Abstract
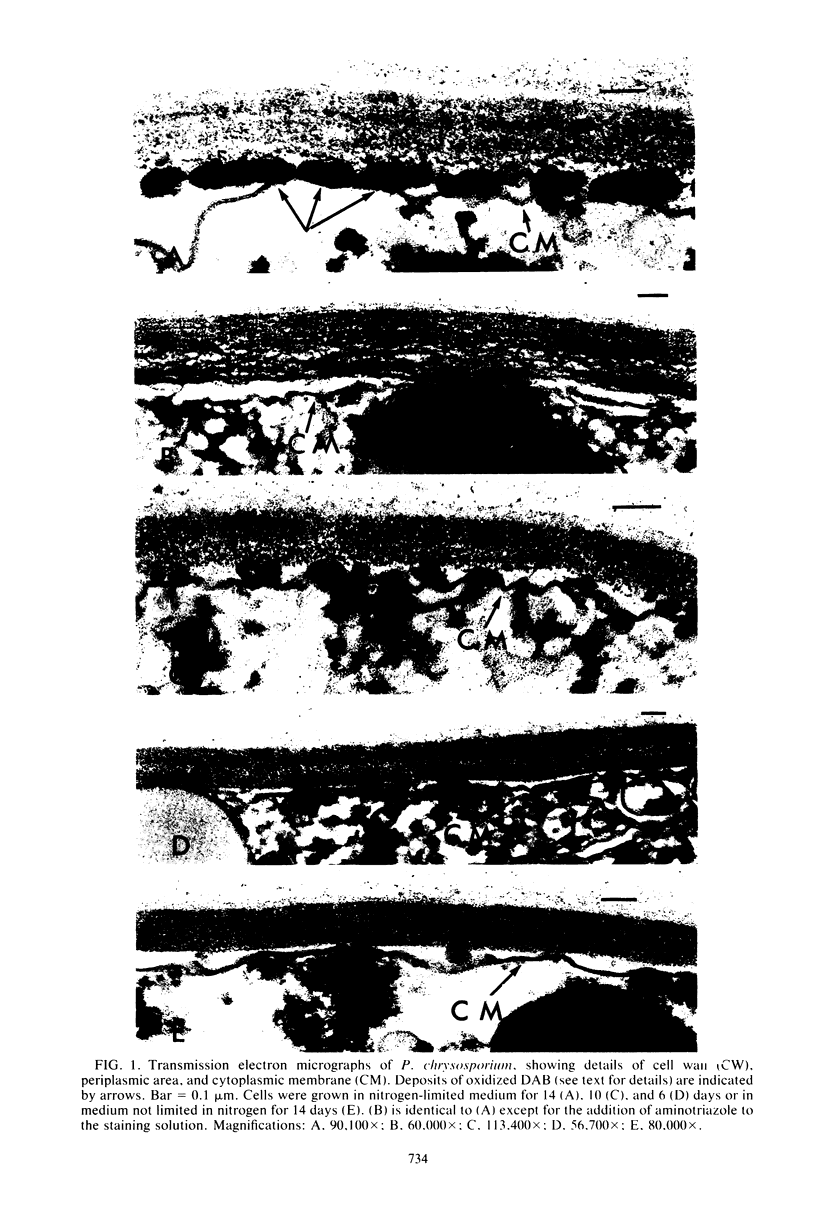
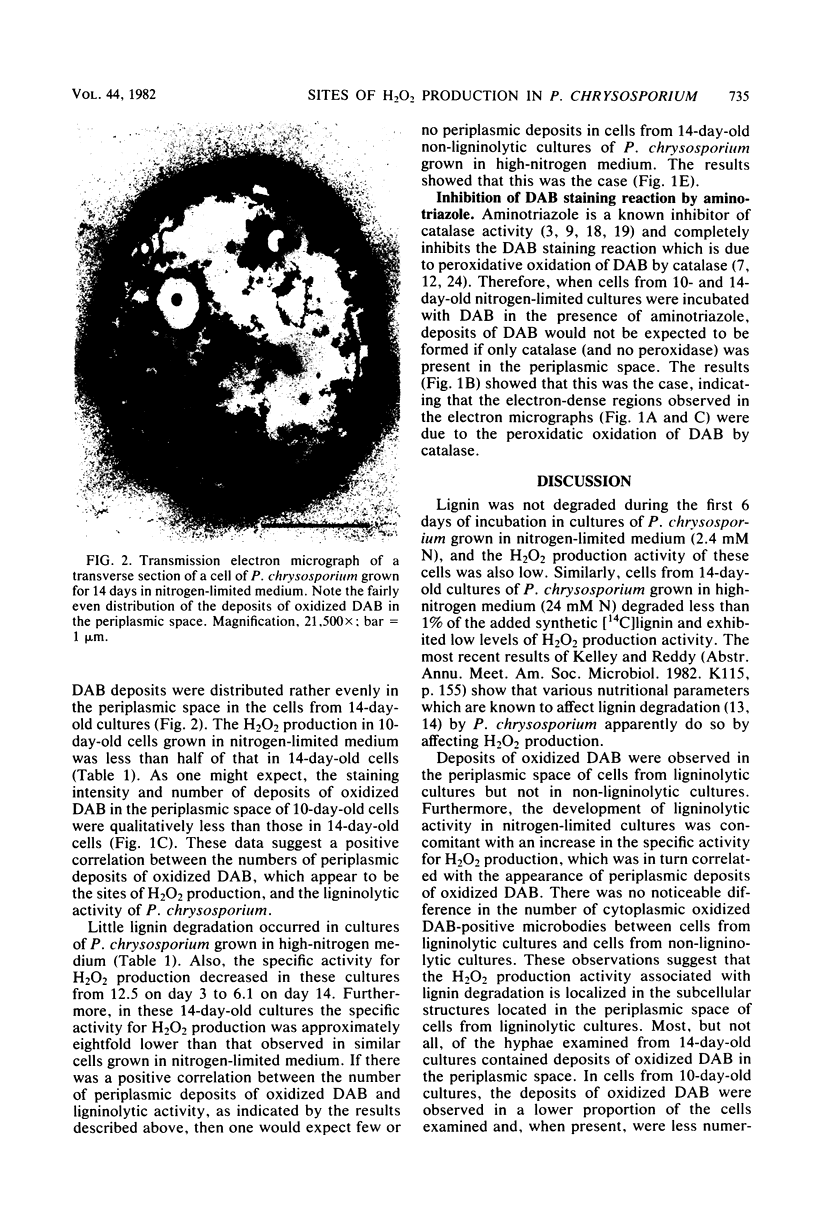
Previous studies have shown that the hydroxyl radical derived from hydrogen peroxide (H2O2) is involved in lignin degradation by Phanerochaete chrysosporium. In the present study, the ultrastructural sites of H2O2 production in ligninolytic cells of P. chrysosporium were demonstrated by cytochemically staining cells with 3,3′-diaminobenzidine (DAB). Hydrogen peroxide production, as evidenced by the presence of oxidized DAB deposits, appeared to be localized in the periplasmic space of cells from ligninolytic cultures grown for 14 days in nitrogen-limited medium. When identical cells were treated with DAB in the presence of aminotriazole, periplasmic deposits of oxidized DAB were not observed, suggesting that the deposits resulted from the H2O2-dependent peroxidatic oxidation of DAB by catalase. Cells from cultures grown for 3 or 6 days in nitrogen-limited medium or for 14 days in nitrogen-sufficient medium had little ligninolytic activity and low specific activity for H2O2 production and did not contain periplasmic oxidized DAB deposits. The results suggest that in cultures grown in nitrogen-limited medium, there is a positive correlation between the occurrence of oxidized DAB deposits, the specific activity for H2O2 production in cell extracts, and ligninolytic activity.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- APPLEMAN D., HEIM W. G., PYFROM H. T. Effects of 3-amino-1, 2, 4-triazole (AT) on catalase and other compounds. Am J Physiol. 1956 Jul;186(1):19–23. doi: 10.1152/ajplegacy.1956.186.1.19. [DOI] [PubMed] [Google Scholar]
- Brawn K., Fridovich I. Superoxide radical and superoxide dismutases: threat and defense. Acta Physiol Scand Suppl. 1980;492:9–18. [PubMed] [Google Scholar]
- COHEN G., HOCHSTEIN P. GENERATION OF HYDROGEN PEROXIDE IN ERYTHROCYTES BY HEMOLYTIC AGENTS. Biochemistry. 1964 Jul;3:895–900. doi: 10.1021/bi00895a006. [DOI] [PubMed] [Google Scholar]
- Cohen G., Somerson N. L. Catalase-aminotriazole method for measuring secretion of hydrogen peroxide by microorganisms. J Bacteriol. 1969 May;98(2):543–546. doi: 10.1128/jb.98.2.543-546.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FARMER V. C., HENDERSON M. E., RUSSELL J. D. Aromatic-alcohol-oxidase activity in the growth medium of Polystictus versicolor. Biochem J. 1960 Feb;74:257–262. doi: 10.1042/bj0740257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fong K. L., McCay P. B., Poyer J. L., Keele B. B., Misra H. Evidence that peroxidation of lysosomal membranes is initiated by hydroxyl free radicals produced during flavin enzyme activity. J Biol Chem. 1973 Nov 25;248(22):7792–7797. [PubMed] [Google Scholar]
- Frederick S. E., Newcomb E. H. Cytochemical localization of catalase in leaf microbodies (peroxisomes). J Cell Biol. 1969 Nov;43(2):343–353. doi: 10.1083/jcb.43.2.343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herzog V., Fahimi H. D. The effect of glutaraldehyde on catalase. Biochemical and cytochemical studies with beef liver catalase and rat liver peroxisomes. J Cell Biol. 1974 Jan;60(1):303–311. doi: 10.1083/jcb.60.1.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keyser P., Kirk T. K., Zeikus J. G. Ligninolytic enzyme system of Phanaerochaete chrysosporium: synthesized in the absence of lignin in response to nitrogen starvation. J Bacteriol. 1978 Sep;135(3):790–797. doi: 10.1128/jb.135.3.790-797.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kong S., Davison A. J. The role of interactions between O2, H2O2, .OH,e- and O2- in free radical damage to biological systems. Arch Biochem Biophys. 1980 Oct 1;204(1):18–29. doi: 10.1016/0003-9861(80)90003-x. [DOI] [PubMed] [Google Scholar]
- MARGOLIASH E., NOVOGRODSKY A. A study of the inhibition of catalase by 3-amino-1:2:4:-triazole. Biochem J. 1958 Mar;68(3):468–475. doi: 10.1042/bj0680468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MARGOLIASH E., NOVOGRODSKY A., SCHEJTER A. Irreversible reaction of 3-amino-1:2:4-triazole and related inhibitors with the protein of catalase. Biochem J. 1960 Feb;74:339–348. doi: 10.1042/bj0740339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seligman A. M., Karnovsky M. J., Wasserkrug H. L., Hanker J. S. Nondroplet ultrastructural demonstration of cytochrome oxidase activity with a polymerizing osmiophilic reagent, diaminobenzidine (DAB). J Cell Biol. 1968 Jul;38(1):1–14. doi: 10.1083/jcb.38.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vigil E. L. Intracellular localization of catalase (peroxidatic) activity in plant microbodies. J Histochem Cytochem. 1969 Jun;17(6):425–428. doi: 10.1177/17.6.425. [DOI] [PubMed] [Google Scholar]





