Abstract
Bone marrow (BM) was for many years primarily envisioned as the “home organ” of hematopoietic stem cells (HSC). Augmenting evidence demonstrates, however that BM in addition to HSC also contains a heterogeneous population of non-hematopoietic stem cells (non-HSC). Recently our group identified in BM and other adult tissues a population of very small embryonic like stem cells (VSELs) which express several markers characteristic for pluripotent stem cells (PSC) that are characteristic for epiblast/germ line- derived stem cells. Thus, we hypothesize that VSELs are a population of epiblast-derived cells that are deposited during early gastrulation in developing tissues/organs and play an important role in turnover of tissue specific/committed stem cells. In this context VSELs deposited in BM may give rise to long term repopulating HSC. VSELs could be also mobilized into peripheral blood (PB) and their number of these cells circulating in PB increases during stress and tissue/organ injuries. Finally, we envision that in pathological situations VSELs are involved in development of some malignancies (e.g., teratomas, germinal tumors).
Keywords: CXCR4, VSEL, Oct-4, Nanog, SSEA, embryonic stem cells
Introduction
Recent evidence indicates that BM in addition to well described hematopoietic stem cells (HSC) contains a heterogeneous population of non-hematopoietic stem cells (non-HSC) [1, 2]. Furthermore it was also postulated that this population of non-HSC in BM contain some very rare cell population that displays several features of pluripotent stem cells (PSC) [3, 4]. These putative PSC have been demonstrated to be able in vitro cultures to differentiate into cells from all three germ layers and were described in the literature as i) mesenchymal stem cells (MSC) [5–7], ii) multipotent adult progenitor cells (MAPC) [8], iii) marrow-isolated adult multilineage inducible (MIAMI) cells [9, 10], iv) multipotent adult stem cells (MASC) [11] and v) very small embryonic like stem cells (VSELs) [12, 13]. We envision the possibility that similar or overlapping populations of primitive stem cells were detected in BM using different experimental strategies and hence were assigned different names.
All of these mentioned above stem cells display pluripotentiality in vitro being able to differentiate into cells from all three germ layers, however, despite several attempts made so far, there is no convincing data proving that any type of these cells may contribute in vivo to the development of multiple organs and tissues when injected into the developing blastocyst. Thus, this important criterion for in vivo pluripotentiality is still missing for stem cells isolated from adult BM. There are several potential explanations for this obvious “deficiency” such as i) developmental lost of proper responsiveness to embryonic factors, ii) asynchronization/differences in cell cycle between cells isolated from adult tissues and cells in blastocyst, and iii) a possibility that these cells erase somatic imprint. It is accepted that proper somatic imprint - methylation of selected maternal- and paternal- inherited genes regulates in vivo pluripotentiality of stem cells [14, 15], and is required for PSC to be able to complete blastocyst development.
In this review paper we will focus on population of VSELs and discuss i) strategies that allow to purify these rare cells, ii) their hypothetical developmental origin and iii) physiological and pathological consequences of their presence in adult tissues.
Overview on the developmental hierarchy of the stem cell compartment – from the totipotent zygote to bone marrow-residing stem cells
Stem cells are endowed with the property of self-renewal and the ability to differentiate into cells that are committed to particular developmental pathways (Figure 1 panel A). The compartment of stem cells is organized in a hierarchical way from the most primitive (totipotent) to already differentiated tissue-committed (monopotent) stem cells. In this context HSC are an example of monopotent stem cells already committed to lympho/hematopoiesis.
Figure 1. Panel A. Developmental hierarchy of the stem cell compartment.

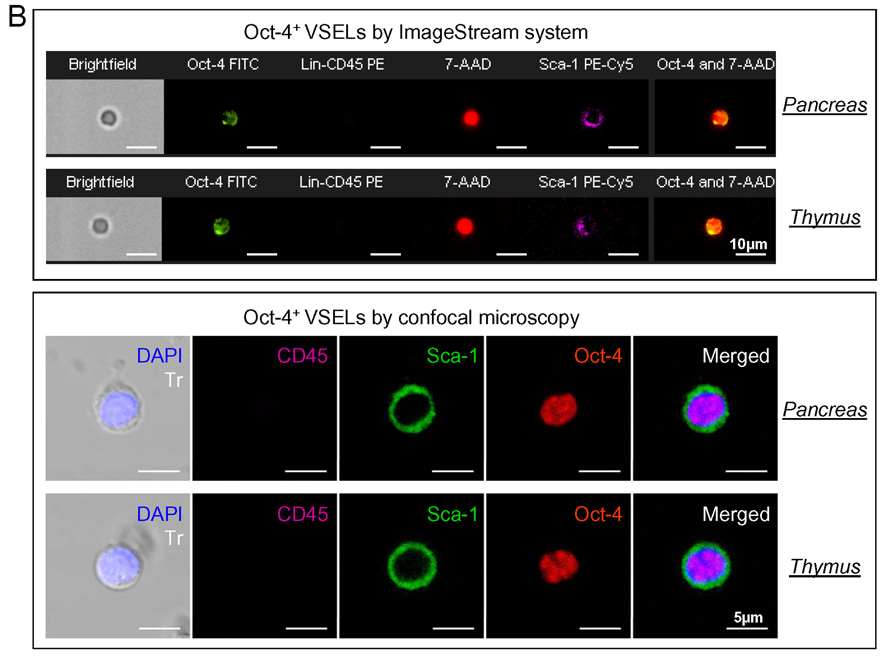
The most primitive stem cell is a totipotent zygote or first blastomers that derives from the first division of the zygote. A totipotent stem cell divides to form both embryo and placenta. Pluripotent stem cells (PSC) are i) cells isolated from the morula, ii) cells from the inner cell mass (ICM) of a blastocyst that give rise to established embryonic stem cell (ESC) lines or iii) embryonic germ cells (EG) that are in vitro derivatives of primordial germ cells (PGC). A potential source of pluripotent stem cells is also epiblast – a part of zylinder stage embryo from which develop all three germ layers. PSC contribute to all three germ layers in the developing embryo (ecto-, meso- and endoderm). Multipotent stem cells give rise to stem cells for particular germ layers (ecto-, meso-, and endoderm). Monopotent stem cells that are already committed to particular organs/tissues. In this context we hypothesize that VSELs are epiblast derived PSC that are deposited in developing organ/tissues as a population of stem cells that gives origin to monopotent tissue committed stem cells. Panel B. Representative images of Oct-4+ VSELs derived from adult murine organs by ImageStream system and confocal microscopy. Upper panel shows images of Sca-1+/Lin−/CD45−/Oct-4+ cells isolated from pancreas and thymus - analyzed by ImageStream system. Cells were fixed and stained in suspension for Oct-4 (FITC; green), CD45 and lineage markers (PE; orange) and Sca-1 antigen (PE-Cy5; magenta). Nuclei were stained with 7-aminoactinomycin D (7-AAD; red). Each photograph presents brightfield image, fluorescence images related to expression of indicated markers, as well as composed photo combining nuclear and Oct-4 images. The scale bar indicates 10µm. Lower panel shows confocal microscopic images of Oct-4+ VSELs derived from murine pancreas and thymus. Cells were fixed and stained for Sca-1 (FITC; green), Oct-4 (TRITC; red) and CD45 (Cy5; magenta). Nuclei were stained with DAPI (blue). Each photograph presents combined transmission (Tr) and nuclear (DAPI) images as well as separated and combined images related to the expression of CD45, Sca-1 and Oct-4 antigens. The scale bar indicates 5µm.
Figure 1 panel A depicts the developmental hierarchy of the stem cell compartment. It begins with the most primitive totipotent stem cell, the zygote, which is the result of the fusion of two germ cells (oocyte and sperm) during the process of fertilization. As a totipotent stem cell, the zygote is able to give rise to both the embryo and the placenta. When the blastomers have divided into the 32-cell stage the embryo is called morula. Cells which form the morula have already lost their totipotency and become pluripotent, what means - being able to contribute to the development of the embryo but have lost the capacity to form the trophoblast (which gives rise to the placenta). Thus the term PSC refers to the stem cells that contribute to all three germ layers, mesoderm, ectoderm and endoderm, but not to the trophoblast.
The growing morula develops a central cavity and becomes the blastocyst [16]. A fully developed blastocyst contains cells that are precursors for extra-embryonic tissues including placenta and a distinct group of cells called the inner cell mass (ICM) [17]. The cells of the ICM are also pluripotent and can give rise to all three germ layers of the developing embryo [18]. ICM cells can be also expanded in vitro as immortalized pluripotent ESC lines.
Another potential source of embryo-derived PSC is embryonic germ cells (EG). EG could be derived ex vivo from primordial germ cells (PGC) [19]. Precursors of PGC are the first distinct population of stem cells that can be identified in mouse at cylinder stage embryo at the beginning of gastrulation in the proximal primitive ectoderm (epiblast), a region adjacent to the extraembryonic ectoderm [20]. These founder cells subsequently move through the primitive streak and give rise to several extra-embryonic mesodermal lineages and to germ cells (oocytes and sperm). From a developmental point of view, PGC are the most important population of stem cells because as precursors of germ cells they are responsible for passing genetic information to the next generation [21]. However, if isolated from the developing embryo and cultured ex vivo, these cells are mortal and undergo terminal differentiation [22]. Furthermore, they neither complement blastocyst development nor are able to provide fully functional nuclei during nuclear transfer [15].
One explanation for this obvious lack of pluripotentiality is the fact that PGC are protected from uncontrolled expansion by epigenetic modification/erasure of the somatic imprint on so-called somatic imprinted genes. Somatic imprinted genes show different methylation of some maternally and paternally inherited genes (e.g., H19, Igf-2, Igf-2R, Snrpn) [14, 15]. As result of the imprint for example, Igf-2 is expressed from paternal and H19 from maternal chromosome [14, 15]. Proper somatic imprint is required for PSC to be able to complement blastocyst development.
The process of erasure of somatic imprint occurs very early during gastrulation, at a time when the PGC begin to migrate to the genital ridges [23] and it is one of the basic mechanisms that prevents i) their uncontrolled proliferation, ii) initiation of potential parthenogenesis and iii) prevents teratoma formation [24, 25]. PGC, however, if plated over murine fetal fibroblasts in the presence of selected growth factors (leukemia inhibitory factor, (LIF); basic fibroblast growth factor, (bFGF); and kit ligand, [26] may undergo epigenetic changes in in vitro culture conditions (Figure 1 panel A) and regain the somatic imprint and thus become an immortalized population of PSC, known as embryonic germ cells (EG) [19]. EG in many aspects are equivalent to ES cells and for example similarly as ES contribute to all three germ layers including the germ cell lineage after injection into a blastocyst (blastocyst complementation assay) as well as provide functional nuclei for clonote after nuclear transfer [19, 27].
Next, since during gastrulation three distinct germ layers of cells are established, it is believed that the embryo must possess three stem cell types specific for mesoderm, ectoderm and endoderm. These stem cells are multipotent and subsequently give rise to monopotent stem cells specific for tissues/organs that develop from a given germ layer (Figure 1 panel A). For example, multipotent mesodermal stem cells give rise to monopotent hematopoietic, skeletal muscle, heart, endothelial, and mesenchymal tissue-committed stem cells, multipotent endodermal stem cells give rise to monopotent liver, pancreas, and gut epithelium, while ectodermal multipotent stem cells give rise to monopotent brain, peripheral ganglions and nerves, eye, epidermis and skin tissues [4].
In this context we envision that isolated recently by our group VSELs are epiblast derived stem cells that are deposited early during gastrulation/organogenesis in developing tissues – being a source of precursors for unipotent (tissue committed) stem cells [4, 28]. These cells survive into adulthood (Figure 1 panel A and B).
We hypothesize, that however VSELs are pluripotent they erase during development similarly as PGC proper methylation of maternal and paternal imprinted genes [4, 28]. Due to the erasure of somatic imprint, VSELs similarly as PGC may loose their ability to complement blastocyst development. However, considering the fact that PGC- derived EG cells may reestablish again somatic imprint and thus regain their pluripotentiality, demonstrates that the erasure of somatic imprint is a reversible process. It is also possible that similarly as PGC, also other epiblast-derived PSC residing in adult tissues (e.g., VSELs) may regain a proper somatic imprint under certain circumstances (e.g., epigenetic changes during tissue/organ injury) [4, 28]. It is no doubt that the identification of genes/proteins involved in this phenomenon could be crucial for manipulating pluripotentiality of stem cells isolated from the adult tissues.
VSELs as most primitive population of stem cells in BM?
Evidence is mounting that stem cells that reside in the adult BM demonstrate also developmental hierarchy (Figure 2). It is hypothesized that at the top of this hierarchy resides the most primitive pluripotent stem cell (PSC) that can give rise to long-term repopulating HSC (LT-HSC), mesenchymal stem cells (MSC) and endothelial progenitor cells (EPC). It is also possible that this PSC may give rise to other monopotent tissue-committed stem cells for various organs (e.g., heart, brain and liver) [4, 29]. The existence of such putative PSC in BM was postulated in the past by several investigators; however, these cells were never purified at the single cell level, and their presence was predicted in some of the stem cell plasticity experiments [30–32].
Figure 2. Versatile stem cells in BM - Concept of hierarchical organization of the stem cell pool.

BM is traditionally the home of self-renewing hematopoietic stem cells (HSC). The question is whether BM contains pluripotent stem cells (PSC) which are functional (e.g., VSEL?) and able to supply all other types of stem cell in BM such as i) long-term repopulating HSC (LT-HSC), ii) endothelial progenitor cells (EPC), iii) mesenchymal stem cells (MSC) and iv) perhaps other tissue-committed stem cells (TCSC). LT-HSC gives rise to HSC (short-term repopulating HSC).
Recently a homogenous population of rare (~0.01% of BM MNC) Sca-1+ lin− CD45− cells was identified in murine BM (Figure 3). They express (as determined by RQ-PCR and immunhistochemistry) markers of pluripotent stem cells such as SSEA-1, Oct-4, Nanog and Rex-1 and Rif-1 telomerase protein [12]. Of note, we employed Oct-4 specific primers that do not amplify Oct-4 pseudogenes [33]. Direct electron microscopical analysis revealed that these cells display several features typical for embryonic stem cells such as i) a small size (~3.6 µm in diameter), ii) a large nucleus surrounded by a narrow rim of cytoplasm, and iii) open-type chromatin (euchromatin).
Figure 3. Gating strategy for isolation of BM-derived VSELs by FACS.
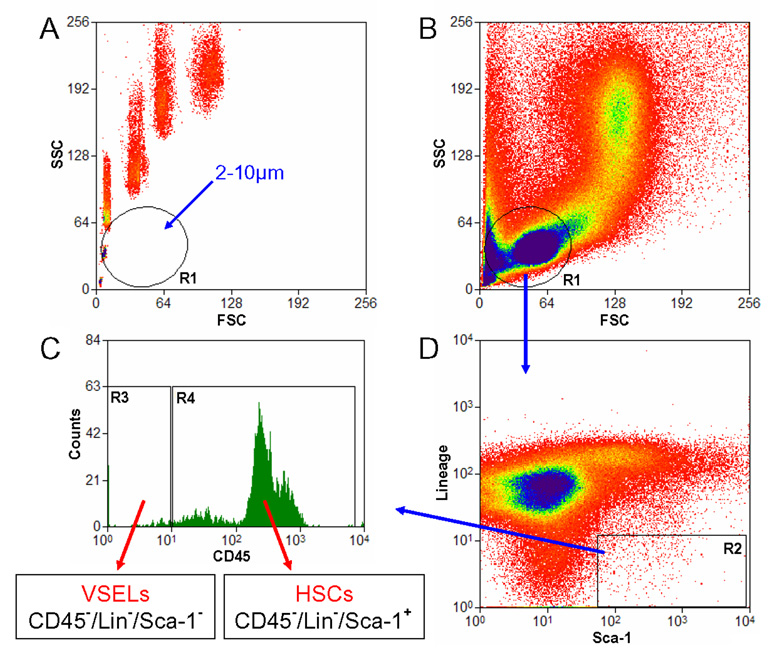
Murine BM-derived VSELs were isolated from full population of bone marrow cells stained for Sca-1, CD45 and hematopoietic lineages markers (Lin) by MoFlo cell sorter (Dako, Carpintera, Ca, USA). Agranular, small events ranging from 2 – 10 µm were included into gate R1 after comparison with six differently sized beads particles with standard diameters of 1, 2, 4, 6, 10 and 15 µm (Flow Cytometry Size beads, Invitrogen; Molecular Probes, Carlsbad, Ca, USA) (Panel A). Cells were visualized by dot plot showing FSC (forward scatter) vs. SSC (side scatter) characteristics, which are related to the size and granularity/complexity of the cell, respectively (Panel B). Objects from region R1 were further analyzed for Sca-1 and Lin expression and only Sca-1+/Lin− events were included into region R2 (Panel D). Population from the region R2 was subsequently sorted based on CD45 marker expression into Sca-1+/Lin−/CD45− VSELs and Sca-1+/Lin−/CD45+ HSCs which are visualized on histogram (Panel C, regions R3 and R4, respectively).
We also employed ImageStream system (ISS) analysis to evaluate better VSELs. This technology was developed as a novel method for multiparameter cell analysis and as a supportive tool for flow cytometry (FC). ISS integrates the features of FC and fluorescent microscopy collecting images of acquired cells for offline digital image analysis [13, 34, 35].
Analysis employing ISS system confirmed a very small size of VSELs as well as other features related to their primitive status such as high nuclear to cytoplasmic ratio [13]. VSELs are smaller than erythrocytes and larger than platelets, as shown in Figure 4. They can be distinguished from large platelets not only based on different surface markers, but also because they contain nuclei. Interestingly, VSELs despite their small size posses diploid DNA, contain numerous mitochondria and high telomerase activity. They do not express MHC-1 and HLA-DR antigens and are CD90− CD105− CD29−. With VSEL isolation, for the first time it has been described a sorting procedure how to purify from adult BM a distinct population of very primitive embryonic-like stem cells and, more importantly, the morphology and the surface markers of these rare cells at the single cell level [13, 36]. Moreover, we reported that VSELs may be released from BM and circulate in blood during tissue/organ injury (e.g., heart infarct and stroke) [37, 38].
Figure 4. Morphology of BM-derived VSELs by ImageStream system.
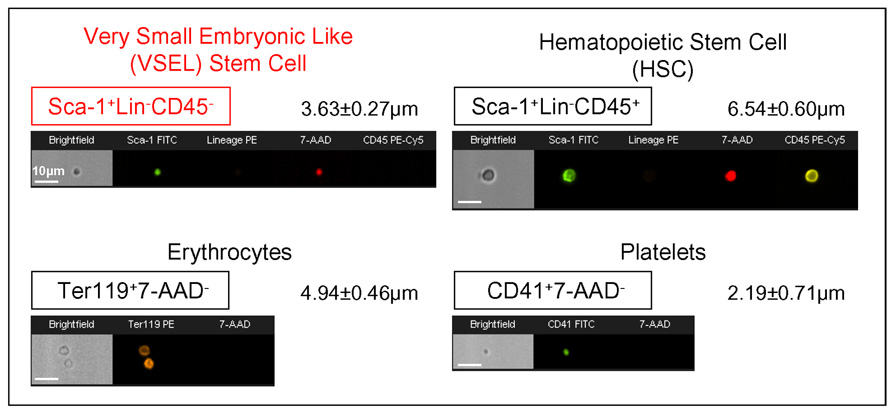
Upper panel shows representative images of Sca-1+/Lin−/CD45− VSEL and Sca-1+/Lin−/CD45+ HSC isolated from murine BM, while the lower panel presents murine erythrocytes and platelets derived from blood. Each population was stained for specific surface markers followed by fixation. Nuclei were visualized with 7-aminoactinomycin D (7-AAD; red). Each photograph presents brightfield image and fluorescence images related to expression of indicated markers and presence of nucleus. Erythrocytes and platelets were identified as anucleated objects (lower panel). The scale bars indicate 10µm. Average size was calculated for each population and is shown as a Mean±SD.
Developmental accumulation of VSELs in BM
We hypothesize that the presence of VSELs in the BM is a result of the “developmental migration” of these epiblast/germ line-derived stem cells during ontogenesis where they find in BM a permissive microenvironment [4, 29]. We established that VSELs express CXCR4 (α-chemokine Gαi protein-coupled seven transmembrane span receptor) and c-met (tyrosine kinase receptor) which are responsible for the robust response of VSELs to the cognate ligands of these receptors - stromal derived factor-1 (SDF-1) and hepatocyte growth factor/scatter factor (HGF/SF), respectively [12]. Since SDF-1 and HGF/SF are secreted by the BM microenvironment, both SDF-1-CXCR4 and HGF/SF-c-met axes may play a pivotal role in the accumulation of VSELs in BM tissue [39]. On the other hand, VSELs residing in the BM could be released/mobilized into circulation if needed, such as during pharmacological G-CSF induced mobilization or during stress related processes related to tissue/organ injury [37, 38]. Accumulating evidence suggests that VSELs residing in the BM play some role in facilitating the regeneration of damaged organs [40]. Since both SDF-1 and HGF/SF are upregulated in the damaged tissues, accumulation of VSEL in damaged organs is again orchestrated by SDF-1-CXCR4 and HGF/SF-c-met axes. Based on this, we hypothesize that VSELs as reserve pool of “mobile” PSC play an important role in turnover of specific tissue committed stem cells and contribute to tissue/organ regeneration both in steady state conditions as well as during organ damage/stress situations.
In vitro pluripotentiality of VSELs
We noticed that ~5–10% of purified VSELs if plated over a C2C12 murine sarcoma cell feeder layer are able to form spheres that resemble embryoid bodies [12]. Cells from these VSEL-derived spheres (VSEL-DS) are composed of immature cells with large nuclei containing euchromatin, and like purified VSELs are CXCR4+SSEA-1+Oct-4+.
Furthermore, VSEL-DS, after re-plating over C2C12 cells, may again grow new secondary or tertiary spheres or, if plated into cultures promoting tissue differentiation, expand into cells from all three germ-cell layers (e.g., cardiomyocytes, neural cells and insulin producing cells). We noticed that VSELs isolated from GFP+ mice grew GFP+ VSEL-DS showing a diploid content of DNA. This confirms that VSEL-DS are derived from VSELs and not from the supportive C2C12 cell line as well as excludes the possibility of cell fusion between VSELs and C2C12 cells respectively. Similar spheres were also formed by VSELs isolated from murine fetal liver, spleen and thymus. Interestingly, formation of VSEL-DS was associated with a young age in mice, and no VSEL-DS were observed in cells isolated from old mice (> 2 years) [1, 12]. This age-dependent content of VSELs in BM may explain why the regeneration processes is more efficient in younger individuals. There are also differences in the content of these cells among BM MNC between long- and short-lived mouse strains. We reported that the concentration of these cells is much higher in BM of long-lived (e.g., C57Bl6) as compared to short-lived (DBA/2J) mice [1]. It would be interesting to identify genes that are responsible for tissue distribution/expansion of these cells as they could be involved in controlling the life span of mammals.
Since VSELs express several markers of primordial germ cells (fetal-type alkaline phosphatase, Oct-4, SSEA-1, CXCR4, Mvh, Stella, Fragilis, Nobox, Hdac6), they could be developmentally closely related to a population of migrating epiblast-derived PGC. VSELs are also highly mobile and respond robustly to an SDF-1 gradient, adhere to fibronectin and fibrinogen, and may interact with BM-derived stromal fibroblasts. Confocal microscopy and time laps studies revealed that these cells attach rapidly to, migrate beneath and undergo emperipolesis in marrow-derived fibroblasts [1]. Since fibroblasts secrete SDF-1 and other chemottractants they may create a homing environment for small CXCR4+ VSELs. This robust interaction of VSELs with BM-derived fibroblasts has an important implication, namely that isolated BM stromal cells may be contaminated by these tiny cells from the beginning. This observation may explain the unexpected “plasticity” of marrow-derived fibroblastic cells (e.g. MSC or MAPC).
Recently, a very similar population of cells that show similar morphology and markers to murine BM-derived VSELs was purified from human CB [41]. Evidence has also mounted that similar cells are also present in the human BM, in particular in young patients. It is anticipated that VSELs could become an important source of pluripotent stem cells for regenerative medicine.
Hematopoiteic differentiation of VSELs
We learned that VSELs freshly isolated from the BM do not show any immediate hematopoietic activity [12]. Accordingly, they do not grow colonies in vitro, do not form spleen colonies in CFU-S assay and do not radioprotect lethally irradiated mice [42].
However, we noticed recently that VSELs that are CD45,− if plated over a supportive OP9 cell line, become primed to hematopoiesis and when subsequently replated in methylocellulose cultures give rise to colonies of CD45+ hematopoietic cells [43]. This hematopoietic differentiation of VSEL is accompanied by upregulation of mRNA for several genes regulating hematopoiesis (e.g., PU-1, c-myb, LMO2, Ikaros). More importantly, these CD45+ cells expanded from CD45−VSEL when transplanted into wild-type animals protect them from lethal irradiation and differentiate in vivo into all the major hematopoietic lineages (e.g., Gr-1+, B220+ and CD3+ cells). Of note, the hematopoietic potential of these cells is maintained after transplantation into secondary recipients [43]. Thus VSEL stem cells could be a population of BM-residing CD45− PSC giving rise to long-term engrafting CD45+ hematopoietic stem cells (Figure 5). As mentioned above, a very similar population of cells that shows similar morphology and markers to murine BM-derived VSEL was purified from human CB [41].
Figure 5. VSELs can give rise to long term repopulating HSC.
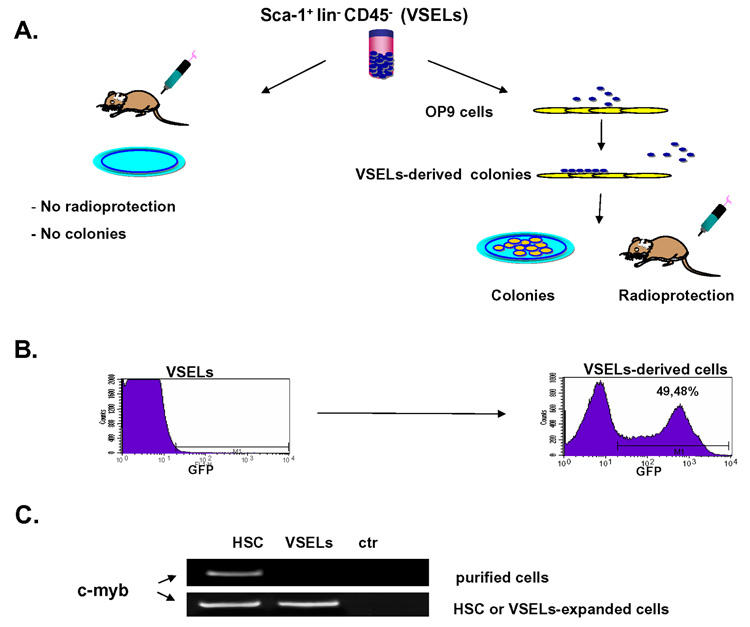
Panel A – VSELs isolated freshly from BM do not grow in vitro hematopoietic colonies, do not show colony froming unit in spleen (CFU-S) activity and do not radioprotect lethally irradiated mice. However, if activated over OP9 cells they acquire hematopoietic potential. Panel B Detection of GFP+ cells in peripheral blood. Left panel - VSELs freshly isolated from GFP+ BM do not show hematopoietic activity in co-transplantation experiments with wild type marrow cell. Right panel – VSELs-derived hematopoietic cells harvested from OP-9 cultures engraft in lethally irradiated mice (~50% of PBMNC are GFP+). Representative data are shown. Panel C Expression of c-myb (RT-PCR) in freshly isolated HSC and VSELs (upper panel) and in HSC and VSELs expanded over OP-9 cells (lower panel). ctr- negative control of RT-PCR reaction (H2O instead of mRNA). Representative data are shown.
Interestingly, other group has also isolated from murine BM a population of very primitive small lymphohematopoietic stem cells that i) do not radioprotect lethally irradiated mice, ii) lack spleen colony-forming (CFU-S) activity and express undetectable or low levels of many antigens presumed to mark LT-HSC, including Thy-1, Sca-1, c-kit and CD34, however, iii) these cells if co-transplanted with short-term engrafting HSC produce delayed multilineage engraftment [44, 45]. These cells are isolated from murine BM based on their very small size by elutriation (low rate of 25 ml/min – FR25), are lin− and express high activity of aldehyde dehydrogenase (ALDHhi) [44, 45]. The relationship of these cells to VSEL requires further study, however it is very likely that these are closely-related populations of the most primitive BM stem cells. This notion is supported by the fact that FR25 lin− ALDHhi cells similarly as VSELs i) are very small, ii) lineage− and iii) do not posses immediate hematopoietic activity (do not grow colonies in vitro, do not form CFU-S in vivo and do not radioprotect lethally irradiated mice) [43, 44].
Furthermore, human BM also contains a population of rare CD34−Lin−CD38− HSC that show decreased clonogeneic activity in vitro, but in vivo engraft robustly in immunodeficient mice [46]. Similar cells were recently also found among human BM-derived CD133+Lin− ALDHhi cells [47]. The relationship of these cells to VSELs requires further studies.
Different strategies to obtain PSC
There are several attempts made to obtain primitive PSC for potential regeneration purposes. There are employed different approaches to obtain PSC such as i) stem cell plasticity, ii) isolation from adult tissues and ii) nuclear reprogramming (Figure 6).
Figure 6. Different strategies to obtain true PSC from non-embryonic tissues.
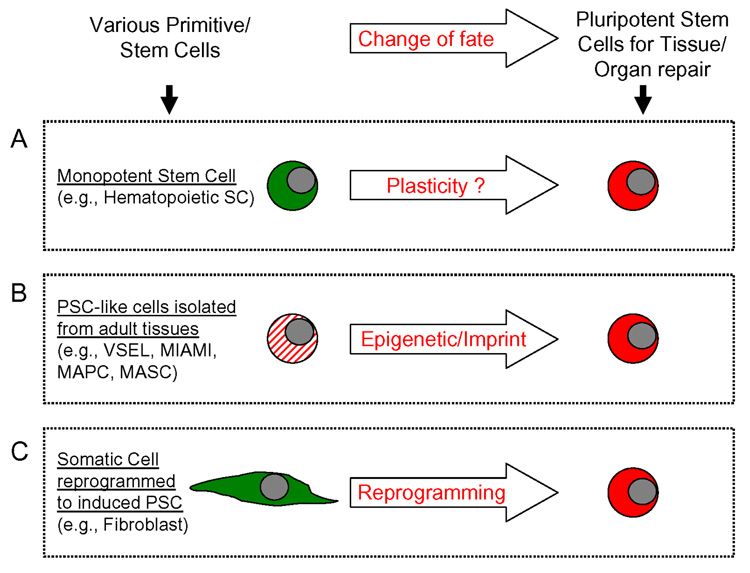
Panel A – Due to the phenomenon of stem cell plasticity of e.g., monopotent HSC may become other types of stem cells. The phenomenon of stem cell plasticity or transdedifferentiation is not confirmed/supported by recent data. Panel B - Isolation of PSC from adult tissues. Several cell candidates are described but so far not one of them was able to contribute in a reproducible way to blastocyst development. In the case of VSEL a problem could be related to erasure of somatic imprint. Panel C - Nuclear reprogramming leads to a generation of PSC-like cells – known as induced PSC (i-PSC). The selected cellular clones, however, form teratomas in syngeneic animals.
Stem Cell Plasticity(Figure 6 panel A)
The phenomenon of stem cell plasticity is based on an assumption that stem cells are “plastic” and thus could trans-dedifferentiate into stem cells committed for various non-hematopoietic organs and tissues [48, 49]. For this phenomenon to occur the following are required (i) a parallel switch of commitment in the compartment of monopotent stem cells or (ii) as postulated, a step back in the differentiation process of monopotent stem cells with their dedifferentiation into multipotent (one germ layer-committed) or even pluripotent (three germ layer-committed) stem cells.
Isolation of PSC from adult tissues(Figure 6 panel B)
Surprisingly, during deliberations concerning stem cell plasticity and the potential contribution of bone marrow (BM) stem cells to organ regeneration, the concept that BM may contain heterogeneous populations of stem cells was not taken into careful consideration [1, 2]. Thus, “positive” data supporting stem cell plasticity or “transdedifferentiation of stem cells” can be re-interpreted by the fact that adult tissue-derived stem cells are heterogeneous and that organs may contain different types of stem cells including perhaps a rare population of PSC. We propose that these cells (e.g., VSELs or MASC), and not HSC, were responsible for positive results with tissue/organ regeneration after infusion of BM-derived cells. Therefore, such PSC isolated from the adult tissues could be a proper non-controversial source of PSC for regeneration.
Reprogramming of somatic cells to iPSC (Figure 6 panel C)
Finally, recent evidence shows that an appropriate “cocktail” of expression vectors encoding four transcription factors such as Oct-4, Sox-2, Myc and Klf4 can reprogram murine fibroblasts to an embryonic status. Nevertheless, cells generated by this strategy (induced pluripotent stem cells – iPSC) have the potential to develop teratomas if injected into syngeneic mice. Thus, the potential clinical application of this strategy to obtain PSC by “debugging cellular reprogramming” by multiple transduced genes requires further studies.
If we compare reprogrammed somatic cells (e.g., fibroblasts – derived induced iPSC) and VSELs important analogies and differences occur [29]. On one hand, the fibroblasts to be transduced with the cocktail of transcription factors which are somatic cells, in contrast to VSEL, possess a proper somatic imprint on maternal and paternal chromosomes. On the other hand, VSEL, which as our preliminary data suggest are germ line-derived cells erase during development somatic imprint, have constitutive expression of all these genes that have to be first over-expressed in fibroblasts (e.g., Oct-4, Sox-2, Myc and Klf4). Therefore, one of the potential strategies to obtain stem cells that will display all the features of PSC (including the potential for blastocyst complementation) would be “debugging cellular reprogramming” by transducing cells by multiple transcription factors (Oct-4, Sox-2, Myc and Klf4) (1st scenario). The other one would rely on the re-establishment of the proper somatic imprint in VSEL (2nd scenario). Our team is currently focused on this second possibility of re-establishment of a proper somatic imprint in VSEL.
The role of bone marrow-derived stem/progenitor cells in pathology – potential involvement of VSELs?
In parallel, evidence has also accumulated that BM-derived stem cells may play an undesirable role in the development of some pathologies. It is likely that if these cells are mobilized at the wrong time and home/seed to the wrong place (e.g., into areas of chronic inflammation) they may exert unwanted effects. For example, BM-derived stem/progenitor cells have been implicated in the pathogenesis of lung fibrosis, ocular pterygia and diabetic neuropathy [50–52].
In 1855 Virchow proposed the “embryonal – rest hypothesis” of tumor formation, based on histological similarities between tumors and embryonic tissues [53]. This theory was later expanded by another pathologist Julius Conheim, who suggested that tumors develop from residual embryonic remnants “lost” during developmental organogenesis. VSELs recently identified in adult tissues could fully support Virchow’s concept. First, if the genomic imprint in these cells is not erased they may retain post-developmental in vivo pluripotency and grow teratomas and teratocarcinomas. Second, if they are closely related to migratory PGC which go astray from the major migratory route to the genital ridges they may ultimately give rise to e.g., germinomas and seminomas. Third, if these cells acquire critical mutations, they may develop into the several types of pediatric sarcomas (e.g., rhabdomyosarcoma, neuroblastoma, Ewing-sarcoma, Willms tumor). In support of this there is a strong correlation with the number of VSELs which persist in postnatal tissues and the coincidence with these types of tumors in pediatric patients. Finally, it is possible that these cells if mobilized at the wrong time into peripheral blood, and deposited in areas of chronic inflammation, instead of playing a role in regeneration may contribute to the development of malignancies. One of the potential mechanisms could be a fusion of VSELs with somatic cells, that through stage of the heterokaryon formation could led to chromosomal aneuploidy, that destabilizes genetic material in the affected cell.
This possibility was recently demonstrated in a model of murine stomach cancer caused by a chronic Helicobacter pylori (HP) infection. In this model BM-derived cells were identified as a source of developing gastric mucosa adenocarcinomas and the SDF-1–CXCR4 axis was implicated in the initiation of this tumor [54]. Accordingly, SDF-1 was found to be upregulated in the gastric mucosa affected by chronic inflammation due to HP infection and postulated to be the chemoattractant responsible for attracting CXCR4+ stem cells (VSELs?) from the BM. Exposed to the chronic inflammatory environment in the gastric mucosa these cells transformed into adenocarcinoma initiating cells. Finally, mounting evidence also suggests that BM may be a source of EPC and MSC for developing cancer tissue. Thus, BM-stem cells (VSELs?) may in different ways contribute to both initiation and expansion of the growing tumor.
Lending further support to the concept that BM-derived cells may initiate the growth of non-hematopoietic malignancies, was the recent report that BM cells exposed in vitro to the carcinogen 3-methycholanthrene could transform into many tumor types including epithelial, neural, muscular, fibroblastic, blood vessel endothelial and poorly differentiated tumors including teratomas [55]. The potential involvement of BM-derived PSC (e.g., VSELs) to this phenomenon requires further studies.
Conclusions
Our data indicate that VSELs could potentially provide a real therapeutic alternative to the controversial use of human ES cells obtained by therapeutic cloning, isolation of single blastomers or from parthenogenetic embryos. Hence, while the ethical debate on the application of ES cells in therapy continues, the potential of VSELs is ripe for exploration. From hematological point of view the fact that CD45− VSELs may differentiate into CD45+ HSC makes from these cells a candidate for long term repopulating HSC. Furthermore, since VSELs may differentiate in vitro into cells from all three germ layers make these cells potential candidates in regenerative medicine. Finally, the mechanism by which VSELs could contribute to development of some malignancies could shed more light on origin of tumors. In conclusion it is of vital importance to evaluate if VSELs could be efficiently employed in the clinic or whether they are merely developmental remnants found in the BM that cannot be harnessed effectively for regeneration. The coming years will bring important answers to these questions.
Acknowledgments
Supported by NIH grant R01 CA106281-01 to MZR.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Kucia M, Reca R, Jala VR, Dawn B, Ratajczak J, Ratajczak MZ. Bone marrow as a home of heterogenous populations of nonhematopoietic stem cells. Leukemia. 2005;19:1118–1127. doi: 10.1038/sj.leu.2403796. [DOI] [PubMed] [Google Scholar]
- 2.Orkin SH, Zon LI. Hematopoiesis and stem cells: plasticity versus developmental heterogeneity. Nat Immunol. 2002;3:323–328. doi: 10.1038/ni0402-323. [DOI] [PubMed] [Google Scholar]
- 3.Nayernia K, Lee JH, Drusenheimer N, et al. Derivation of male germ cells from bone marrow stem cells. Lab Invest. 2006;86:654–663. doi: 10.1038/labinvest.3700429. [DOI] [PubMed] [Google Scholar]
- 4.Ratajczak MZ, Machalinski B, Wojakowski W, Ratajczak J, Kucia M. A hypothesis for an embryonic origin of pluripotent Oct-4+ stem cells in adult bone marrow and other tissues. Leukemia. 2007;21:860–867. doi: 10.1038/sj.leu.2404630. [DOI] [PubMed] [Google Scholar]
- 5.Friedenstein AJ, Petrakova KV, Kurolesova AI, Frolova GP. Heterotopic of bone marrow.Analysis of precursor cells for osteogenic and hematopoietic tissues. Transplantation. 1968;6:230–247. [PubMed] [Google Scholar]
- 6.Friedenstein AJ, Piatetzky-Shapiro II, Petrakova KV. Osteogenesis in transplants of bone marrow cells. J Embryol Exp Morphol. 1966;16:381–390. [PubMed] [Google Scholar]
- 7.Prockop DJ. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science. 1997;276:71–74. doi: 10.1126/science.276.5309.71. [DOI] [PubMed] [Google Scholar]
- 8.Jiang Y, Jahagirdar BN, Reinhardt RL, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature. 2002;418:41–49. doi: 10.1038/nature00870. [DOI] [PubMed] [Google Scholar]
- 9.D'Ippolito G, Diabira S, Howard GA, Menei P, Roos BA, Schiller PC. Marrow-isolated adult multilineage inducible (MIAMI) cells, a unique population of postnatal young and old human cells with extensive expansion and differentiation potential. J Cell Sci. 2004;117:2971–2981. doi: 10.1242/jcs.01103. [DOI] [PubMed] [Google Scholar]
- 10.D'Ippolito G, Howard GA, Roos BA, Schiller PC. Isolation and characterization of marrow-isolated adult multilineage inducible (MIAMI) cells. Exp Hematol. 2006;34:1608–1610. doi: 10.1016/j.exphem.2006.07.016. [DOI] [PubMed] [Google Scholar]
- 11.Beltrami AP, Cesselli D, Bergamin N, et al. Multipotent cells can be generated in vitro from several adult human organs (heart, liver and bone marrow) Blood. 2007;110:3438–3446. doi: 10.1182/blood-2006-11-055566. [DOI] [PubMed] [Google Scholar]
- 12.Kucia M, Reca R, Campbell FR, et al. A population of very small embryonic-like (VSEL) CXCR4(+)SSEA-1(+)Oct-4+ stem cells identified in adult bone marrow. Leukemia. 2006;20:857–869. doi: 10.1038/sj.leu.2404171. [DOI] [PubMed] [Google Scholar]
- 13.Zuba-Surma EK, Kucia M, Abdel-Latif A, et al. Morphological characterization of Very Small Embryonic-Like stem cells (VSELs) by ImageStream system analysis. J Cell Mol Med. 2007 Nov 20; doi: 10.1111/j.1582-4934.2007.00154.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mann JR. Imprinting in the germ line. Stem Cells. 2001;19:287–294. doi: 10.1634/stemcells.19-4-287. [DOI] [PubMed] [Google Scholar]
- 15.Yamazaki Y, Mann MR, Lee SS, et al. Reprogramming of primordial germ cells begins before migration into the genital ridge, making these cells inadequate donors for reproductive cloning. Proc Natl Acad Sci U S A. 2003;100:12207–12212. doi: 10.1073/pnas.2035119100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.O'Farrell PH, Stumpff J, Su TT. Embryonic cleavage cycles: how is a mouse like a fly? Curr Biol. 2004;14:R35–R45. doi: 10.1016/j.cub.2003.12.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Evans MJ, Kaufman MH. Establishment in culture of pluripotential cells from mouse embryos. Nature. 1981;292:154–156. doi: 10.1038/292154a0. [DOI] [PubMed] [Google Scholar]
- 18.Bradley A, Evans M, Kaufman MH, Robertson E. Formation of germ-line chimaeras from embryo-derived teratocarcinoma cell lines. Nature. 1984;309:255–256. doi: 10.1038/309255a0. [DOI] [PubMed] [Google Scholar]
- 19.Matsui Y, Zsebo K, Hogan BL. Derivation of pluripotential embryonic stem cells from murine primordial germ cells in culture. Cell. 1992;70:841–847. doi: 10.1016/0092-8674(92)90317-6. [DOI] [PubMed] [Google Scholar]
- 20.McLaren A, Lawson KA. How is the mouse germ-cell lineage established? Differentiation. 2005;73:435–437. doi: 10.1111/j.1432-0436.2005.00049.x. [DOI] [PubMed] [Google Scholar]
- 21.Donovan PJ. The germ cell--the mother of all stem cells. Int J Dev Biol. 1998;42:1043–1050. [PubMed] [Google Scholar]
- 22.De Felici M, McLaren A. In vitro culture of mouse primordial germ cells. Exp Cell Res. 1983;144:417–427. doi: 10.1016/0014-4827(83)90421-4. [DOI] [PubMed] [Google Scholar]
- 23.Lee J, Inoue K, Ono R, et al. Erasing genomic imprinting memory in mouse clone embryos produced from day 11.5 primordial germ cells. Development. 2002;129:1807–1817. doi: 10.1242/dev.129.8.1807. [DOI] [PubMed] [Google Scholar]
- 24.Macchiarini P, Ostertag H. Uncommon primary mediastinal tumours. Lancet Oncol. 2004;5:107–118. doi: 10.1016/S1470-2045(04)01385-3. [DOI] [PubMed] [Google Scholar]
- 25.Oosterhuis JW, Looijenga LH. Testicular germ-cell tumours in a broader perspective. Nat Rev Cancer. 2005;5:210–222. doi: 10.1038/nrc1568. [DOI] [PubMed] [Google Scholar]
- 26.Donovan PJ. Growth factor regulation of mouse primordial germ cell development. Curr Top Dev Biol. 1994;29 doi: 10.1016/s0070-2153(08)60551-7. [DOI] [PubMed] [Google Scholar]
- 27.Zwaka TP, Thomson JA. A germ cell origin of embryonic stem cells? Development. 2005;132:227–233. doi: 10.1242/dev.01586. [DOI] [PubMed] [Google Scholar]
- 28.Kucia M, Wu W, Ratajczak MZ. Bone marrow-derived very small embryonic-like stem cells: Their developmental origin and biological significance. Dev Dyn. 2007;11 doi: 10.1002/dvdy.21180. [DOI] [PubMed] [Google Scholar]
- 29.Ratajczak MZ, Zuba-Surma EK, Wysoczynski M, et al. Hunt for pluripotent stem cell - Regenerative medicine search for almighty cell. J Autoimmun. 2008;30:151–162. doi: 10.1016/j.jaut.2007.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Di Campli C, Piscaglia AC, Pierelli L, et al. A human umbilical cord stem cell rescue therapy in a murine model of toxic liver injury. Dig Liver Dis. 2004;36:603–613. doi: 10.1016/j.dld.2004.03.017. [DOI] [PubMed] [Google Scholar]
- 31.Lagasse E, Connors H, Al-Dhalimy M, et al. Purified hematopoietic stem cells can differentiate into hepatocytes in vivo. Nat Med. 2000 Jun;:1229–1234. doi: 10.1038/81326. [DOI] [PubMed] [Google Scholar]
- 32.Orlic D, Kajstura J, Chimenti S, et al. Bone marrow cells regenerate infarcted myocardium. Nature. 2001;410:701–705. doi: 10.1038/35070587. [DOI] [PubMed] [Google Scholar]
- 33.Lengner CJ, Camargo FD, Hochedlinger K, et al. Oct4 expression is not required for mouse somatic stem cell self-renewal. Cell Stem Cell. 2007;1:403–415. doi: 10.1016/j.stem.2007.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zuba-Surma EK, Kucia M, Abdel-Latif A, Lillard JJ, Ratajczak MZ. The ImageStream System: a key step to a new era in imaging. Folia Histochem Cytobiol. 2007;45:279–290. [PubMed] [Google Scholar]
- 35.Zuba-Surma EK, Kucia M, Ratajczak MZ. “Decoding of Dot”: The ImageStream System (ISS) as a Supportive Tool for Flow Cytometric Analysis. Cent Eur J Biol. 2008;3:1–10. [Google Scholar]
- 36.Kucia M, Wysoczynski MJ, Ratajczak J, Ratajczak MZ. Identification of Very Small Embryonic Like (VSEL) stem cells in bone marrow. Cell Tissue Res. 2008;33:125–134. doi: 10.1007/s00441-007-0485-4. [DOI] [PubMed] [Google Scholar]
- 37.Kucia M, Zhang YP, Reca R, et al. Cells enriched in markers of neural tissue-committed stem cells reside in the bone marrow and are mobilized into the peripheral blood following stroke. Leukemia. 2006;20:18–28. doi: 10.1038/sj.leu.2404011. [DOI] [PubMed] [Google Scholar]
- 38.Zuba-Surma EK, Kucia M, Dawn B, Guo Y, Ratajczak MZ, Bolli R. Bone marrow-derived pluripotent Very Small Embryonic-Like stem cells (VSELs) are mobilized after acute myocardial infarction. J Mol Cell Cardiol. 2008 doi: 10.1016/j.yjmcc.2008.02.279. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ratajczak MZ, Zuba-Surma E, Kucia M, Reca R, Wojakowski W, Ratajczak J. The pleiotropic effects of the SDF-1-CXCR4 axis in organogenesis, regeneration and tumorigenesis. Leukemia. 2006;20:1915–1924. doi: 10.1038/sj.leu.2404357. [DOI] [PubMed] [Google Scholar]
- 40.Zuba-Surma EK, Taher H, Kucia M, et al. AHA Scientific Sessions. Orlando, Fl: Circulation; 2007. Transplantation of bone marrow-derived very small embryonic-like stem cells (VSELs) improves left ventricular function and remodeling after myocardial infarction; p. II-204. [Google Scholar]
- 41.Kucia M, Halasa M, Wysoczynski M, et al. Morphological and molecular characterization of novel population of CXCR4(+) SSEA-4(+) Oct-4(+) very small embryonic-like cells purified from human cord blood - preliminary report. Leukemia. 2007;21:297–303. doi: 10.1038/sj.leu.2404470. [DOI] [PubMed] [Google Scholar]
- 42.Kucia M, Ratajczak J, Ratajczak MZ. Bone marrow as a source of circulating CXCR4+ tissue-committed stem cells. Biol Cell. 2005;97:133–146. doi: 10.1042/BC20040069. [DOI] [PubMed] [Google Scholar]
- 43.Wysoczynski M, Kucia M, Zuba-Surma EK, Wu W, Ratajczak MZ, Ratajczak J. An in vivo evidence that the CD45negative adult marrow-derived CXCR4+ SSEA-1+ Oct-4+ Very Small Embryonic Like (VSEL) stem cells may differentiate into CD45positive long term repopulating hematopoietic stem cells. ASH Meeting: Blood. 2007:505. [Google Scholar]
- 44.Jones RJ, Collector MI, Barber JP, et al. Characterization of mouse lymphohematopoietic stem cells lacking spleen colony-forming activity. Blood. 1996;88:487–491. [PubMed] [Google Scholar]
- 45.Jones RJ, Wagner JE, Celano P, Zicha MS, Sharkis SJ. Separation of pluripotent haematopoietic stem cells from spleen colony-forming cells. Nature. 1990;347:188–189. doi: 10.1038/347188a0. [DOI] [PubMed] [Google Scholar]
- 46.Larochelle A, Vormoor J, Hanenberg H, et al. Identification of primitive human hematopoietic cells capable of repopulating NOD/SCID mouse bone marrow: implications for gene therapy. Nat Med. 1996;2:1329–1337. doi: 10.1038/nm1296-1329. [DOI] [PubMed] [Google Scholar]
- 47.Hess DA, Wirthlin L, Craft TP, et al. Selection based on CD133 and high aldehyde dehydrogenase activity isolates long-term reconstituting human hematopoietic stem cells. Blood. 2006;107:2162–2169. doi: 10.1182/blood-2005-06-2284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hess DC, Abe T, Hill WD, et al. Hematopoietic origin of microglial and perivascular cells in brain. Exp Neurol. 2004;186:134–144. doi: 10.1016/j.expneurol.2003.11.005. [DOI] [PubMed] [Google Scholar]
- 49.Mezey E, Chandross KJ, Harta G, Maki RA, McKercher SR. Turning blood into brain: cells bearing neuronal antigens generated in vivo from bone marrow. Science. 2000;290:1779–1782. doi: 10.1126/science.290.5497.1779. [DOI] [PubMed] [Google Scholar]
- 50.Hasegawa T, Kosaki A, Shimizu K, et al. Amelioration of diabetic peripheral neuropathy by implantation of hematopoietic mononuclear cells in streptozotocin-induced diabetic rats. Exp Neurol. 2006;199:274–280. doi: 10.1016/j.expneurol.2005.11.001. [DOI] [PubMed] [Google Scholar]
- 51.Hashimoto N, Jin H, Liu T, Chensue SW, Phan SH. Bone marrow-derived progenitor cells in pulmonary fibrosis. J Clin Invest. 2004;113:243–252. doi: 10.1172/JCI18847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Song YS, Ryu YH, Choi SR, Kim JC. The involvement of adult stem cells originated from bone marrow in the pathogenesis of pterygia. Yonsei Med J. 2005;46:687–692. doi: 10.3349/ymj.2005.46.5.687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Virchow R. Editorial Archive fuer pathologische Anatomie und Physiologie fuer klinische Medizin. 1855;8:23–54. [Google Scholar]
- 54.Houghton J, Stoicov C, Nomura S, et al. Gastric cancer originating from bone marrow-derived cells. Science. 2004:306. doi: 10.1126/science.1099513. [DOI] [PubMed] [Google Scholar]
- 55.Liu C, Chen Z, Chen Z, Zhang T, Lu Y. Multiple tumor types may originate from bone marrow-derived cells. Neoplasia. 2006;8:716–724. doi: 10.1593/neo.06253. [DOI] [PMC free article] [PubMed] [Google Scholar]


