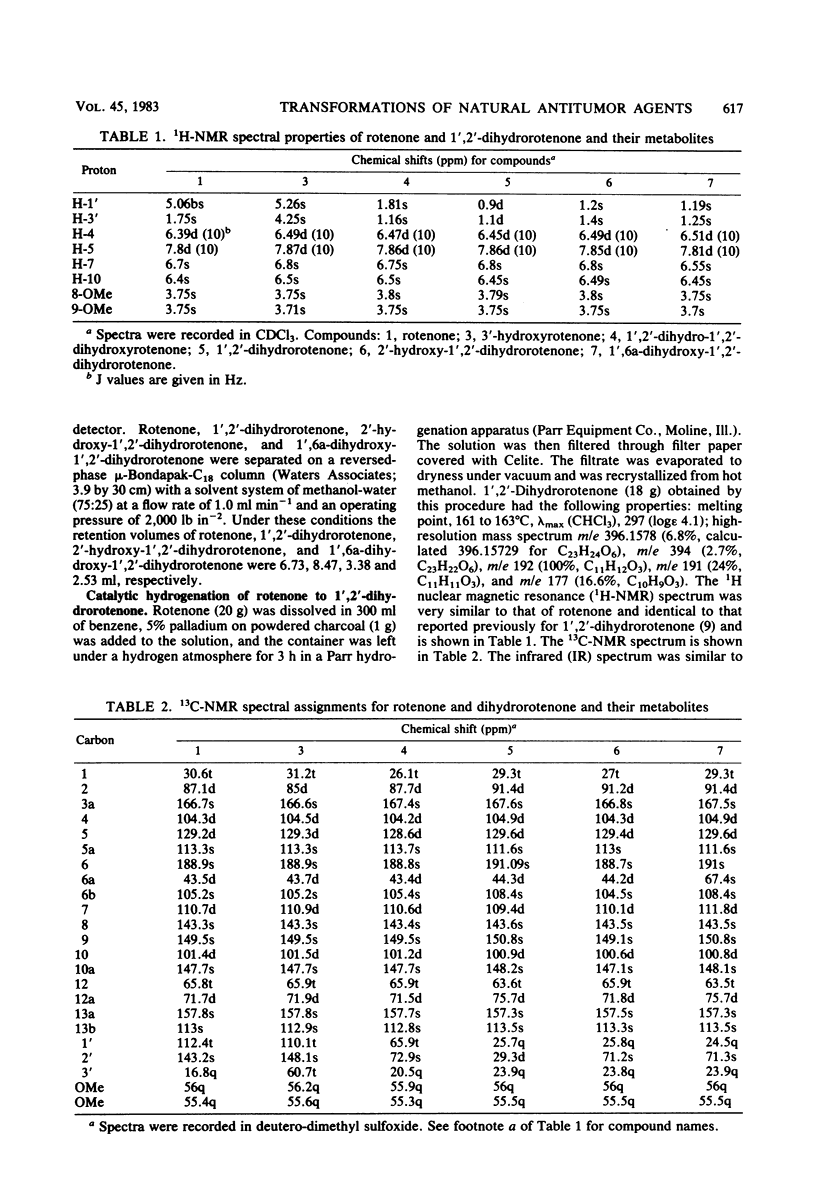
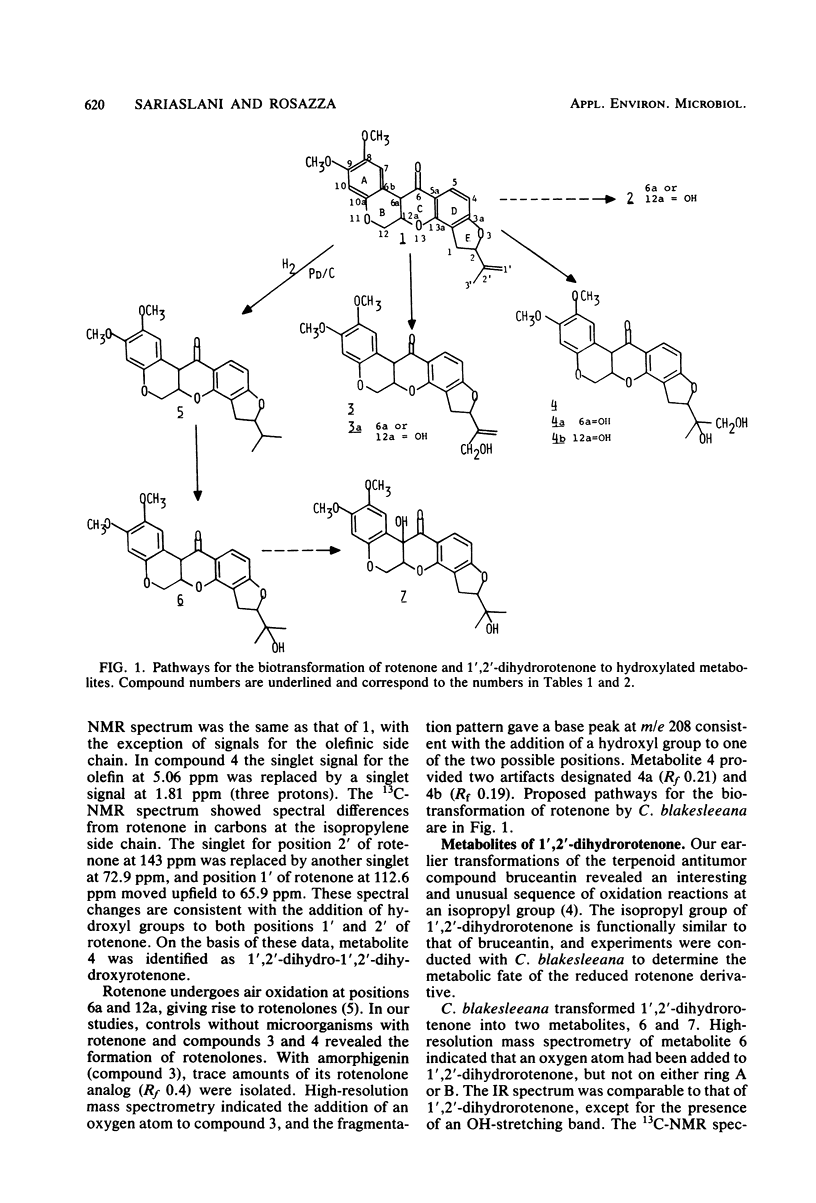
Abstract
Various species of Absidia, Aspergillus, Cunninghamella, Trichothecium, Penicillium, and Phanerochaete were found to transform rotenone to one or more metabolites. Two biotransformation products were isolated from a preparative-scale incubation of rotenone with Cunninghamella blakesleeana and identified as 1′,2′-dihydro-1′,2′-dihydroxyrotenone and 3′-hydroxyrotenone (amorphigenin). The catalytic reduction of the isopropylene side chain of rotenone resulted in the formation of 1′,2′-dihydrorotenone. The latter was transformed by C. blakesleeana to 2′-hydroxy-1′,2′-dihydrorotenone.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Betts R. E., Walters D. E., Rosazza J. P. Microbial transformations of antitumor compounds. 1. Conversion of acronycine to 9-hydroxyacronycine by Cunninghamella echinulata. J Med Chem. 1974 Jun;17(6):599–602. doi: 10.1021/jm00252a006. [DOI] [PubMed] [Google Scholar]
- CHANCE B., WILLIAMS G. R., HOLLUNGER G. Inhibition of electron and energy transfer in mitochondria. I. Effects of Amytal, thiopental, rotenone, progesterone, and methylene glycol. J Biol Chem. 1963 Jan;238:418–431. [PubMed] [Google Scholar]
- Haley T. J. A review of the literature of rotenone, 1,2,12,12a-tetrahydro-8,9-dimethoxy-2-(1-methylethenyl)-1-benzopyrano[3,5-b]furo[2,3-h][1]benzopyran-6(6h)-one. J Environ Pathol Toxicol. 1978 Jan-Feb;1(3):315–337. [PubMed] [Google Scholar]



