associated with numerous X-linked genes. These results revealed complex patterns of acetylation that were not previously detected.
First, a low-level H4 acetylation is present along all examined genes on the Xi, except at the promoters of silenced genes. This low-level acetylation is comparable to that associated with genes on the Xa and does not correlate with transcription. Second, promoter regions of expressed genes, regardless of whether they are located on the Xi or Xa, are hyperacetylated relative to their downstream regions. Third and most significantly, promoter regions of X-inactivated genes are dramatically hypoacetylated, even compared with the low-level acetylation found in downstream regions of the same loci. Thus, promoter hypoacetylation is another feature, along with CG methylation and late replication, that correlates with silencing of individual genes on the Xi. Finally, DNA methylation coincided with promoter-proximal hypoacetylation: methylation of CG dinucleotides was observed at hypoacetylated promoters of silenced genes on the Xi but not at hyperacetylated promoters of active genes.
It has been shown in yeast cells that hyperacetylation of chromatin at an active promoter is the direct result of recruitment of the histone acetyltransferase Gcn5p (30). A similar recruitment process may account for the hyperacetylation of active promoters on the mammalian X chromosomes observed here. For instance, histone acetyltransferase activity may be recruited to active promoters on X chromosomes by a variety of transcriptional activators. In addition, some of the general transcription factors associated with most active promoters possess intrinsic histone acetyltransferase activity (31), which may facilitate promoter hyperacetylation and transcription of the adjacent genes.
Just as histone hyperacetylation at active promoters may be due to the recruitment of histone acetyltransferases, the hypoacetylation observed at promoters of X-inactivated genes may be due to recruitment of histone deacetylases. Given the correlation between DNA methylation and histone hypoacetylation in promoter regions in these hybrid cell lines, it is likely that the hypoacetylation at X-inactivated promoters in differentiated cells may be maintained through cell division by methyl-directed recruitment of deacetylase-containing complexes. In fact, it was shown recently that methylated CG sequences bind MeCP2, which can recruit complexes with histone deacetylase activity (32, 33). Alternatively, it is possible that specific factors recognize sequences other than 5-methylcytosine near the promoters of X-inactivated genes and target deacetylase complexes to those regions.
Histone deacetylation is probably critical for the initiation of X inactivation as well as for its maintenance. This notion is supported by the observation that chemical inhibition of deacetylase activity in differentiating embryonic stem cells prevents the appearance of several properties normally associated with the Xi chromosome, including its global hypoacetylation (34). Because hypoacetylation of the Xi precedes methylation of CG islands as differentiating embryonic stem cells undergo X inactivation (35), it is unlikely that recognition of methylated nucleotides is responsible for recruiting deacetylase-containing complexes to promoters during this transition. It is more likely that the hypoacetylation that occurs during initiation of X inactivation depends on sequence-specific targeting of deacetylase-containing complexes to particular promoters.
Although the metaphase Xi, as observed by immunofluorescence microscopy, may be hypoacetylated relative to all other chromosomes (6–8), gene-containing regions of the Xi are clearly acetylated in asynchronous cells, as measured by the chromatin IP assay. In fact, promoters of expressed genes are hyperacetylated on the Xi. The apparent differences between immunofluorescence microscopy and the chromatin IP assay in detecting histone acetylation status could reflect a multitude of differences between the two experimental systems, including detection threshold, signal resolution, and cell-cycle-associated variations in histone acetylation. Indeed, it has been shown that bulk histone acetylation is reduced in metaphase cells relative to interphase cells (26, 36).
Several features of the Xi chromatin, such as colocalization with XIST RNA and formation of a Barr body, have been described at the level of the entire chromosome, leading to the view that changes in chromatin structure of the Xi occur on a chromosome-wide basis. Yet, the data presented here, that acetylation differences between expressed and silenced alleles are confined primarily to promoter regions, as well as previous data showing that methylation of CG islands is uniquely confined to X inactivated genes (3, 4), indicate that this model is incomplete. Instead, the data suggest that there are several, perhaps hierarchical levels of control that regulate X-linked gene expression. One level of control may indeed operate at a broad, regional level to initiate differential regulation of the two X chromosomes and to produce alterations that are generally inhibitory to transcription. At another level, because the acetylation status of individual promoters correlates with the activity of adjacent genes, local sequences may ultimately specify the transcriptional activity of individual promoters.
Evolutionary studies are consistent with a model in which regulation of gene expression from the Xi is specified on a local basis. Phylogenetic comparisons have shown that X inactivation did not evolve in a single, broad sweep; rather, genes acquired the potential for X inactivation throughout evolution on an individual or regional basis (12). For each gene, acquisition of X inactivation was an eventual response to the decay of its homolog on the Y chromosome. The data presented here suggest that this acquisition of X inactivation potential may result from sequence evolution within the upstream regulatory regions of target genes, so as to alter the transcriptional competence of promoters.
Initiation of X inactivation during embryonic development involves a cascade of events. The first detectable event is a dramatic increase in the level of Xist RNA associated with the X that will become inactivated (37–39). Xist expression is essential for the occurrence of downstream events (40–42)—such as late replication, H4 hypoacetylation, DNA methylation, and down-regulation of gene expression—which seem to proceed in a defined order during differentiation (35). If Xist RNA directs these downstream processes, it may do so—directly or indirectly—by targeting factors that are repressive to transcription to promoters of X-inactivated genes. Histone deacetylases are likely to be one component of an X inactivation machinery that is recruited to the promoters of genes that are silenced on the Xi. The resulting promoter-specific hypoacetylation may then provide a mechanistic basis for the transcriptional silencing of X-inactivated genes.
Acknowledgments
We thank R. Scott Hansen for providing us with the X8–6T2S1 human/hamster cell hybrid; Dave Allis and Richard Treisman for technical advice; Dave Allis, Julian Borrow, Steve Buratowski, Derek Dykxhoorn, Hristo Houbaviy, Rudolph Jaenisch, Jae B. Kim, Bob Kingston, Bruce Lahn, David Page, Barbara Panning, John Pehrson, and Rick Young for helpful discussions and/or critical reading of this manuscript. This work was supported by United States Public Health Service Grants RO1-AI32486 and PO1-CA42063 from the National Institutes of Health and, partially, by National Cancer Institute Cancer Center Support (Core) Grant P30-CA14051.
Abbreviations
- Xi
inactive X chromosome
- Xa
active X chromosome
- IP
immunoprecipitation
- kb
kilobase
References
- 1.Schmidt M, Migeon B R. Proc Natl Acad Sci USA. 1990;87:3685–3689. doi: 10.1073/pnas.87.10.3685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hansen R S, Canfield T K, Fjeld A D, Gartler S M. Hum Mol Genet. 1996;5:1345–1353. doi: 10.1093/hmg/5.9.1345. [DOI] [PubMed] [Google Scholar]
- 3.Riggs A D. Cytogenet Cell Genet. 1975;14:9–25. doi: 10.1159/000130315. [DOI] [PubMed] [Google Scholar]
- 4.Norris D P, Brockdorff N, Rastan S. Mamm Genome. 1991;1:78–83. doi: 10.1007/BF02443782. [DOI] [PubMed] [Google Scholar]
- 5.Costanzi C, Pehrson J R. Nature (London) 1998;393:599–601. doi: 10.1038/31275. [DOI] [PubMed] [Google Scholar]
- 6.Jeppesen P, Turner B M. Cell. 1993;74:281–289. doi: 10.1016/0092-8674(93)90419-q. [DOI] [PubMed] [Google Scholar]
- 7.Belyaev N, Keohane A M, Turner B M. Hum Genet. 1996;97:573–578. doi: 10.1007/BF02281863. [DOI] [PubMed] [Google Scholar]
- 8.Boggs B A, Connors B, Sobel R E, Chinault A C, Allis C D. Chromosoma. 1996;105:303–309. doi: 10.1007/BF02524648. [DOI] [PubMed] [Google Scholar]
- 9.Brown C J, Ballabio A, Rupert J L, Lafreniere R G, Grompe M, Tonlorenzi R, Willard H F. Nature (London) 1991;349:38–44. doi: 10.1038/349038a0. [DOI] [PubMed] [Google Scholar]
- 10.Brown C J, Carrel L, Willard H F. Am J Hum Genet. 1997;60:1333–1343. doi: 10.1086/515488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Disteche C M. Am J Hum Genet. 1997;60:1312–1315. doi: 10.1086/515484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Jegalian K, Page D C. Nature (London) 1998;394:776–780. doi: 10.1038/29522. [DOI] [PubMed] [Google Scholar]
- 13.Mohandas T, Sparkes R S, Shapiro L J. Science. 1981;211:393–396. doi: 10.1126/science.6164095. [DOI] [PubMed] [Google Scholar]
- 14.Goodfellow P J, Mondello C, Darling S M, Pym B, Little P, Goodfellow P N. Proc Natl Acad Sci USA. 1988;85:5605–5609. doi: 10.1073/pnas.85.15.5605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Braunstein M, Rose A B, Holmes S G, Allis C D, Broach J R. Genes Dev. 1993;7:592–604. doi: 10.1101/gad.7.4.592. [DOI] [PubMed] [Google Scholar]
- 16.Alberts A S, Geneste O, Treisman R. Cell. 1998;92:475–487. doi: 10.1016/s0092-8674(00)80941-1. [DOI] [PubMed] [Google Scholar]
- 17.Rundlett S E, Carmen A A, Suka N, Turner B M, Grunstein M. Nature (London) 1998;392:831–835. doi: 10.1038/33952. [DOI] [PubMed] [Google Scholar]
- 18.Hansen R S, Ellis N A, Gartler S M. Mol Cell Biol. 1988;8:4692–4699. doi: 10.1128/mcb.8.11.4692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hendrich B D, Brown C J, Willard H F. Hum Mol Genet. 1993;2:663–672. doi: 10.1093/hmg/2.6.663. [DOI] [PubMed] [Google Scholar]
- 20.Hansen R S, Gartler S M. Proc Natl Acad Sci USA. 1990;87:4174–4178. doi: 10.1073/pnas.87.11.4174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hornstra I K, Yang T P. Mol Cell Biol. 1994;14:1419–1430. doi: 10.1128/mcb.14.2.1419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lin R, Leone J W, Cook R G, Allis C D. J Cell Biol. 1989;108:1577–1588. doi: 10.1083/jcb.108.5.1577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Schneider-Gadicke A, Beer-Romero P, Brown L G, Nussbaum R, Page D C. Cell. 1989;57:1247–1258. doi: 10.1016/0092-8674(89)90061-5. [DOI] [PubMed] [Google Scholar]
- 24.Agulnik A I, Mitchell M J, Mattei M G, Borsani G, Avner P A, Lerner J L, Bishop C E. Hum Mol Genet. 1994;3:879–884. doi: 10.1093/hmg/3.6.879. [DOI] [PubMed] [Google Scholar]
- 25.Wu J, Salido E C, Yen P H, Mohandas T K, Heng H H, Tsui L C, Park J, Chapman V M, Shapiro L J. Nat Genet. 1994;7:491–496. doi: 10.1038/ng0894-491. [DOI] [PubMed] [Google Scholar]
- 26.Turner B M, Fellows G. Eur J Biochem. 1989;179:131–139. doi: 10.1111/j.1432-1033.1989.tb14530.x. [DOI] [PubMed] [Google Scholar]
- 27.McCabe V, Formstone E J, O’Neill L P, Turner B M, Brockdorff N. Proc Natl Acad Sci USA. 1999;96:7155–7160. doi: 10.1073/pnas.96.13.7155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tribioli C, Tamanini F, Patrosso C, Milanesi L, Villa A, Pergolizzi R, Maestrini E, Rivella S, Bione S, Mancini M, et al. Nucleic Acids Res. 1992;20:727–733. doi: 10.1093/nar/20.4.727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hansen R S, Canfield T K, Stanek A M, Keitges E A, Gartler S M. Proc Natl Acad Sci USA. 1998;95:5133–5138. doi: 10.1073/pnas.95.9.5133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kuo M H, Zhou J, Jambeck P, Churchill M E, Allis C D. Genes Dev. 1998;12:627–639. doi: 10.1101/gad.12.5.627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mizzen C A, Yang X J, Kokubo T, Brownell J E, Bannister A J, Owen-Hughes T, Workman J, Wang L, Berger S L, Kouzarides T, Nakatani Y, Allis C D. Cell. 1996;87:1261–1270. doi: 10.1016/s0092-8674(00)81821-8. [DOI] [PubMed] [Google Scholar]
- 32.Jones P L, Veenstra G J, Wade P A, Vermaak D, Kass S U, Landsberger N, Strouboulis J, Wolffe A P. Nat Genet. 1998;19:187–191. doi: 10.1038/561. [DOI] [PubMed] [Google Scholar]
- 33.Nan X, Ng H H, Johnson C A, Laherty C D, Turner B M, Eisenman R N, Bird A. Nature (London) 1998;393:386–389. doi: 10.1038/30764. [DOI] [PubMed] [Google Scholar]
- 34.O’Neill L P, Keohane A M, Lavender J S, McCabe V, Heard E, Avner P, Brockdorff N, Turner B M. EMBO J. 1999;18:2897–2907. doi: 10.1093/emboj/18.10.2897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Keohane A M, O’Neill L P, Belyaev N D, Lavender J S, Turner B M. Dev Biol. 1996;180:618–630. doi: 10.1006/dbio.1996.0333. [DOI] [PubMed] [Google Scholar]
- 36.D’Anna J A, Gurley L R, Tobey R A. Exp Cell Res. 1983;147:407–417. doi: 10.1016/0014-4827(83)90222-7. [DOI] [PubMed] [Google Scholar]
- 37.Beard C, Li E, Jaenisch R. Genes Dev. 1995;9:2325–2334. doi: 10.1101/gad.9.19.2325. [DOI] [PubMed] [Google Scholar]
- 38.Lee J T, Strauss W M, Dausman J A, Jaenisch R. Cell. 1996;86:83–94. doi: 10.1016/s0092-8674(00)80079-3. [DOI] [PubMed] [Google Scholar]
- 39.Panning B, Dausman J, Jaenisch R. Cell. 1997;90:907–916. doi: 10.1016/s0092-8674(00)80355-4. [DOI] [PubMed] [Google Scholar]
- 40.Penny G D, Kay G F, Sheardown S A, Rastan S, Brockdorff N. Nature (London) 1996;379:131–137. doi: 10.1038/379131a0. [DOI] [PubMed] [Google Scholar]
- 41.Marahrens Y, Panning B, Dausman J, Strauss W, Jaenisch R. Genes Dev. 1997;11:156–166. doi: 10.1101/gad.11.2.156. [DOI] [PubMed] [Google Scholar]
- 42.Lee J T, Jaenisch R. Nature (London) 1997;386:275–279. doi: 10.1038/386275a0. [DOI] [PubMed] [Google Scholar]
- 43.Brown C J, Hendrich B D, Rupert J L, Lafreniere R C, Xing Y, Lawrence J, Willard H F. Cell. 1992;71:527–542. doi: 10.1016/0092-8674(92)90520-m. [DOI] [PubMed] [Google Scholar]