Abstract
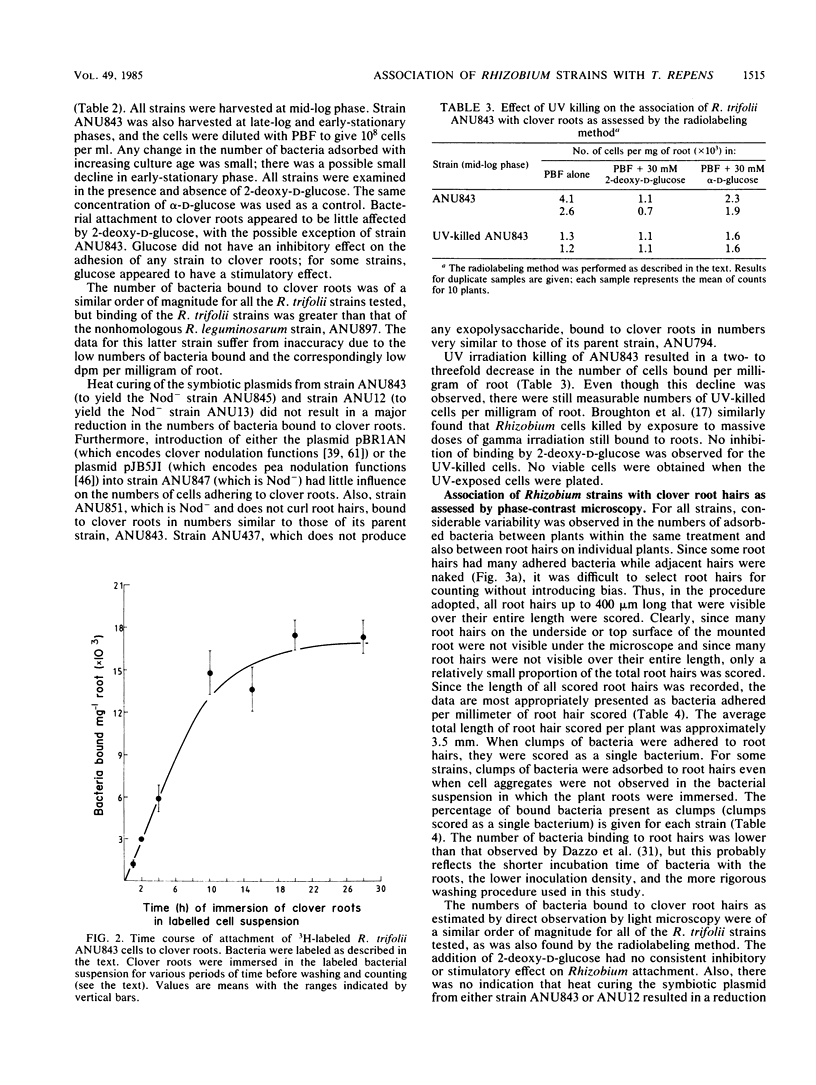
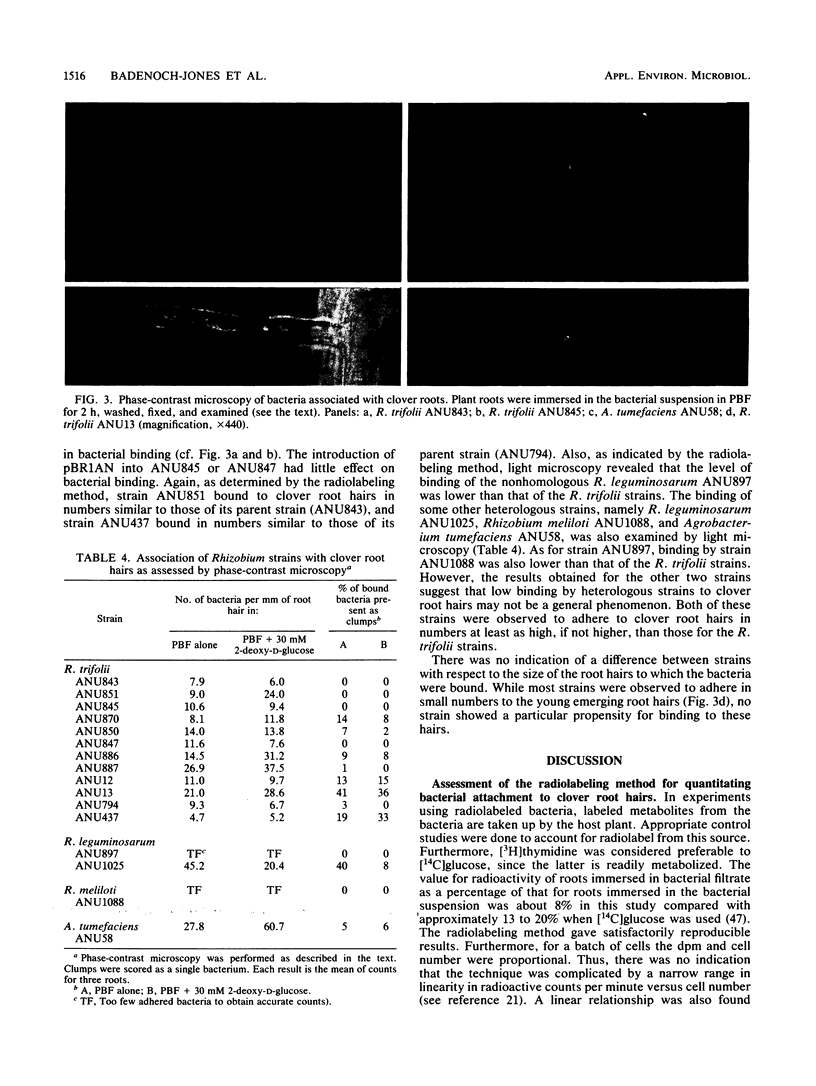
Two techniques were used to assess the binding of rhizobia to clover roots: indirect counting after radiolabeling the bacteria and direct counting by using phase-contrast microscopy. Microscopic observations revealed a large variability in the number of bacteria associated with individual root hairs. This variability made unbiased counting by microscopy difficult. Systematic examination of all visible root hairs and “blind” counting of coded strains and treatments were adopted to minimize observer bias. The validity of the radiolabeling method was also examined in some detail. The reproducibility of results from this method was satisfactory. However, drawbacks of this method included its lack of sensitivity and its failure to distinguish between bacteria attached to mature root hairs, emerging root hairs, and undifferentiated epidermal cells. The method also failed to distinguish between individual bacteria and any aggregates that may be present. The ability of a number of chosen mutant strains of Rhizobium trifolii and their corresponding parent strains, as well as a number of nonhomologous strains, to bind to clover roots was assessed by using both of these methods. Our results gave no indication of specificity of R. trifolii binding to clover roots. 2-Deoxy-d-glucose did not appear to have a major inhibitory effect on the attachment of rhizobia to the host root, which suggests that lectin cross-bridging is not an obligatory step in the initiation of infection even though it may occur under some conditions. The presence or absence of the symbiotic plasmid was not correlated with bacterial adherence to the host plant root. Since host specificity functions are carried on this plasmid, our results suggest that binding of rhizobia to the legume root is not the basis of host specificity.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Badenoch-Jones J., Summons R. E., Djordjevic M. A., Shine J., Letham D. S., Rolfe B. G. Mass spectrometric quantification of indole-3-acetic Acid in Rhizobium culture supernatants: relation to root hair curling and nodule initiation. Appl Environ Microbiol. 1982 Aug;44(2):275–280. doi: 10.1128/aem.44.2.275-280.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bal A. K., Shantharam S., Ratnam S. Ultrastructure of Rhizobium japonicum in relation to its attachment to root hairs. J Bacteriol. 1978 Mar;133(3):1393–1400. doi: 10.1128/jb.133.3.1393-1400.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bauer W. D. Lectins as determinants of specificity in legume-Rhizobium symbiosis. Basic Life Sci. 1977;9:283–297. doi: 10.1007/978-1-4684-0880-5_18. [DOI] [PubMed] [Google Scholar]
- Beringer J. E., Brewin N., Johnston A. W., Schulman H. M., Hopwood D. A. The Rhizobium--legume symbiosis. Proc R Soc Lond B Biol Sci. 1979 Apr 11;204(1155):219–233. doi: 10.1098/rspb.1979.0024. [DOI] [PubMed] [Google Scholar]
- Bhuvaneswari T. V., Bauer W. D. Role of Lectins in Plant-Microorganism Interactions: III. Influence of Rhizosphere/Rhizoplane Culture Conditions on the Soybean Lectin-binding Properties of Rhizobia. Plant Physiol. 1978 Jul;62(1):71–74. doi: 10.1104/pp.62.1.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhuvaneswari T. V., Bhagwat A. A., Bauer W. D. Transient susceptibility of root cells in four common legumes to nodulation by rhizobia. Plant Physiol. 1981 Nov;68(5):1144–1149. doi: 10.1104/pp.68.5.1144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhuvaneswari T. V., Mills K. K., Crist D. K., Evans W. R., Bauer W. D. Effects of culture age on symbiotic infectivity of Rhizobium japonicum. J Bacteriol. 1983 Jan;153(1):443–451. doi: 10.1128/jb.153.1.443-451.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhuvaneswari T. V., Pueppke S. G., Bauer W. D. Role of lectins in plant-microorganism interactions: I. Binding of soybean lectin to rhizobia. Plant Physiol. 1977 Oct;60(4):486–491. doi: 10.1104/pp.60.4.486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhuvaneswari T. V., Turgeon B. G., Bauer W. D. Early Events in the Infection of Soybean (Glycine max L. Merr) by Rhizobium japonicum: I. LOCALIZATION OF INFECTIBLE ROOT CELLS. Plant Physiol. 1980 Dec;66(6):1027–1031. doi: 10.1104/pp.66.6.1027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohlool B. B., Schmidt E. L. Immunofluorescent polar tips of Rhizobium japonicum: possible site of attachment or lectin binding. J Bacteriol. 1976 Mar;125(3):1188–1194. doi: 10.1128/jb.125.3.1188-1194.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohlool B. B., Schmidt E. L. Lectins: a possible basis for specificity in the Rhizobium--legume root nodule symbiosis. Science. 1974 Jul 19;185(4147):269–271. doi: 10.1126/science.185.4147.269. [DOI] [PubMed] [Google Scholar]
- Broughton W. J., van Egeraat A. W., Lie T. A. Dynamics of Rhizobium competition for nodulation of Pisum sativum cv. Afghanistan. Can J Microbiol. 1980 Apr;26(4):562–565. doi: 10.1139/m80-099. [DOI] [PubMed] [Google Scholar]
- Chakravorty A. K., Zurkowski W., Shine J., Rolfe B. G. Symbiotic nitrogen fixation: molecular cloning of Rhizobium genes involved in exopolysaccharide synthesis and effective nodulation. J Mol Appl Genet. 1982;1(6):585–596. [PubMed] [Google Scholar]
- Dazzo F. B. Bacterial attachment as related to cellular recognition in the Rhizobium-legume symbiosis. J Supramol Struct Cell Biochem. 1981;16(1):29–41. doi: 10.1002/jsscb.1981.380160104. [DOI] [PubMed] [Google Scholar]
- Dazzo F. B., Brill W. J. Bacterial polysaccharide which binds Rhizobium trifolii to clover root hairs. J Bacteriol. 1979 Mar;137(3):1362–1373. doi: 10.1128/jb.137.3.1362-1373.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dazzo F. B., Brill W. J. Receptor site on clover and alfalfa roots for Rhizobium. Appl Environ Microbiol. 1977 Jan;33(1):132–136. doi: 10.1128/aem.33.1.132-136.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dazzo F. B., Brill W. J. Regulation by fixed nitrogen of host-symbiont recognition in the Rhizobium-clover symbiosis. Plant Physiol. 1978 Jul;62(1):18–21. doi: 10.1104/pp.62.1.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dazzo F. B., Hrabak E. M. Presence of trifoliin A, a Rhizobium-binding lectin, in clover root exudate. J Supramol Struct Cell Biochem. 1981;16(2):133–138. doi: 10.1002/jsscb.1981.380160204. [DOI] [PubMed] [Google Scholar]
- Dazzo F. B., Hubbell D. H. Cross-reactive antigens and lectin as determinants of symbiotic specificity in the Rhizobium-clover association. Appl Microbiol. 1975 Dec;30(6):1017–1033. doi: 10.1128/am.30.6.1017-1033.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dazzo F. B., Napoli C. A., Hubbell D. H. Adsorption of bacteria to roots as related to host specificity in the Rhizobium-clover symbiosis. Appl Environ Microbiol. 1976 Jul;32(1):166–171. doi: 10.1128/aem.32.1.166-171.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dazzo F. B., Truchet G. L., Sherwood J. E., Hrabak E. M., Abe M., Pankratz S. H. Specific phases of root hair attachment in the Rhizobium trifolii-clover symbiosis. Appl Environ Microbiol. 1984 Dec;48(6):1140–1150. doi: 10.1128/aem.48.6.1140-1150.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dazzo F. B., Truchet G. L., Sherwood J. E., Hrabak E. M., Gardiol A. E. Alteration of the Trifoliin A-Binding Capsule of Rhizobium trifolii 0403 by Enzymes Released from Clover Roots. Appl Environ Microbiol. 1982 Aug;44(2):478–490. doi: 10.1128/aem.44.2.478-490.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dazzo F. B., Yanke W. E., Brill W. J. Trifolin: a Rhizobium recognition protein from white clover. Biochim Biophys Acta. 1978 Mar 20;539(3):276–286. doi: 10.1016/0304-4165(78)90032-6. [DOI] [PubMed] [Google Scholar]
- Dhaese P., De Greve H., Decraemer H., Schell J., Van Montagu M. Rapid mapping of transposon insertion and deletion mutations in the large Ti-plasmids of Agrobacterium tumefaciens. Nucleic Acids Res. 1979 Dec 11;7(7):1837–1849. doi: 10.1093/nar/7.7.1837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Djordjevic M. A., Zurkowski W., Shine J., Rolfe B. G. Sym plasmid transfer to various symbiotic mutants of Rhizobium trifolii, R. leguminosarum, and R. meliloti. J Bacteriol. 1983 Dec;156(3):1035–1045. doi: 10.1128/jb.156.3.1035-1045.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hrabak E. M., Urbano M. R., Dazzo F. B. Growth-phase-dependent immunodeterminants of Rhizobium trifolii lipopolysaccharide which bind trifoliin A, a white clover lectin. J Bacteriol. 1981 Nov;148(2):697–711. doi: 10.1128/jb.148.2.697-711.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li D., Hubbell D. H. Infection thread formation as a basis of nodulation specificity in Rhizobium--strawberry clover associations. Can J Microbiol. 1969 Oct;15(10):1133–1136. doi: 10.1139/m69-206. [DOI] [PubMed] [Google Scholar]
- Marshall K. C., Cruickshank R. H., Bushby H. V. The orientation of certain root-nodule bacteria at interfaces, including legume root-hair surfaces. J Gen Microbiol. 1975 Nov;91(1):198–200. doi: 10.1099/00221287-91-1-198. [DOI] [PubMed] [Google Scholar]
- Menzel G., Uhlig H., Weichsel G. Uber die Besiedlung der Wurzeln einiger Leguminosen und Nichtleguminosen mit Rhizobien und anderen Bodenbakterien. Zentralbl Bakteriol Parasitenkd Infektionskr Hyg. 1972;127(4):348–358. [PubMed] [Google Scholar]
- Napoli C. A., Hubbell D. H. Ultrastructure of Rhizobium-induced infection threads in clover root hairs. Appl Microbiol. 1975 Dec;30(6):1003–1009. doi: 10.1128/am.30.6.1003-1009.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pueppke S. G. Adsorption of slow- and fast-growing rhizobia to soybean and cowpea roots. Plant Physiol. 1984 Aug;75(4):924–928. doi: 10.1104/pp.75.4.924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pueppke S. G., Bauer W. D. Role of Lectins in Plant-Microorganism Interactions: II. Distribution of Soybean Lectin in Tissues of Glycine max (L.) Merr. Plant Physiol. 1978 May;61(5):779–784. doi: 10.1104/pp.61.5.779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolfe B. G., Gresshoff P. M., Shine J., Vincent J. M. Interaction Between a Non-Nodulating and an Ineffective Mutant of Rhizobium trifolli Resulting in Effective (Nitrogen-Fixing) Nodulation. Appl Environ Microbiol. 1980 Feb;39(2):449–452. doi: 10.1128/aem.39.2.449-452.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt E. L. Initiation of plant root-microbe interactions. Annu Rev Microbiol. 1979;33:355–376. doi: 10.1146/annurev.mi.33.100179.002035. [DOI] [PubMed] [Google Scholar]
- Sherwood J. E., Vasse J. M., Dazzo F. B., Truchet G. L. Development and trifoliin A-binding ability of the capsule of Rhizobium trifolii. J Bacteriol. 1984 Jul;159(1):145–152. doi: 10.1128/jb.159.1.145-152.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimshick E. J., Hebert R. R. Adsorption of rhizobia to cereal roots. Biochem Biophys Res Commun. 1978 Oct 16;84(3):736–742. doi: 10.1016/0006-291x(78)90766-0. [DOI] [PubMed] [Google Scholar]
- Stacey G., Paau A. S., Brill W. J. Host recognition in the Rhizobium-soybean symbiosis. Plant Physiol. 1980 Oct;66(4):609–614. doi: 10.1104/pp.66.4.609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szabó B., Tóth F. D., Váczi L., Réthy A., Kiss A., Rák K. Cytotoxicity of lymphocytes and antibodies against autologous tumor cells in patients with myeloid leukemias and preleukemic disorders. I. Blocking activity of gp70 antigens of primate type C viruses. Neoplasma. 1983;30(2):129–135. [PubMed] [Google Scholar]
- Werner D., Wilcockson J., Zimmermann E. Adsorption and selection of rhizobia with ion-exchange papers. Arch Microbiol. 1975 Sep 30;105(1):27–32. doi: 10.1007/BF00447108. [DOI] [PubMed] [Google Scholar]
- Wong P. P. Interactions between Rhizobia and Lectins of Lentil, Pea, Broad Bean, and Jackbean. Plant Physiol. 1980 Jun;65(6):1049–1052. doi: 10.1104/pp.65.6.1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zurkowski W. Specific adsorption of bacteria to clover root hairs, related to the presence of the plasmid pWZ2 in cells of Rhizobium trifolii. Microbios. 1980;27(107):27–32. [PubMed] [Google Scholar]
- van Rensburg H. J., Strijdom B. W. Root Surface Association in Relation to Nodulation of Medicago sativa. Appl Environ Microbiol. 1982 Jul;44(1):93–97. doi: 10.1128/aem.44.1.93-97.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]



