Abstract
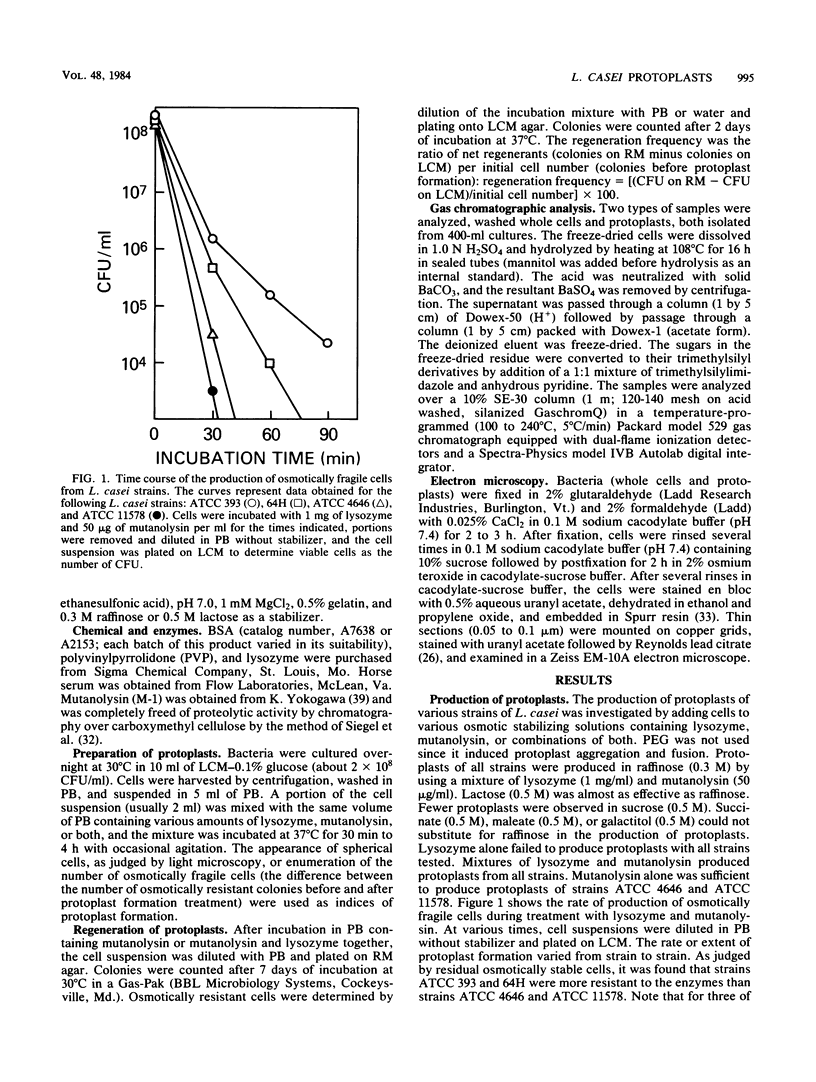
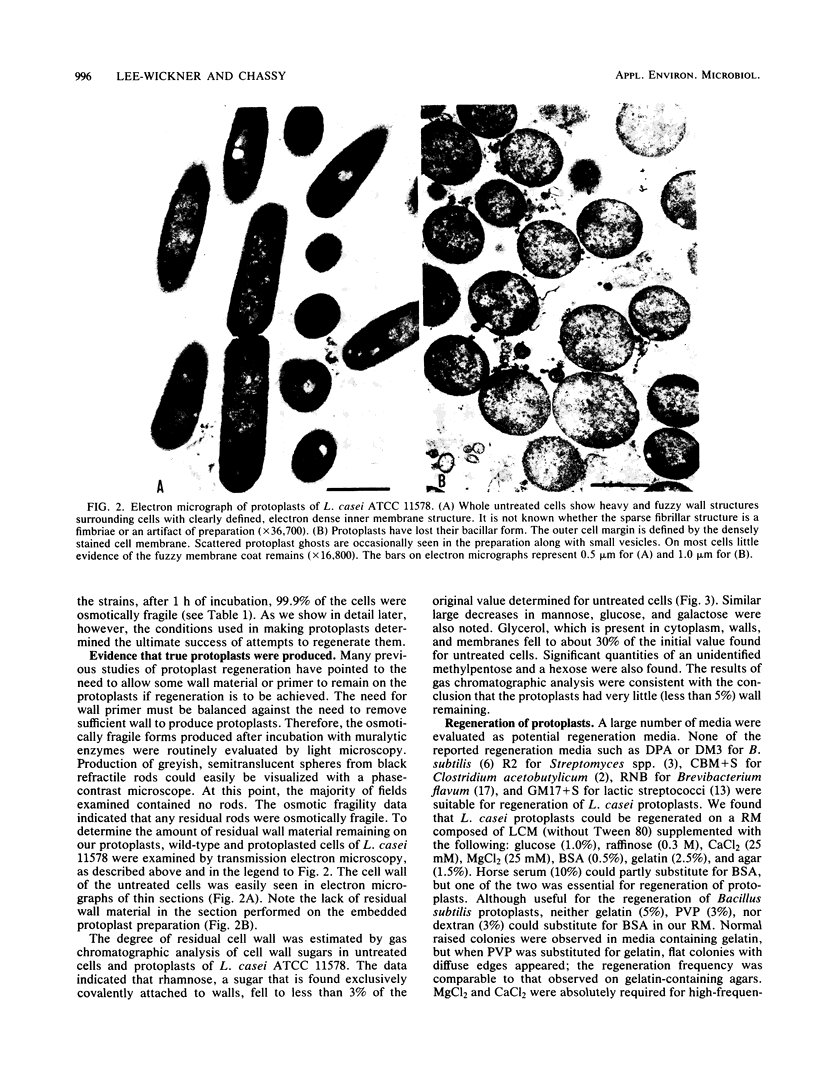
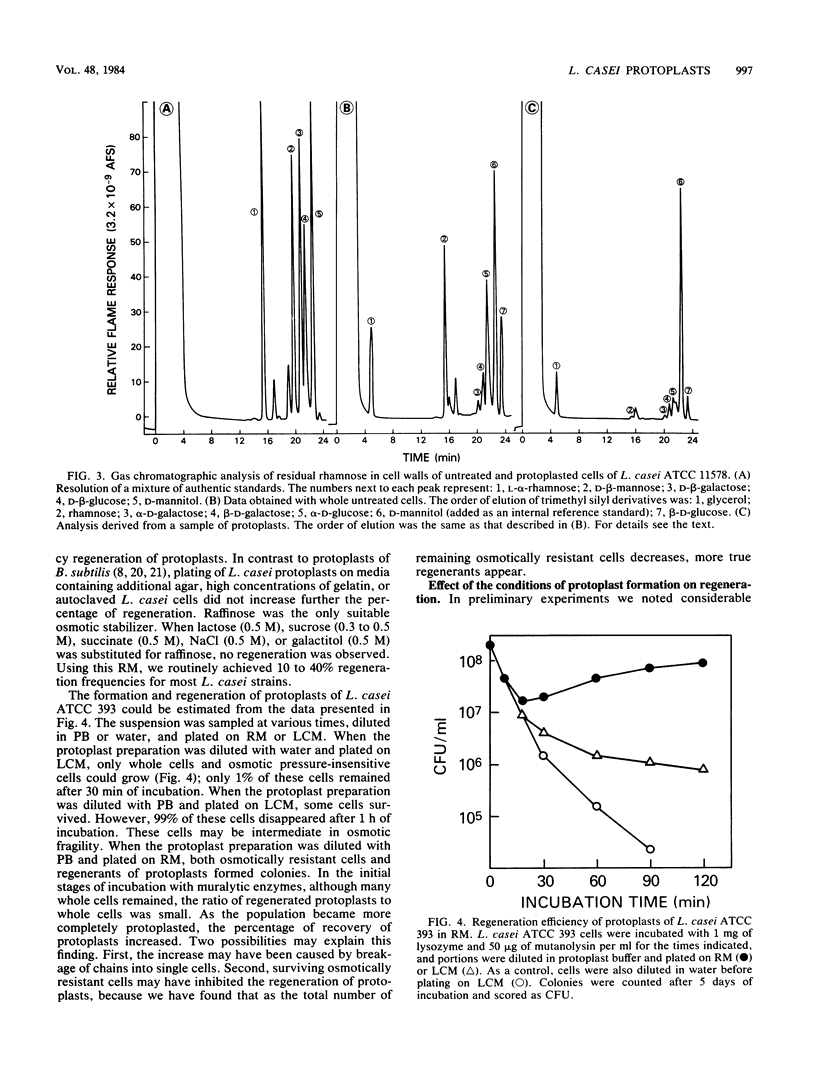
Methods for the production and regeneration of Lactobacillus casei protoplasts are described. Protoplasts of L. casei strains were obtained by treatment with mutanolysin or with mutanolysin and lysozyme together in a protoplast formation buffer containing 0.02 M HEPES (N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid) (pH 7.0), 1 mM MgCl2, 0.5% gelatin, and 0.3 M raffinose. Cells were regenerated on a complex medium supplemented with bovine serum albumin, MgCl2, CaCl2, gelatin, and raffinose. Lengthy digestion with lytic enzymes inhibited the capacity of protoplasts to regenerate. The optimum conditions of protoplast formation varied from strain to strain. Using predetermined optimal conditions it was possible to prepare protoplasts of several L. casei strains and regenerate them with 10 to 40% efficiency. The methods were applicable to other species of lactobacilli as well.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allcock E. R., Reid S. J., Jones D. T., Woods D. R. Clostridium acetobutylicum Protoplast Formation and Regeneration. Appl Environ Microbiol. 1982 Mar;43(3):719–721. doi: 10.1128/aem.43.3.719-721.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baltz R. H., Matsushima P. Protoplast fusion in Streptomyces: conditions for efficient genetic recombination and cell regeneration. J Gen Microbiol. 1981 Nov;127(1):137–146. doi: 10.1099/00221287-127-1-137. [DOI] [PubMed] [Google Scholar]
- Bibb M. J., Ward J. M., Hopwood D. A. Transformation of plasmid DNA into Streptomyces at high frequency. Nature. 1978 Jul 27;274(5669):398–400. doi: 10.1038/274398a0. [DOI] [PubMed] [Google Scholar]
- Bott K. F., Wilson G. A. Development of competence in the Bacillus subtilis transformation system. J Bacteriol. 1967 Sep;94(3):562–570. doi: 10.1128/jb.94.3.562-570.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang S., Cohen S. N. High frequency transformation of Bacillus subtilis protoplasts by plasmid DNA. Mol Gen Genet. 1979 Jan 5;168(1):111–115. doi: 10.1007/BF00267940. [DOI] [PubMed] [Google Scholar]
- Chassy B. M., Giuffrida A. Method for the lysis of Gram-positive, asporogenous bacteria with lysozyme. Appl Environ Microbiol. 1980 Jan;39(1):153–158. doi: 10.1128/aem.39.1.153-158.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clive D., Landman O. E. Reversion of Bacillus subtilis protoplasts to the bacillary form induced by exogenous cell wall, bacteria and by growth in membrane filters. J Gen Microbiol. 1970 May;61(2):233–243. doi: 10.1099/00221287-61-2-233. [DOI] [PubMed] [Google Scholar]
- Davies R. W., Gronenborn A. M. Molecular cloning of the gene for dihydrofolate reductase from Lactobacillus casei. Gene. 1982 Feb;17(2):229–233. doi: 10.1016/0378-1119(82)90077-4. [DOI] [PubMed] [Google Scholar]
- EFTHYMIOU C., HANSEN P. A. An antigenic analysis of Lactobacillus acidophilus. J Infect Dis. 1962 May-Jun;110:258–267. doi: 10.1093/infdis/110.3.258. [DOI] [PubMed] [Google Scholar]
- Fodor K., Alföldi L. Fusion of protoplasts of Bacillus megaterium. Proc Natl Acad Sci U S A. 1976 Jun;73(6):2147–2150. doi: 10.1073/pnas.73.6.2147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Götz F., Ahrné S., Lindberg M. Plasmid transfer and genetic recombination by protoplast fusion in staphylococci. J Bacteriol. 1981 Jan;145(1):74–81. doi: 10.1128/jb.145.1.74-81.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henderson G. B., Zevely E. M., Huennekens F. M. Purification and properties of a membrane-associated, folate-binding protein from Lactobacillus casei. J Biol Chem. 1977 Jun 10;252(11):3760–3765. [PubMed] [Google Scholar]
- Ito H., Fukuda Y., Murata K., Kimura A. Transformation of intact yeast cells treated with alkali cations. J Bacteriol. 1983 Jan;153(1):163–168. doi: 10.1128/jb.153.1.163-168.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kondo J. K., McKay L. L. Transformation of Streptococcus lactis Protoplasts by Plasmid DNA. Appl Environ Microbiol. 1982 May;43(5):1213–1215. doi: 10.1128/aem.43.5.1213-1215.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LANDMAN O. E., HALLE S. ENZYMICALLY AND PHYSICALLY INDUCED INHERITANCE CHANGES IN BACILLUS SUBTILIS. J Mol Biol. 1963 Dec;7:721–738. doi: 10.1016/s0022-2836(63)80119-9. [DOI] [PubMed] [Google Scholar]
- Landman O. E., Forman A. Gelatin-induced reversion of protoplasts of Bacillus subtilis to the bacillary form: biosynthesis of macromolecules and wall during successive steps. J Bacteriol. 1969 Aug;99(2):576–589. doi: 10.1128/jb.99.2.576-589.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landman O. E., Ryter A., Fréhel C. Gelatin-induced reversion of protoplasts of Bacillus subtilis to the bacillary form: electron-microscopic and physical study. J Bacteriol. 1968 Dec;96(6):2154–2170. doi: 10.1128/jb.96.6.2154-2170.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LeBlanc D. J., Hassell F. P. Transformation of Streptococcus sanguis Challis by plasmid deoxyribonucleic acid from Streptococcus faecalis. J Bacteriol. 1976 Oct;128(1):347–355. doi: 10.1128/jb.128.1.347-355.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee L. J., Hansen J. B., Jagusztyn-Krynicka E. K., Chassy B. M. Cloning and expression of the beta-D-phosphogalactoside galactohydrolase gene of Lactobacillus casei in Escherichia coli K-12. J Bacteriol. 1982 Dec;152(3):1138–1146. doi: 10.1128/jb.152.3.1138-1146.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- REYNOLDS E. S. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol. 1963 Apr;17:208–212. doi: 10.1083/jcb.17.1.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose A. H. The microbiological production of food and drink. Sci Am. 1981 Sep;245(3):126-34, 136, 138. doi: 10.1038/scientificamerican0981-126. [DOI] [PubMed] [Google Scholar]
- Saunders C. W., Guild W. R. Monomer plasmid DNA transforms Streptococcus pneumoniae. Mol Gen Genet. 1981;181(1):57–62. doi: 10.1007/BF00339005. [DOI] [PubMed] [Google Scholar]
- Saunders C. W., Guild W. R. Properties and transforming activities of two plasmids in Streptococcus pneumoniae. Mol Gen Genet. 1980;180(3):573–578. doi: 10.1007/BF00268062. [DOI] [PubMed] [Google Scholar]
- Scott K. F., Rolfe B. G., Shine J. Biological nitrogen fixation: primary structure of the Klebsiella pneumoniae nifH and nifD genes. J Mol Appl Genet. 1981;1(1):71–81. [PubMed] [Google Scholar]
- Siegel J. L., Hurst S. F., Liberman E. S., Coleman S. E., Bleiweis A. S. Mutanolysin-induced spheroplasts of Streptococcus mutants are true protoplasts. Infect Immun. 1981 Feb;31(2):808–815. doi: 10.1128/iai.31.2.808-815.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spurr A. R. A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res. 1969 Jan;26(1):31–43. doi: 10.1016/s0022-5320(69)90033-1. [DOI] [PubMed] [Google Scholar]
- Takahashi W., Yamagata H., Yamaguchi K., Tsukagoshi N., Udaka S. Genetic transformation of Bacillus brevis 47, a protein-secreting bacterium, by plasmid DNA. J Bacteriol. 1983 Dec;156(3):1130–1134. doi: 10.1128/jb.156.3.1130-1134.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorne K. J., Barker D. C. Bactoprenol, ATPase and acetate activating enzymes of a vesicular fraction from Lactobacillus casei. Eur J Biochem. 1969 Dec;11(3):582–591. doi: 10.1111/j.1432-1033.1969.tb00810.x. [DOI] [PubMed] [Google Scholar]
- Tobian J. A., Macrina F. L. Helper plasmid cloning in Streptococcus sanguis: cloning of a tetracycline resistance determinant from the Streptococcus mutans chromosome. J Bacteriol. 1982 Oct;152(1):215–222. doi: 10.1128/jb.152.1.215-222.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomochika K., Funabashi M., Nagata A., Fujii T., Kanemasa Y. [Protoplast formation of Lactobacillus casei induced by endo-N-acetylmuramidase]. Nihon Saikingaku Zasshi. 1982 Jul;37(4):777–785. [PubMed] [Google Scholar]
- Yokogawa K., Kawata S., Nishimura S., Ikeda Y., Yoshimura Y. Mutanolysin, bacteriolytic agent for cariogenic Streptococci: partial purification and properties. Antimicrob Agents Chemother. 1974 Aug;6(2):156–165. doi: 10.1128/aac.6.2.156. [DOI] [PMC free article] [PubMed] [Google Scholar]