Abstract
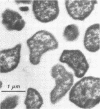
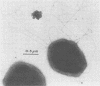
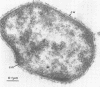
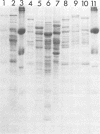
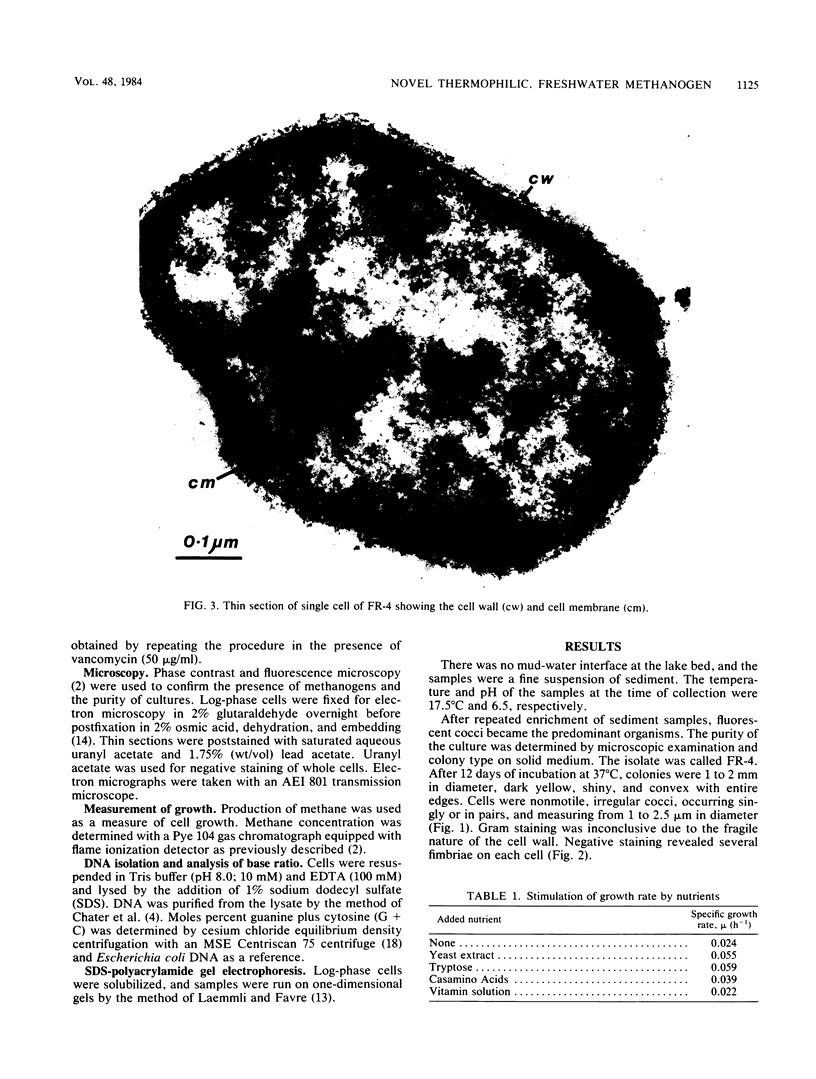
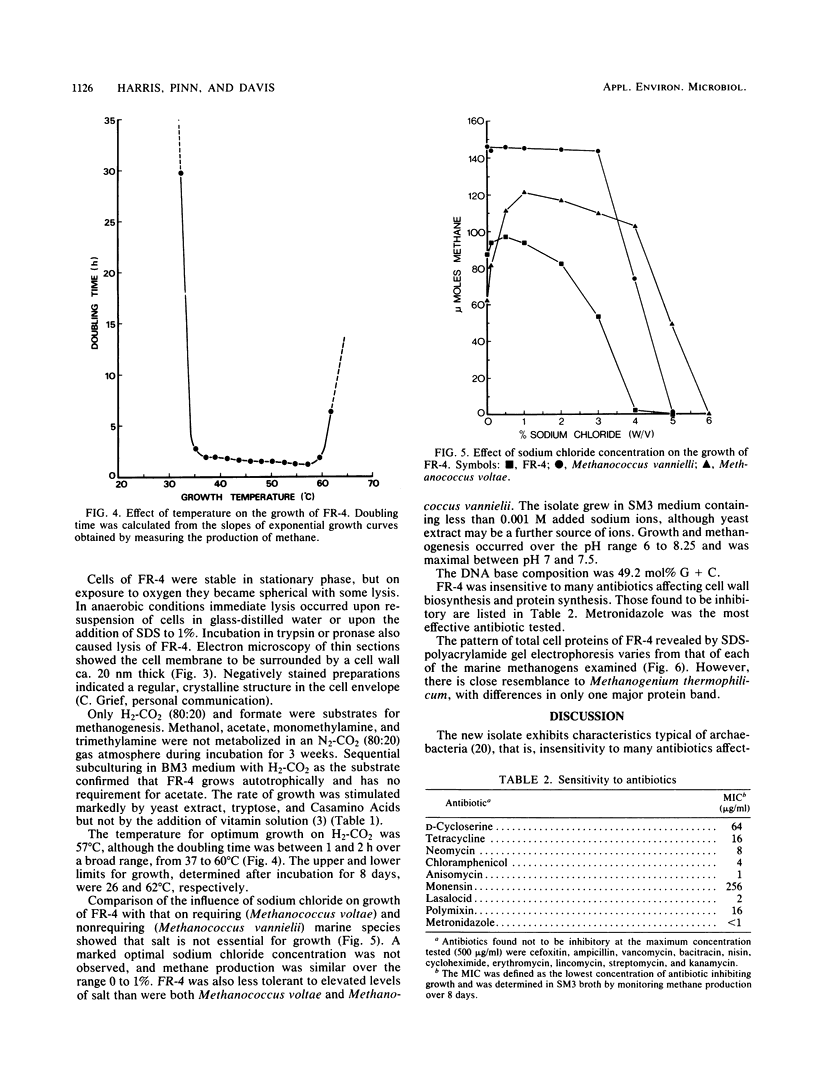
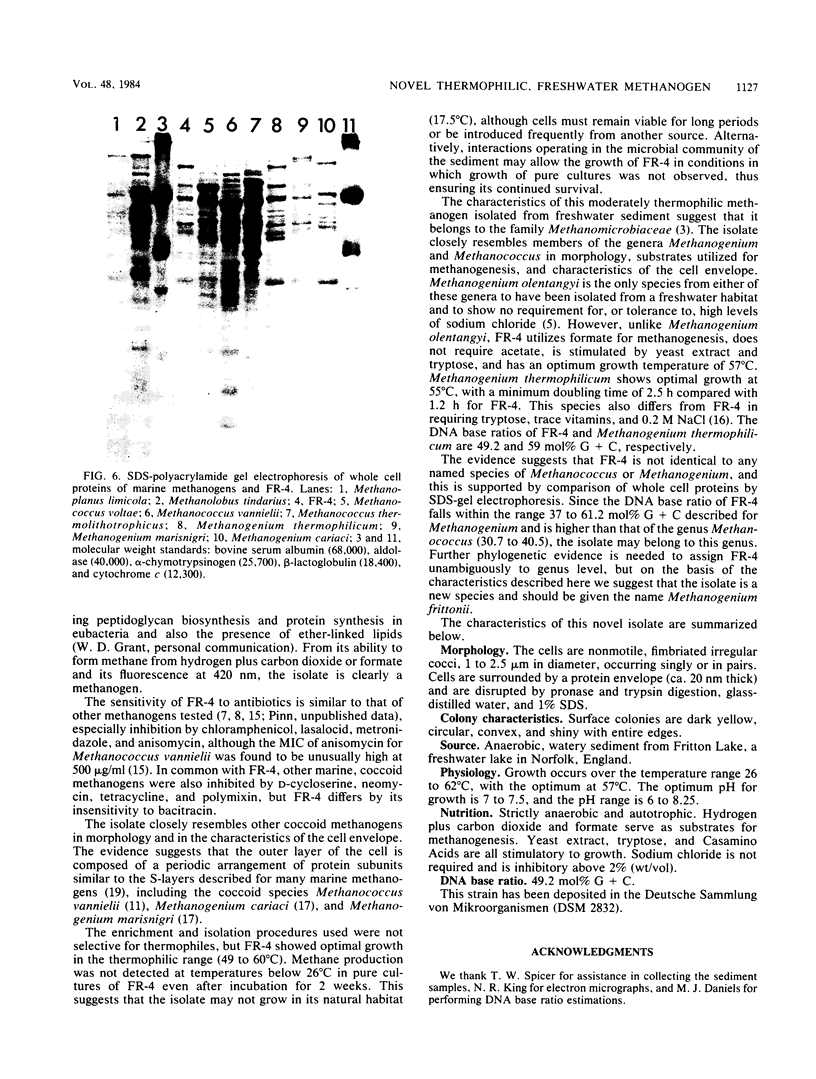
A novel thermophilic, coccoid methanogen isolated from nonthermal freshwater sediments is described. Hydrogen plus carbon dioxide and formate were substrates for methanogenesis, and methane production was stimulated by yeast extract, Casamino Acids, and tryptose. Growth also occurred autotrophically. Elevated levels of sodium chloride were not required for maximum growth and were inhibitory above 2%. The minimum doubling time occurred at 57°C, and the upper and lower limits for methane production were 62 and 26°C, respectively. The optimum pH for growth was between 7.0 and 7.5. Inhibitory antibiotics included metronidazole, anisomycin, chloramphenicol, and lasalocid. Colonies were circular, dark yellow, shiny, and convex with entire edges. Cells were 1 to 2.5 μm in diameter, nonmotile, occurring singly or in pairs, and fimbriated. Cells were lysed by pronase or trypsin digestion, glass-distilled water, and 1% sodium dodecyl sulfate. Electron micrographs of thin sections showed a monolayered cell wall ca. 20 nm thick. The DNA base ratio was 49.2 mol% guanine plus cytosine. The whole cell protein pattern differed from that of other named coccoid methanogens.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Balch W. E., Fox G. E., Magrum L. J., Woese C. R., Wolfe R. S. Methanogens: reevaluation of a unique biological group. Microbiol Rev. 1979 Jun;43(2):260–296. doi: 10.1128/mr.43.2.260-296.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chater K. F., Hopwood D. A., Kieser T., Thompson C. J. Gene cloning in Streptomyces. Curr Top Microbiol Immunol. 1982;96:69–95. doi: 10.1007/978-3-642-68315-2_5. [DOI] [PubMed] [Google Scholar]
- Ferguson T. J., Mah R. A. Isolation and characterization of an h(2)-oxidizing thermophilic methanogen. Appl Environ Microbiol. 1983 Jan;45(1):265–274. doi: 10.1128/aem.45.1.265-274.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones J. B., Bowers B., Stadtman T. C. Methanococcus vannielii: ultrastructure and sensitivity to detergents and antibiotics. J Bacteriol. 1977 Jun;130(3):1357–1363. doi: 10.1128/jb.130.3.1357-1363.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K., Favre M. Maturation of the head of bacteriophage T4. I. DNA packaging events. J Mol Biol. 1973 Nov 15;80(4):575–599. doi: 10.1016/0022-2836(73)90198-8. [DOI] [PubMed] [Google Scholar]
- SCHILDKRAUT C. L., MARMUR J., DOTY P. Determination of the base composition of deoxyribonucleic acid from its buoyant density in CsCl. J Mol Biol. 1962 Jun;4:430–443. doi: 10.1016/s0022-2836(62)80100-4. [DOI] [PubMed] [Google Scholar]
- Sleytr U. B., Messner P. Crystalline surface layers on bacteria. Annu Rev Microbiol. 1983;37:311–339. doi: 10.1146/annurev.mi.37.100183.001523. [DOI] [PubMed] [Google Scholar]
- Woese C. R., Magrum L. J., Fox G. E. Archaebacteria. J Mol Evol. 1978 Aug 2;11(3):245–251. doi: 10.1007/BF01734485. [DOI] [PubMed] [Google Scholar]