Abstract
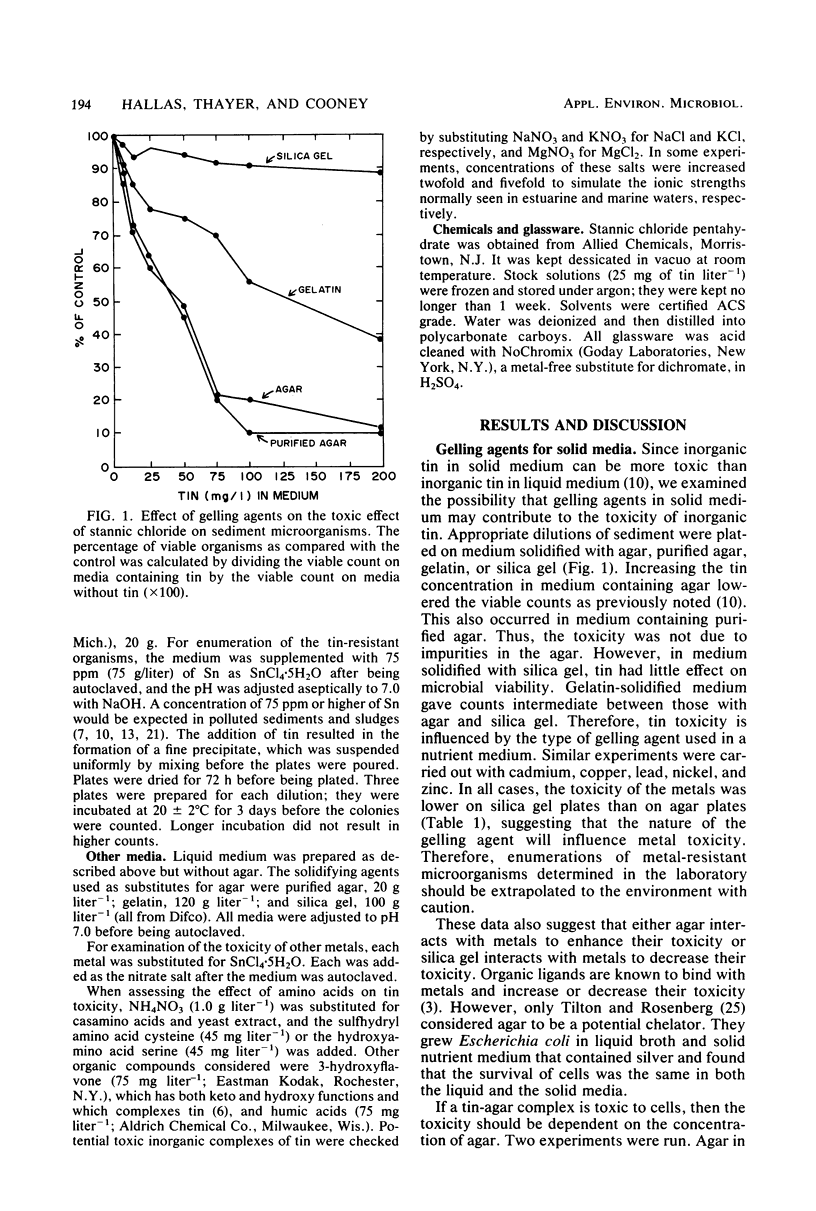
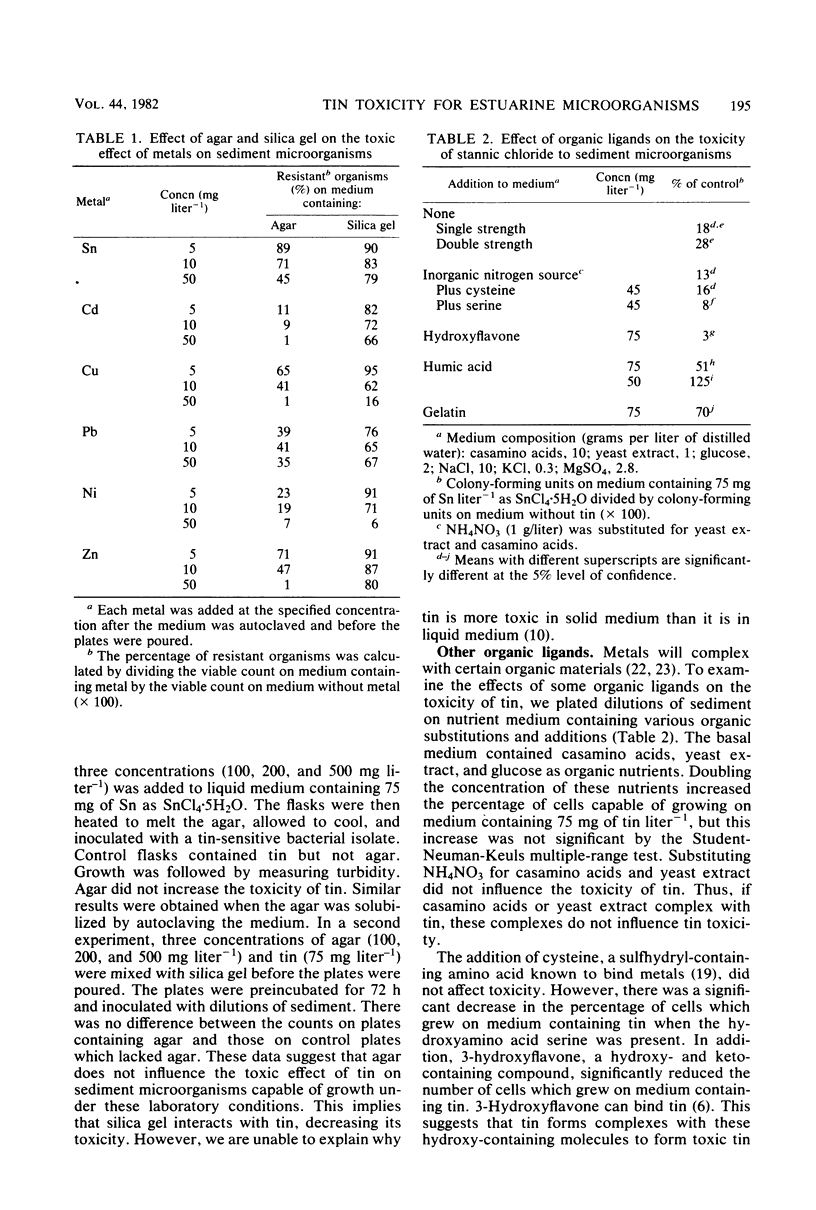
Inorganic tin (SnCl4·H2O) is toxic to microbial populations obtained from estuarine sediments plated on nutrient medium solidified with either agar or purified agar. The use of gelatin as a gelling agent decreased the apparent toxicity of tin, and toxicity was markedly reduced in medium solidified with silica gel. There was no evidence that toxic agar-tin complexes were involved. Cd, Cu, Pb, Ni, and Zn exhibited similar toxicity patterns; therefore, toxicity levels determined in the laboratory should be extrapolated to the environment with caution. The addition of cysteine to the medium had no effect on tin toxicity. Serine or 3-hydroxyflavone enhanced toxicity, while humic acids or gelatin inhibited toxicity. Replacement of SO42− with NO3− did not alter tin toxicity, but replacement of Cl− with NO3− decreased tin toxicity. Thus, the toxic effect(s) of tin depend as much on the chemical speciation of the metal as on the total concentration of the metal in the medium.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aldridge W. N., Street B. W., Skilleter D. N. Oxidative phosphorylation. Halide-dependent and halide-independent effects of triorganotin and trioganolead compounds on mitochondrial functions. Biochem J. 1977 Dec 15;168(3):353–364. doi: 10.1042/bj1680353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babich H., Stotzky G. Abiotic factors affecting the toxicity of lead to fungi. Appl Environ Microbiol. 1979 Sep;38(3):506–513. doi: 10.1128/aem.38.3.506-513.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babich H., Stotzky G. Differential toxicities of mercury to bacteria and bacteriophages in sea and in lake water. Can J Microbiol. 1979 Nov;25(11):1252–1257. doi: 10.1139/m79-197. [DOI] [PubMed] [Google Scholar]
- Babich H., Stotzky G. Environmental factors that influence the toxicity of heavy metal and gaseous pollutants to microorganisms. Crit Rev Microbiol. 1980;8(2):99–145. doi: 10.3109/10408418009081123. [DOI] [PubMed] [Google Scholar]
- Hallas L. E., Cooney J. J. Tin and tin-resistant microorganisms in chesapeake bay. Appl Environ Microbiol. 1981 Feb;41(2):466–471. doi: 10.1128/aem.41.2.466-471.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamdy M. K., Wheeler S. R. Inhibition of Bacterial growth by mercury and the effects of protective agents. Bull Environ Contam Toxicol. 1978 Sep;20(3):378–386. doi: 10.1007/BF01683535. [DOI] [PubMed] [Google Scholar]
- Jernelöv A., Martin A. L. Ecological implications of metal metabolism by microorganisms. Annu Rev Microbiol. 1975;29:61–77. doi: 10.1146/annurev.mi.29.100175.000425. [DOI] [PubMed] [Google Scholar]
- Lighthart B. Effects of certain cadmium species on pure and litter populations of microorganisms. Antonie Van Leeuwenhoek. 1980;46(2):161–167. doi: 10.1007/BF00444071. [DOI] [PubMed] [Google Scholar]
- Mills A. L., Colwell R. R. Microbiological effects of metal ions in Chesapeake Bay water and sediment. Bull Environ Contam Toxicol. 1977 Jul;18(1):99–103. doi: 10.1007/BF01686313. [DOI] [PubMed] [Google Scholar]
- Tilton R. C., Rosenberg B. Reversal of the silver inhibition of microorganisms by agar. Appl Environ Microbiol. 1978 Jun;35(6):1116–1120. doi: 10.1128/aem.35.6.1116-1120.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walter R. G., Windeler A. S. Solubilization of interfering tin precipitate in bacterial growth media. J Dent Res. 1976 Mar-Apr;55(2):311–311. doi: 10.1177/00220345760550022701. [DOI] [PubMed] [Google Scholar]


