Summary
Group III metabotropic glutamate receptors (mGluRs) reduce synaptic transmission at the Schaffer collateral-CA1 (SC-CA1) synapse in rats by a presynaptic mechanism. Previous studies show that low concentrations of the group III-selective agonist, L-AP4, reduce synaptic transmission in slices from neonatal but not adult rats, whereas high micromolar concentrations reduce transmission in both age groups. L-AP4 activates mGluRs 4 and 8 at much lower concentrations than those required to activate mGluR7, suggesting that the group III mGluR subtype modulating transmission is a high affinity receptor in neonates and a low affinity receptor in adults. The previous lack of subtype selective ligands has made it difficult to test this hypothesis. We have measured fEPSPs in the presence of novel subtype selective agents to address this question. We show that the effects of L-AP4 can be blocked by LY341495 in both neonates and adults, verifying that these effects are mediated by mGluRs. In addition, the selective mGluR8 agonist, DCPG, has a significant effect in slices from neonatal rats but does not reduce synaptic transmission in adult slices. The mGluR4 selective allosteric potentiator, PHCCC, is unable to potentiate the L-AP4-induced effects at either age. Taken together, our data suggest that group III mGluRs regulate transmission at the SC-CA1 synapse throughout development but there is a developmental regulation of the subtypes involved so that that both mGluR8 serves this role in neonates but not adults whereas mGluR7 is involved in regulating transmission at this synapse in throughout postnatal development.
Keywords: metabotropic glutamate receptor, group III, hippocampus, DCPG, L-AP4
Introduction
Glutamate is the major excitatory neurotransmitter in the brain and can exert its effects through activation of both ionotropic and metabotropic glutamate receptors (mGluRs). The mGluRs are members of the family C G protein-coupled receptors (GPCRs). Eight mGluR subtypes have been identified mammalian brain and are grouped into three groups based on their sequence homologies, ligand selectivity, and downstream effector molecules. Group I mGluRs include mGluR1 and mGluR5 which couple to Gq and activate PLCβ. mGluRs 2 and 3 comprise Group II and mGluRs 4, 6, 7, and 8 comprise Group III. The members of Groups II and III couple to Gi/o which results in modulation of multiple effector systems, including various ion channels and inhibition of adenylyl cyclase (see (Conn and Pin, 1997) for review).
One of the most common physiological effects of mGluR activation that is consistent throughout the central nervous system is a role as presynaptic autoreceptors involved in reducing transmission at glutamatergic synapses. For instance, activation of mGluRs reduces transmission at each of the major glutamatergic synapses in the hippocampal formation. Interestingly, evidence suggests that different mGluR subtypes serve this role in each major hippocampal synapse (Gereau and Conn, 1995b; Kamiya and Ozawa, 1999; Koerner and Cotman, 1981, 1982; Lanthorn et al., 1984; Macek et al., 1996). One of the most important synapses in the hippocampal circuit that is often implicated in learning and memory is the synapse between CA3 pyramidal cell axons, termed Schaffer collaterals (SC), and CA1 pyramidal cells (SC-CA1 synapse). Previous studies suggest that Group III mGluRs attenuate transmission at the SC-CA1 synapse by a presynaptic mechanism (Baskys and Malenka, 1991; Gereau and Conn, 1995a, 1995b; Vignes et al., 1995). Baskys and Malenka (1991) found that 50µM L-AP4, a selective agonist of group III mGluRs, attenuates transmission at this synapse in slices from neonatal but not adult rats. These data suggested that group III mGluRs may only participate in regulation of transmission at the SC-CA1 synapse at early stages of postnatal development. However, subsequent studies revealed that higher concentrations of L-AP4 (500µM) have similar effects on transmission at this synapse in slices from neonatal and adult animals (Gereau and Conn, 1995a), suggesting that group III mGluRs regulate SC-CA1 transmission throughout postnatal development. A difference in the effect of 50 µM vs 500µM L-AP4 raises the possibility of a developmental regulation of the specific group III mGluR subtype(s) that serve this role in neonatal versus adult animals. For instance, L-AP4 has nM potencies at mGluR4 and mGluR8 but µM potency at mGluR7 (Schoepp et al., 1999). Thus, mGluR4 or 8 could predominate at early postnatal stages while mGluR7 could serve as the predominant receptor mediating this response in adults. However, the reduction in sensitivity to L-AP4 could also be due to the presence of high receptor reserve in neonatal animals and a developmental decrease in expression of a single group III mGluR subtype, leading to loss of receptor reserve and reduced agonist potency. In support of the possibility that mGluR7 is the only group III mGluR subtype involved in regulating transmission at this synapse in adults, immunohistochemical analysis of Group III mGluRs in the adult rat hippocampus revealed that mGluR4a is primarily localized to the inner third of the molecular layer and mGluR8 is found in the lateral perforant path terminal zone. In contrast, mGluR7a is distributed in all dendritic layers throughout the hippocampus (Bradley et al., 1996; Corti et al., 2002; Kosinski et al., 1999; Shigemoto et al., 1997). While immunohistochemical analysis has not been performed for all group III mGluR subtypes in neonates, antibodies to mGluR7a in the neonatal (p7) rat hippocampus revealed that this receptor is not as highly expressed in the stratum radiatum of the CA1 region (Bradley et al., 1998). Thus, it is unlikely that mGluR7 expression and receptor reserve are higher in neonates than adults. Therefore, it is possible that another mGluR subtype is expressed in neonates and is responsible for the higher potency of L-AP4.
Unfortunately, pharmacological reagents that clearly differentiate between the group III mGluR subtypes have not been available, making it impossible to directly determine the mechanism for this difference. However, recent efforts have provided novel pharmacological tools that provide greater selectivity between group III mGluR subtypes (Marino et al., 2003; Mitsukawa et al., 2005; Thomas et al., 2001; Zhai et al., 2002) than previously available compounds. We have used these newer reagents to rigorously test the hypothesis that there is a developmental regulation of the presynaptic mGluR subtypes involved in regulating transmission at this synapse and to determine which mGluR subtypes mediate this effect at the different stages of postnatal development.
Methods
Compounds
L-(+)-2-amino-4-phosphonobutyric acid (L-AP4) and N,N’-dibenzhydrylethane-1,2-diamine (AMN082) were purchased from Ascent Scientific (Weston-Super-Mare, UK) or Tocris Bioscience (Ellisville, MO). (S)-3,4-Dicarboxyphenylglycine ((S)-DCPG), N-Phenyl-7-(hydroxyimino) cyclopropa[b] chromen-1a-carboxamide (PHCCC), 2S)-2-amino-2-[(1S,2S)-2-carboxycycloprop-1-yl]-3-xanth-9-yl) propanoic acid (LY341495), were purchased from Tocris Bioscience (Ellisville, MO). Z-Cyclopentyl AP4 ((Z)-1(RS)-amino-3(RS)-phosphonocyclopentanecarboxylic acid) (Crooks et al., 1986) was supplied by Dr. Rodney Johnson (University of Minnesota).
Animals
All animals used in these studies were cared for in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Experimental protocols were in accordance with all applicable guidelines regarding the care and use of animals. Animals were housed in an Association for Assessment and Accreditation of Laboratory Animal Care (AALAC) International approved facility with free access to food and water. All efforts were made to minimize animal suffering and to reduce the number of animals used.
Electrophysiology
Nine- to 11-day-old (neonatal) or >80-day-old (adult) male Sprague Dawley rats (Charles River, Wilmington, MA) were anesthetized with isoflorane, decapitated and the brains were quickly removed and submerged into ice cold cutting solution (in mM: 110 sucrose, 60 NaCl, 3 KCl, 1.25 NaH2PO4, 28 NaHCO3, 5 glucose, 0.6 (+)-sodium-L-7 ascorbate, 0.5 CaCl2, 7 MgCl2) continuously bubbled with 95% O2/5% CO2. The brains were then hemisected and 400µm transverse slices were made using a vibratome (Leica VT100S). Individual hippocampi were removed from the slice and transferred to a room temperature mixture containing equal volumes of cutting solution and artificial cerebrospinal fluid (ACSF; in mM: 125 NaCl, 2.5 KCl, 1.25 NaH2PO4, 25 NaHCO3, 25 glucose, 2 CaCl2, 1 MgCl2) where they were allowed to equilibrate for 30 minutes. The hippocampi were then placed into a holding chamber containing room temperature ACSF continuously bubbled with 95% O2/5% CO2 and allowed to recover for at least 1 hour.
Following recovery, slices were moved to a submersion chamber continuously perfused with oxygenated ACSF at a rate of 1.5 milliliter per minute. ASCF temperature was maintained between 24 and 25°C. Field excitatory postsynaptic potentials (fEPSPs) were evoked at 0.05Hz by placing a bipolar nichrome stimulating electrode in the stratum radiatum near the CA3-CA1 border in order to stimulate the Schaffer collaterals. Baseline intensities that evoked half-maximal fEPSPs were chosen for all experiments. Recording electrodes were pulled on a Flaming/Brown micropipette puller (Sutter Instruments) to a resistance of 4-5MΩ, filled with ACSF and placed in the stratum radiatum of area CA1. Field potentials were recorded using either an Axoclamp 2 (Molecular Devices, Sunnyvale, CA) or a Microelectrode AC Amplifier Model 1800 (A–M Systems) and analyzed using Clampex 9.2. Sampled data was analyzed off-line using Clampfit 9.2. Three sequential fEPSPs were averaged and their slopes calculated. All fEPSP slopes were normalized to the average slope calculated during the predrug period (percent of predrug).
Cell-based assays
HEK 293 cells stably expressing GIRK 1, GIRK 2 were generously provided by Dr. Huai Hu Chang and Dr. Lily Jan (University of San Francisco, San Francisco, CA). HEK/GIRK cells were stably transfected with either human mGluR7 or human mGluR8 (mGluR7/GIRK or mGluR8/GIRK) and grown in 45% Dulbecco’s Modified Eagle Media (DMEM), 45% Ham’s F12, 10% fetal bovine serum (FBS), 100 units/ml penicillin/streptomycin, 20 mM HEPES (pH 7.3), 1 mM sodium pyruvate, and 2 mM glutamine. Cells were plated into 384 well, black walled, clear bottom poly-D-lysine coated plates (Greiner) at a density of 15×105 cells/20 µl/well in glutamate/glutamine-free medium. Plated cells were incubated overnight at 37°C in an atmosphere of 95% air, 5% CO2.
Thallium flux assays were carried out as described in Niswender et al (2008). Briefly, on the day of the assay, medium was removed from the cells and 20 µl/well of 1.67 µg/ml of the indicator dye BTC-AM (Invitrogen) in assay buffer (Hanks’ Balanced Salt Solution (Invitrogen) containing 20 mM HEPES pH 7.3; HBSS) was added to the plated cells. Cells were incubated for one hour at room temperature and the dye was replaced with 40 µl/well HBSS (agonist studies) or 20 µl/well HBSS (potentiator experiments). For agonist studies, compounds were diluted in thallium assay buffer (125 mM sodium gluconate or 125 mM sodium bicarbonate, 1 mM magnesium sulfate, 1.8 mM calcium gluconate, 5 mM glucose, 12 mM thallium sulfate, 10 mM HEPES, pH 7.3) at 5x the final concentration to be assayed. For antagonist or potentiator experiments, compounds were diluted to 2.5X their final desired concentration in HBSS. Compounds were added in a 20 µl volume and incubated for five minutes prior to the addition of 10 µl of thallium buffer ± agonist. Thallium flux was measured using the Functional Drug Screening System (Hamamatsu Photonic Systems). All raw data was analyzed by Excel and then imported to GraphPad Prism for graphing and statistical analysis.
Statistical Analysis
All data from cell-based assays were plotted and analyzed using GraphPad Prism software to calculate standard errors. EC50 values were calculated in Prism using the sigmoidal dose-response variable slope equation. Statistical analysis of electrophysiology data was performed in Prism using either student’s unpaired t-test or one way ANOVA with Bonferroni's multiple comparison test or Dunnett’s post hoc test to compare multiple sets of data. P values less than 0.05 (p<0.05) were considered to be statistically significant. All data are presented as mean ± SEM.
Results
L-AP4 has higher potency at reducing synaptic transmission at the SC-CA1 synapse in neonatal rats than in adults
As a first step to testing the hypothesis that a group III mGluR with relatively high affinity for L-AP4 (mGluR4 or mGluR8) regulates transmission at the SC-CA1 synapse in neonates, we determined the effects of multiple concentrations of L-AP4 on evoked fEPSPs at this synapse in slices from neonatal animals. Consistent with a previous study (Baskys and Malenka, 1991), 50µM L-AP4 induced a significant reduction of fEPSP slope (65±8.7% of predrug; p<0.01) (Fig.1A). A concentration response curve (CRC) analysis (Fig. 1B) revealed an EC50 value for L-AP4 of 25µM.
Figure 1.
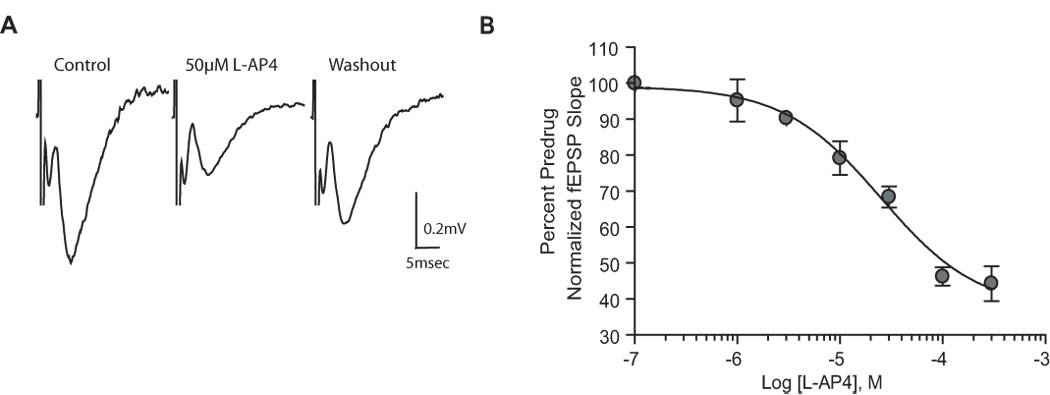
L-AP4 reduces synaptic transmission at the SC-CA1 synapse in the neonatal rat hippocampus. A: representative field excitatory postsynaptic potentials (fEPSP) recorded from the dendritic layer of CA1 with the use of extracellular field recording techniques. Traces are the average of 5 trials immediately before and 10 min after application of L-AP4 (50µM). Field EPSPs were elicited by stimulating the Schaffer collaterals. B: concentration-response curve for L-AP4. Shown is the depression (mean ± SEM) of fEPSP slope induced by increasing concentrations of L-AP4. Data are expressed as a percentage of predrug fEPSP slope. The concentration producing half-maximal depression is 25µM. N = 3–6 for each point.
In contrast to slices from neonatal animals, 50µM L-AP4 did not significantly reduce fEPSPs at the SC-CA1 synapse in slices from adult animals (Fig. 2A; 98.4±5.4% of predrug, p>0.05) whereas 1mM L-AP4 significantly reduced fEPSP slope (Fig. 2A; 50±7.5% of predrug; p<0.01). We then performed a full CRC analysis to directly compare potencies of L-AP4 at the different ages (Fig. 2B). The EC50 value for L-AP4 in slices from adult animals was 1mM. These data extend the previous studies and confirm that L-AP4 has much higher potency in regulating transmission at the SC-CA1 synapse at early stages of postnatal development than in adults.
Figure 2.
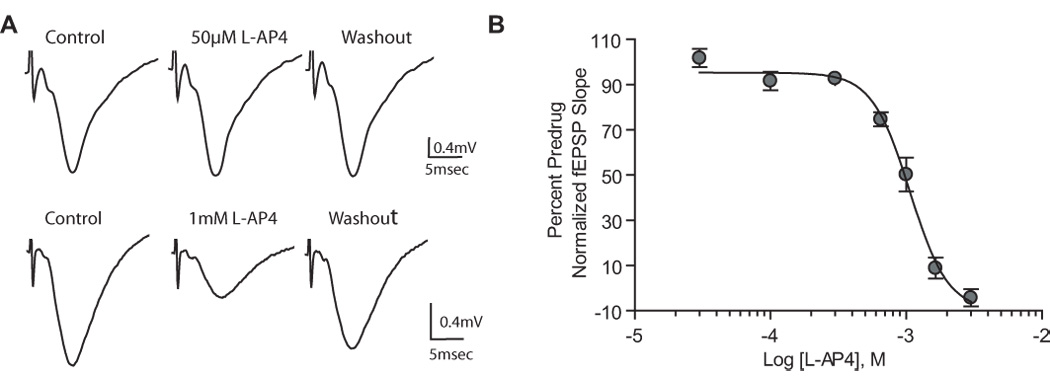
High concentrations of L-AP4 are required to reduce synaptic transmission at the SC-CA1 synapse of the adult rat hippocampus. A: representative field excitatory postsynaptic potential (fEPSP) recorded from the dendritic layer of CA1 with the use of extracellular field recording techniques, showing the effects of the group III-selective agonist L-AP4 (50µM or 1mM). Field EPSPs were elicited by stimulating the Schaffer collaterals. Traces are the averages of 5 trials immediately before and 10 min after application of L-AP4. High concentrations of L-AP4 (1mM) are required to reduce the slope of the fEPSP. B: concentration-response curve for L-AP4. Shown is the depression (mean ± SEM) of fEPSP slope induced by increasing concentrations of L-AP4. Data are expressed as a percentage of predrug fEPSP slope. The concentration producing half-maximal depression is 1mM. N = 3–5 for each point.
The finding that low concentrations of L-AP4 reduce transmission at the SC-CA1 synapse in neonates than in adults does not rule out the possibility that higher concentrations of L-AP4 could further reduce transmission at this synapse, as they do in slices from adult animals. Thus, we also determined the effect of 1 mM L-AP4 in slices from neonates. Interestingly, we found that increasing the concentrations of L-AP4 to 1mM resulted in a further reduction of synaptic transmission relative to the maximal reduction observed with the lower µM concentrations (2.62 ± 2.12% of predrug; p<0.0001). These results suggest that the low potency L-AP4 effect occurs at both ages but that the response to L-AP4 in neonates is biphasic so that there is also a higher potency component.
Previous data from our lab and others have shown that the effects of L-AP4 in both neonatal and adult rats are mediated by a presynaptic mechanism (Baskys and Malenka, 1991; Gereau and Conn, 1995a). Thus, lower µM concentrations of L-AP4 increase paired pulse facilitation in neonates (Baskys and Malenka, 1991). In adult rats, 1mM L-AP4 reduces the frequency of miniature EPSCs as well as increasing the paired pulse facilitation ratio (Gereau and Conn, 1995a). Thus, it is likely that effects of both low and high concentrations of L-AP4 are mediated by activation of presynaptic L-AP4-sensitive receptors.
LY341495 antagonizes the L-AP4-induced reduction of synaptic transmission in both neonatal and young adult rats
LY341495 is an orthosteric antagonist of all mGluR subtypes. While this compound is selective for group II mGluRs relative to other mGluR subtypes, it is a relatively potent antagonist each of the group III mGluR subtypes as well (Edl et al., 2006; Kingston et al., 1998; Niswender et al., 2008; Schoepp et al., 1999). To confirm that the effect of L-AP4 is mediated by group III mGluRs and not by a non-specific action or off target activity, we determined the effect of LY341495 on the response to L-AP4 in slices from both neonatal and adult rats. The effect of 10µM L-AP4 at the SC-CA1 synapse in neonatal animals was blocked by 30µM LY341495 (Fig. 3A; 10µM L-AP4, 76.7±4% of predrug; 3µM LY341495 + 10µM L-AP4, 100.5±3.4% of predrug; p<0.01). LY341495 also significantly blocked the response of 650µM L-AP4 at the adult rat SC-CA1 synapse (Fig. 3B; 650µM L-AP4, 74.6±3% of predrug; 100µM LY341495 + 650µM L-AP4, 88.2±2.4% predrug; p<0.05), however it was necessary to increase the concentration of LY341495 in adult animals due to the lower potency of LY341495 at inhibiting mGluR7 realtive to mGluR8 (Edl et al., 2006; Niswender et al., 2008; Schoepp et al., 1999).
Figure 3.
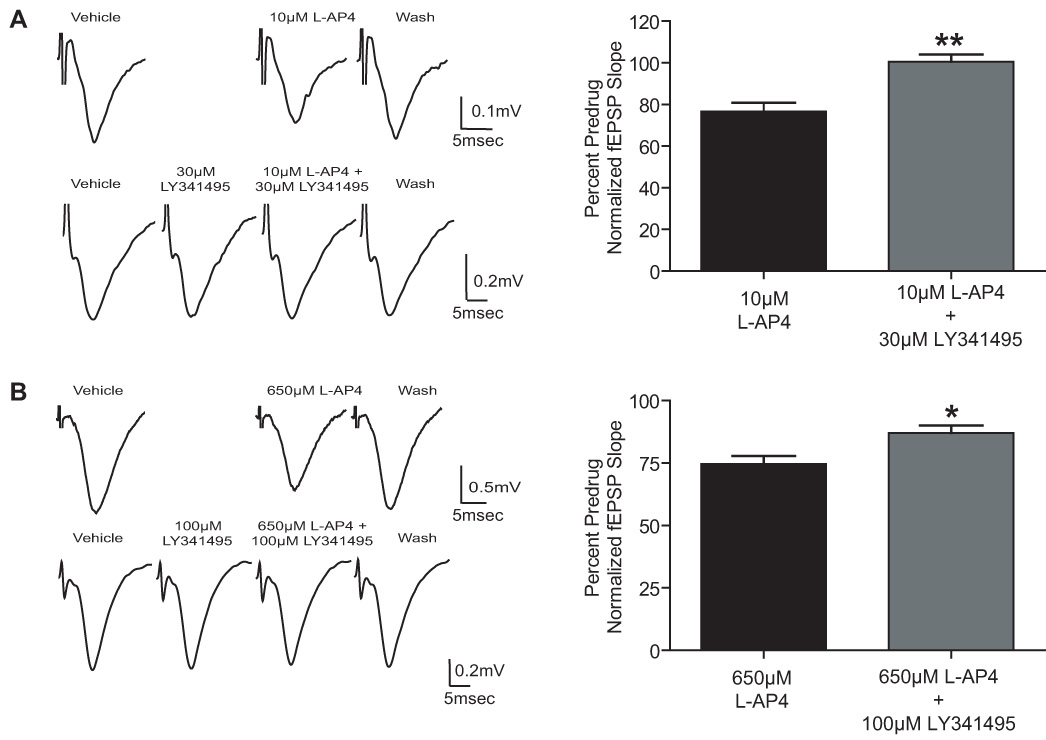
LY341495 antagonizes the effect of L-AP4 at the SC-CA1 synapse in the neonatal and adult rat hippocampus. A: Representative fEPSPs and bar graph depicting the effect of 10µM L-AP4 on fEPSP slope in the absence and presence of 30µM LY341495 on slices from neonatal rats. B: Representative fEPSPs and bar graph showing the effect of 650µM L-AP4 of fEPSP slope in the absence and presence of 100µM LY341495 on slices from adult rats. Data (mean ± SEM) are expressed as a percentage of predrug value. N = 3–5 for each experiment; * P < 0.05, ** P < 0.01.
While LY341495 also inhibits group I and II mGluRs (see (Schoepp et al., 1999) for review), the possibility that L-AP4 may be acting through one of these receptors can be ruled out based on previous work showing that 1mM L-AP4 does not activate goup I or group II mGluRs (Gereau and Conn, 1995b). Furthermore, we have found that 500µM L-AP4 is not blocked by (+) MCPG, an antagonist that inhibits group I and group II mGluRs (Gereau and Conn, 1995a). Finally, neither mGluR2 nor mGluR3 are expressed in the CA1 region of the hippocampus (Shigemoto et al., 1997).and the group II mGluR agonists DCG-IV does not reduce synaptic transmission at the SC-CA1 synapse in 10–40 day old rats (Fitzjohn et al., 1999; Gereau and Conn, 1995a; Kamiya et al., 1996; Yoshino et al., 1996) at the SC-CA1 synapse. Therefore the ability of LY341495 to block responses to L-AP4 at the SC-CA1 synapse confirms a role for a group III mGluR in mediating these responses.
DCPG reduces synaptic transmission at the SC-CA1 synapse in neonatal, but not adult, rats
The data presented thus far are consistent with the hypothesis that there is a developmental regulation of group III mGluR-mediated regulation of transmission at the SC-CA1 synapse. If this is due to a developmental regulation of the mGluR subtypes involved in regulation of transmission at this synapse, these data are most consistent with a role for mGluR4 or mGluR8 (which have high affinity for L-AP4) in neonatal animals and mGluR7 (low affinity for L-AP4) in adults as well as neonates. Furthermore, the ability of 30 µM LY341495 to block these responses is most consistent with a role for mGluR8 in neonatal animals. To more directly test this hypothesis, we determined the effect of the mGluR8-selective agonist, (S)-DCPG. We tested the effects of multiple concentrations of DCPG at the SC-CA1 synapse in neonates and found that 1µM was sufficient to elicit a maximal reduction of synaptic transmission to 71±6.7% of control (Figure 4 A and C, p<0.05). In contrast, DCPG did not reduce fEPSP slopes in slices from adults even at concentrations as high as 10µM (Fig. 4B and D; 10µM DCPG, 96±6.4% of predrug, p>0.05). These data suggest that mGluR8 is the most likely group III mGluR subtype responsible for regulation of transmission at the SC-CA1 synapse in neonatal animals. Furthermore, these data suggest that mGluR8 is not likely to play a role in attenuating synaptic transmission in the adult SC-CA1 synapse.
Figure 4.
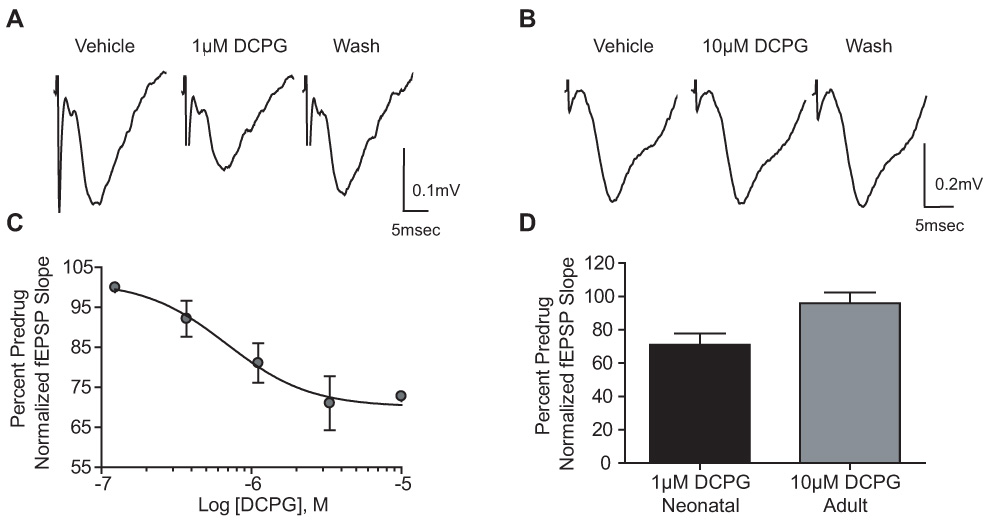
DCPG reduces synaptic transmission at the SC-CA1 synapse in the neonatal, but not adult rat hippocampus. A,D: Representative fEPSPs and bar graph depicting the effects of 1µM DCPG on fEPSP slope in hippocampal slices from neonatal rats. C: Concentration response curve for DCPG. Shown is the depression (mean ± SEM) of fEPSP slope induced by increasing concentrations of DCPG. Data are expressed as a percentage of predrug fEPSP slope. The concentration producing half-maximal depression is 688nM. N = 3–7 for each point. B,D: Representative fEPSPs and bar graph depicting the effects of 10µM DCPG in slices from adult rats. Data (mean ± SEM) are expressed as a percentage of predrug value. N = 4–7 for each experiment; * P < 0.05.
PHCCC does not potentiate the L-AP4 effect in neonatal or young adult rats
Another ligand that has recently been shown to have high selectivity for an individual group III mGluR subtype is PHCCC, a highly selective allosteric potentiator of mGluR4 (Canudas et al., 2004; Maj et al., 2003; Marino et al., 2003). As with other allosteric potentiators, PHCCC has no effect on mGluR4 when added alone but dramatically increases the potency of L-AP4 at activating this receptor. Furthermore, PHCCC enhances the effect of L-AP4 at synapses in the globus pallidus (Marino et al., 2003) and substantia nigra (Valenti et al., 2005) where mGluR4 is the predominant mGluR subtype involved in regulating synaptic transmission. To further test the hypothesis that mGluR8 is responsible for L-AP4-induced regulation of synaptic transmission at the SC-CA1 synapse in neonatal animals, we determined the effect of PHCCC (30µM) on the response to a concentration of L-AP4 that induces a clear but relatively small response. This concentration of PHCCC was chosen based on our previous findings that this concentration potentiates mGluR4-mediated responses at other synapses in brain slice preparations (Marino et al., 2003; Valenti et al., 2005). Consistent with predominant roles of mGluR8 and mGluR7 in neonatal and adult animals, PHCCC had no effect on the responses to submaximal concentrations of L-AP4 in slices from either age group (Fig. 5). These data suggest that mGluR4 is not likely involved in mediating L-AP4-induced attenuation of synaptic transmission in rats of either age group.
Figure 5.
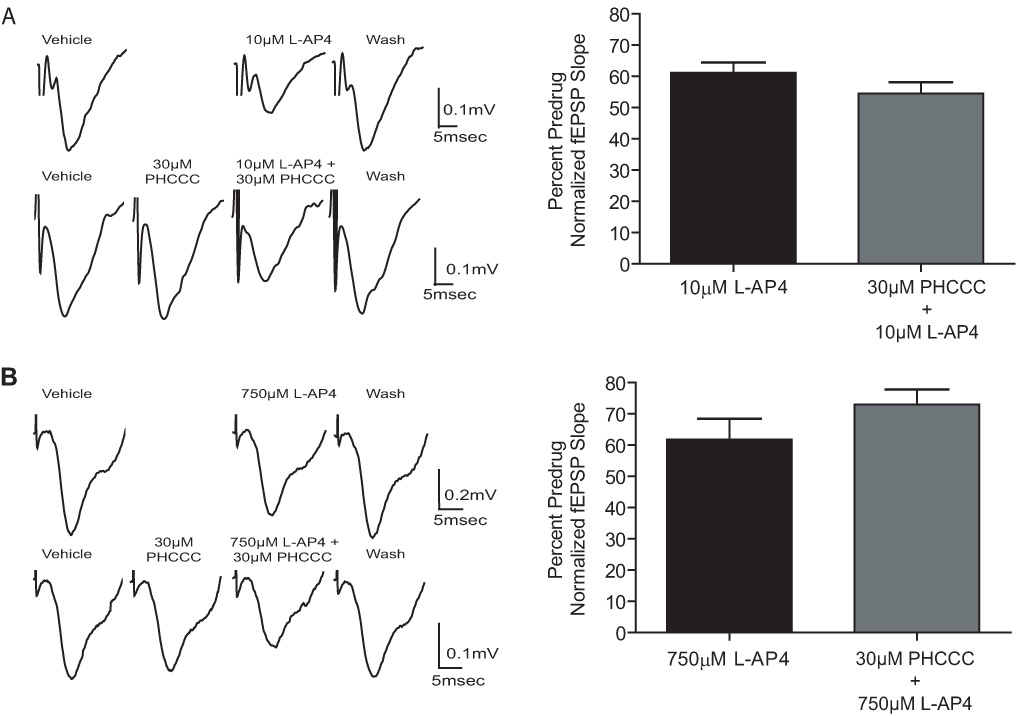
PHCCC does not potentiate the effects of L-AP4 at the SC-CA1 synapse in slices from neonatal and adult hippocampus. A: Representative fEPSPs and bar graph depicting the effects of 10µM L-AP4 on fEPSP slope in the presence and absence of 30µM PHCCC on slices from neonatal rats. 10µM L-AP4, 61±3.3% of predrug; 10µM L-AP4 + 30µM PHCCC, 54.5±3.6% of predrug; p>0.05. B: Representative fEPSPs and bar graph depicting the effects of 750µM L-AP4 on fEPSP slope in the presence and absence of 30µM PHCCC on slices from adult rats. 750µM L-AP4, 61.6±7% of predrug; 750µM L-AP5 + 30µM PHCCC, 71±3% of predrug; p>0.05. Data (mean ± SEM) are presented as percentage of predrug value. N = 3–4 for each experiment.
Z-Cylcopentyl AP4 does not reduce synaptic transmission
Z-Cylcopentyl AP4 is a conformationally constrained analog of L-AP4 that acts as a group III mGluR agonist with higher potency at mGluR4 than 8 and has no activity at mGluR7. As described above, PHCCC increases the potency of orthosteric agonists at activating mGluR4, suggesting that we may be able to increase the selectivity of Z-cylcopentyl AP4 for mGluR4 even further by combining this compound with PHCCC. To test this possibility, we determined the effect of 10µM PHCCC on the response of mGluR4 to multiple concentrations of Z-cylcopentyl AP4 in a cell line stably expressing the rat mGluR4 receptor. The assay used for these cell-based studies involves coupling of Gi/o-coupled receptors, such as the group III mGluRs, to the G protein gated Inwardly Rectifying Potassium Channel, GIRK (Niswender et al., 2006). Consistent with its effects on responses to other agonists, PHCCC increased the potency of Z-cylcopentyl AP4 by 3.39 fold and the efficacy by 49.73% at activating mGluR4 (Figure 6A). In contrast, PHCCC did not potentiate Z-cylcopentyl AP4’s responses on rat mGluR7 or rat mGluR8 (Figure 6B and C). Thus, Z-cylcopentyl AP4, used in the presence of PHCCC, provides a highly selective agonist of mGluR4 that fully activates mGluR4 at concentrations that have no effect on mGluR7 or mGluR8 (Fig. 6D).
Figure 6.
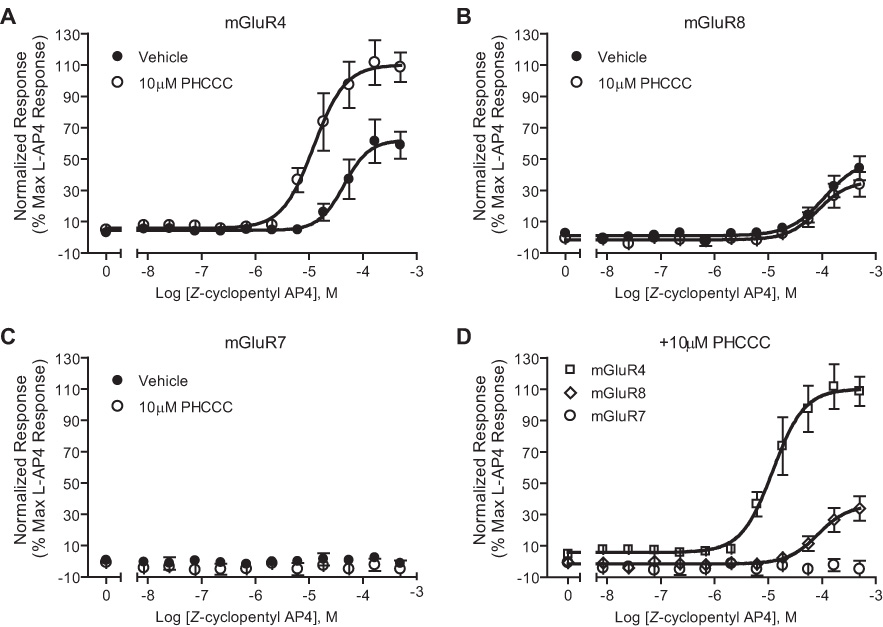
Concentration-response relationship of Z-cyclopentyl AP4 in the presence of PHCCC. Cells lines expressing the group III mGluRs were screened for activation by Z-cyclopentyl AP4. The addition of 10µM PHCCC potentiated the effects of Z-cyclopentyl AP4 at mGluR4 (A, D), but not mGluR8 (B, D). Z-Cyclopentyl AP4 has no effect on mGluR7 (C, D). mGluR4: Z-cyclopentyl AP4 EC50 = 49.13 ± 10.86µM versus Z-cyclopentyl AP4 + PHCCC EC50 = 14.5 ± 5.1 µM, p<0.01. mGluR8: (z)-cyclopentyl AP4 EC50 = 124.9 ± 21 µM versus Z-cyclopentyl AP4 + PHCCC EC50 = 85 ± 9.7 µM, p<0.05. Data (mean ± SEM) are expressed as a percentage of the maximal L-AP4 response for each individual cell line and were calculated based on means of three independent experiments performed in triplicate.
To further rule out a possible role for mGluR4 in regulating transmission at the SC-CA1 synapse, we determined the effects of Z-cylcopentyl AP4 in combination with PHCCC in both neonatal and adult rats. As expected, neither 50µM Z-cylcopentyl AP4 alone or in the presence of 30µM PHCCC caused a significant reduction of fEPSP slopes in slices from either neonatal or adult animals (Fig 7). These data lend further support to the hypothesis that mGluR4 is not involved in L-AP4-induced decreases in transmission at the SC-CA1 synapse of either neonates or adult rats.
Figure 7.
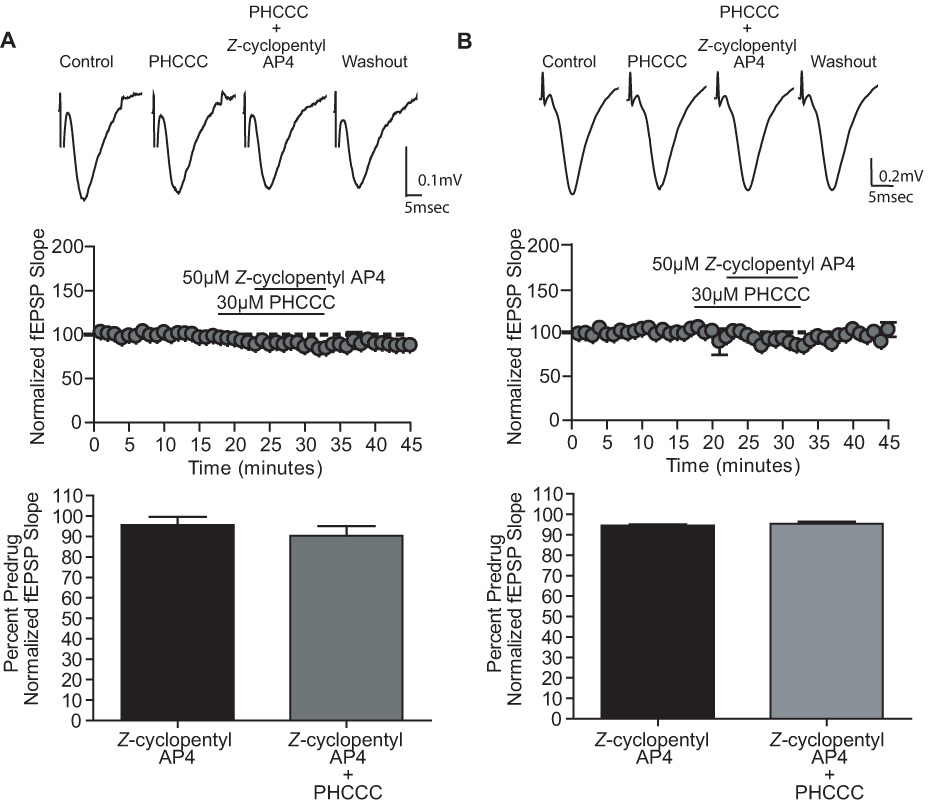
PHCCC does not potentiate the effects of Z-cyclopentyl AP4 at the SC-CA1 synapse in slices from neonatal and adult hippocampus. A (neonatal) and B (adult): Representative fEPSPs, time course and graphic representation showing the effects of Z-cyclopentyl AP4 (50µM) in the presence and absence of PHCCC (30µM). Neonatal: 50µM Z-cyclopentyl AP4 95.6±3.8% of predrug, 50µM Z-cyclopentyl AP4 + 30µM PHCCC 90±4.8% of predrug; p>0.05. Adult: 50µM Z-cyclopentyl AP4 94.5±0.5% of predrug, 50µM Z-cyclopentyl AP4 + 30µM PHCCC 95.4±1% of predrug; p>0.05. Data (mean ± SEM) are presented as percentage of predrug value. N = 3–6 for each experiment.
AMN082 does not reduce synaptic transmission at the SC-CA1 synapse
N,N’-dibenzhydrylethane-1,2-diamine (AMN082) was recently reported to be a selective allosteric agonist of mGluR7 (Conn and Niswender, 2006; Mitsukawa et al., 2005). In this previous paper, AMN082 elicited a full agonist response with nanomolar potency when measuring inhibition of cAMP accumulation or GTPγ[35S] binding in cells expressing mGluR7 (Mitsukawa et al., 2005; Suzuki et al., 2007). Previous studies suggest that allosteric modulators such as AMN802 may differentially modulate coupling of a receptor to different signaling pathways or in distinct cellular contexts. AMN802 has not been rigorously characterized at established mGluR7-mediated responses in native systems or across multiple cell populations and signaling pathways. Furthermore, it was recently reported that AMN082 does not activate mGluR7 in CHO cells as assessed by measuring mGluR7-mediated calcium responses in cells expressing the promiscuous G protein, Gα15 (Suzuki et al., 2007). This suggests that the pharmacology of AMN082 may be complex, and that this compound may not serve as a general agonist of mGluR7 across multiple responses and cell populations.
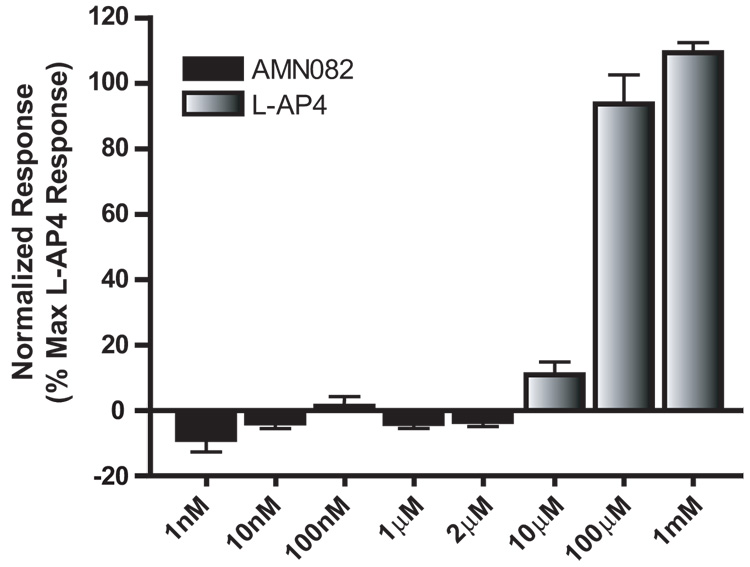
We tested the ability of AMN082 to decrease synaptic transmission at the SC-CA1 synapse. Surprisingly, AMN802 (1µM - 10µM) did not reduce synaptic transmission relative to baseline at the SC-CA1 synapse (Figure 8. 1µM AMN082 97.86 ± 4.99% of predrug; p>0.05). On the surface, these data may suggest that mGluR7 does not regulate transmission at the SC-CA1 synapse. Thus, we used the assay described above in which mGluR7 is coupled to the GIRK channel (Niswender et al., 2008), to examine the ability of AMN082 to activate mGluR7. In contrast to the robust response induced by L-AP4, AMN082 had no effect on GIRK mGluR7 as assessed by activation of GIRK at concentrations well above those shown by Mitsukawa et al. (2005) to inhibit cAMP responses in CHO cells (Figure 9).
Figure 8.
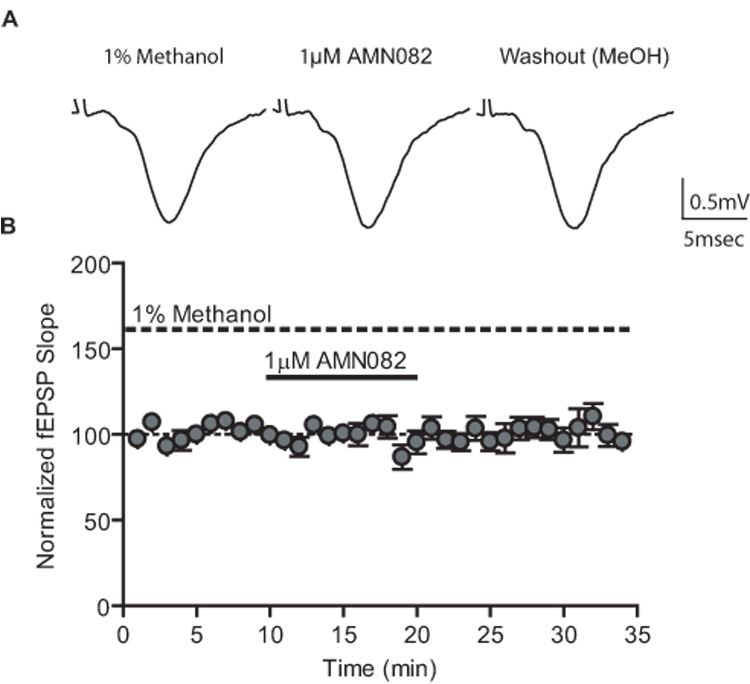
AMN082 does not reduce synaptic transmission at the SC-CA1 synapse of adult animals. Representative fEPSPs (inset) and time course showing the lack of effect of AMN082 (1µM). Data (mean ± SEM) are presented as percentage of predrug value. N = 5.
Figure 9.
AMN082 does not result in activation of GIRK channels in mGluR7/GIRK cells. Increasing concentrations of AMN082 or L-AP4 were added to mGluR7/GIRK cells and thallium flux was measured. L-AP4 EC50 = 43.3 ± 4.1 µM. Data (mean ± SEM) are expressed as a percentage of the maximal L-AP4 response and were calculated based on means of four independent experiments performed in triplicate.
Discussion
The present data suggest that there is a dramatic developmental regulation of presynaptic receptors involved in regulating transmission at the SC-CA1 synapse. Interestingly, while group III mGluRs provide robust modulation of transmission at this synapse throughout development, there is a developmental switch in the specific subtypes responsible for this effect. Specifically, our data suggest that mGluR7 and mGluR8 are the most likely candidates for receptors involved in regulating synaptic transmission at the SC-CA1 synapse in neonatal animals whereas mGluR7 is likely to play a predominant role in adults. A role for mGluR4 at the SC-CA1 synapse can be ruled out based on the finding that the response to L-AP4 is not potentiated by the mGluR4-selective potentiator PHCCC and is not mimicked by the mGluR4-selective agonist Z-cyclopentyl AP4 either in the presence or absence of PHCCC. This leaves mGluR7 and mGluR8 as the most likely candidates for regulation of transmission at this synapse. Consistent with this, the response at both ages is blocked by the mGluR antagonist LY341495, which has higher potencies for mGluR7 and mGluR8 than for mGluR4. The concentration response curve of L-AP4 is neonates is biphasic. 100 and 300µM L-AP4 result in an initial plateau of the CRC, however increasing the concentration of L-AP4 to a range that can activate mGluR7 results in a further decline in transmission, suggesting that mGluR7 may also be present in neonates, but is only activated when very high glutamate concentrations are achieved in the synapse. The conclusion that mGluR8 is likely the receptor mediating the response to low concentrations of L-AP4 in neonates is based on the finding that L-AP4 has relatively high potency in slices from neonatal animals and reduces transmission at concentrations that are not effective in activating mGluR7. Furthermore, the effect of L-AP4 is mimicked by relatively low concentrations of the mGluR8-selective agonist DCPG. In contrast, high concentrations (mM) of L-AP4 were required for activation of the presynaptic receptor in adults, which is consistent with the potency of L-AP4 at mGluR7. This response was not elicited by DCPG at concentrations ten times higher than the concentration required to regulate transmission in neonates. When taken together with the finding that the response in adults was blocked by LY31495 and was not altered by PHCCC or Z-cylcopentyl AP4, these data strongly support a role for mGluR7 in mediating this response.
Our findings are consistent with previous immunohistochemical studies of localization of Group III mGluRs. For instance, mGluR7a is abundantly distributed in all dendritic layers throughout the adult hippocampus (Bradley et al., 1996; Corti et al., 2002; Kosinski et al., 1999; Shigemoto et al., 1997). Additionally, immunogold studies reveal that mGluR7 is clustered in the presynaptic terminal at the site of neurotransmitter release in area CA1 of adult rats (Shigemoto et al., 1996). These data are consistent with a major role of mGluR7 as a presynaptic receptor involved in regulating transmission at the SC-CA1 synapse in adults. In contrast, mGluR4a is less abundantly expressed in adult hippocampus and is primarily localized to the inner third of the molecular layer of the dentate gyrus (Shigemoto et al., 1997), far removed from the SC-CA1 synapse. Likewise, mGluR8 immunoreactivity is largely absent from area CA1 of adults but is found in the dentate gyrus lateral perforant path terminal zone (Shigemoto et al., 1997). Fewer studies have been performed in neonatal animals. Taken together, these studies are consistent with our findings and support the hypothesis that there is a developmental change in the group III mGluR that modulates synaptic transmission at the SC-CA1 synapse.
Despite the converging evidence from pharmacological and anatomical studies supporting a role of mGluR7 in regulating transmission at the SC-CA1 synapse in adult animals, we found that AMN082, a compound recently reported to be an mGluR7-selective agonist, did not inhibit synaptic transmission at the SC-CA1 synapse. Furthermore, we found that AMN082 was without effect in cell-based assays of mGluR7-induced activation of the GIRK potassium channel. A recent study also reported that AMN082 does not induce calcium mobilization downstream of mGluR7 in a cell line coexpressing the promiscuous G protein Gα15 (Suzuki et al., 2007). This is in contrast to previous reports demonstrating activity of AMN082 in reduction of cAMP and GTPγ[35S] binding assays (Mitsukawa et al., 2005; Suzuki et al., 2007). These data suggest that the effects of AMN082 may be context-specific. Thus, agonist activity of this compound at mGluR7 could depend on the G proteins or second messenger systems that are activated in a particular cell type or on other cell or pathway-specific factors. Previous studies have led to the suggestion that activation of mGluR7 leads to inhibition of P/Q-type calcium channel activity in hippocampal nerve terminals by βγ subunits (Martin et al., 2007; Perroy et al., 2000). Since activation of GIRK channels also occurs through activation of βγ subunits (Saugstad et al., 1996), it is interesting that we observed a correlation between the lack of effect of AMN082 in a cell-based system that relies upon βγ-mediated signaling after mGluR7 activation and synaptic transmission. In future studies, it will be important to develop a further understanding of the mechanisms by which AMN082 activates mGluR7 and the specific conditions under which this activation can occur. Furthermore, it will be important to exercise caution in using AMN082 as a general use selective mGluR7 agonist in more complex systems, such as behavioral studies.
The current finding that there is a developmental regulation in the mGluR subtype responsible for regulation of transmission at the SC-CA1 synapse raises a major question as to the functional impact of a developmental regulation of the subtype of group III mGluRs involved in regulating transmission at this important hippocampal synapse. At a qualitative level, both receptors serve similar roles in reducing excitatory synaptic transmission. Importantly, the relative affinities of group III mGluRs for L-AP4 are reflected by the affinities of these receptors for glutamate. Thus, glutamate activates mGluR8 at low µM concentrations whereas mM concentrations of glutamate are required for activation of mGluR7. Thus, it is possible that these receptors play fundamentally different roles in vivo and in the context of ongoing synaptic transmission. It is conceivable that the developmental switch in expression of a receptor with high affinity for glutamate to one with low affinity suggests that there is a requirement for tighter control of glutamate release during early stages of synapse formation. To a great extent, the formation of inhibitory synapses occurs postnatally in the developing rat brain (Fritschy et al., 1994). Particularly in the CA1 subfield of the rat hippocampus, the development of GABAergic inhibitory control is ongoing in animals up to postnatal day 10 (Swann et al., 1989) and some studies suggest that inhibitory responses can not be evoked until p14–17 (Dunwiddie, 1981). It is possible that this lack of inhibitory control increases vulnerability to excessive excitatory activity and expression of a receptor with high sensitivity to glutamate concentrations during these early stages of postnatal development provides a protective mechanism. However, it is also possible that mGluR8 and mGluR7 play fundamentally different roles in neonates and adult animals. For instance, as mentioned above, mGluR7 is specifically localized in the active zone of glutamatergic synapses in area CA1 of adult animals (Ohishi et al., 1995; Shigemoto et al., 1996). This clear synaptic localization would place mGluR7 in a position where it could be exposed to relatively high concentrations of glutamate during ongoing synaptic transmission. It has been suggested that this localization, paired with the low potency of glutamate at this receptor, may allow mGluR7 to reduce glutamate release when action potentials arrive at high-frequency, only allowing glutamate to be released when the cell is firing at a lower frequency, thus acting as a low-pass filter (Scanziani et al., 1997; Shigemoto et al., 1996). The synaptic localization of mGluR8 in neonatal CA1 is not known. However, it is possible that this receptor plays a fundamentally different role and that the high affinity for glutamate renders it sensitive to changes in ambient extrasynaptic glutamate concentrations. In future studies, it will be important to determine the synaptic localization of mGluR8 in neonatal hippocampus and determine the precise roles of group III mGluRs in regulating synaptic transmission at different developmental stages.
Aknowledgements
The authors thank Dr. Rodney Johnson (University of Minnesota, Minneapolis, MN) for the Z-cyclopentyl AP4. The authors also thank Dr. Huai Hu Chang and Dr. Lily Jan (University of San Francisco, San Francisco, CA) for stable HEK/GIRK cells and K. Johnson and C. Kim for stable transfection of HEK/GIRK cells with mGluR constructs.
This work was supported by grants from National Institute of Mental Health and National Institute of Neurological Disorders and Stroke. Vanderbilt is a site in the National Institutes of Health-supported Molecular Libraries Screening Center Network.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Baskys A, Malenka RC. Agonists at metabotropic glutamate receptors presynaptically inhibit EPSCs in neonatal rat hippocampus. J Physiol. 1991;444:687–701. doi: 10.1113/jphysiol.1991.sp018901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradley SR, Levey AI, Hersch SM, Conn PJ. Immunocytochemical localization of group III metabotropic glutamate receptors in the hippocampus with subtype-specific antibodies. J Neurosci. 1996;16:2044–2056. doi: 10.1523/JNEUROSCI.16-06-02044.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradley SR, Rees HD, Yi H, Levey AI, Conn PJ. Distribution and developmental regulation of metabotropic glutamate receptor 7a in rat brain. J Neurochem. 1998;71:636–645. doi: 10.1046/j.1471-4159.1998.71020636.x. [DOI] [PubMed] [Google Scholar]
- Canudas AM, Di Giorgi-Gerevini V, Iacovelli L, Nano G, D'Onofrio M, Arcella A, Giangaspero F, Busceti C, Ricci-Vitiani L, Battaglia G, Nicoletti F, Melchiorri D. PHCCC, a specific enhancer of type 4 metabotropic glutamate receptors, reduces proliferation and promotes differentiation of cerebellar granule cell neuroprecursors. J Neurosci. 2004;24:10343–10352. doi: 10.1523/JNEUROSCI.3229-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conn PJ, Niswender CM. mGluR7's lucky number. Proc Natl Acad Sci U S A. 2006;103:251–252. doi: 10.1073/pnas.0510051103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conn PJ, Pin JP. Pharmacology and functions of metabotropic glutamate receptors. Annu Rev Pharmacol Toxicol. 1997;37:205–237. doi: 10.1146/annurev.pharmtox.37.1.205. [DOI] [PubMed] [Google Scholar]
- Corti C, Aldegheri L, Somogyi P, Ferraguti F. Distribution and synaptic localisation of the metabotropic glutamate receptor 4 (mGluR4) in the rodent CNS. Neuroscience. 2002;110:403–420. doi: 10.1016/s0306-4522(01)00591-7. [DOI] [PubMed] [Google Scholar]
- Crooks SL, Robinson MB, Koerner JF, Johnson RL. Cyclic analogues of 2-amino-4-phosphonobutanoic acid (APB) and their inhibition of hippocampal excitatory transmission and displacement of [3H]APB binding. J Med Chem. 1986;29:1988–1995. doi: 10.1021/jm00160a031. [DOI] [PubMed] [Google Scholar]
- Dunwiddie TV. Age-related differences in the in vitro rat hippocampus. Development of inhibition and the effects of hypoxia. Dev Neurosci. 1981;4:165–175. doi: 10.1159/000112753. [DOI] [PubMed] [Google Scholar]
- Edl J, Banko J, Myers K, Niswender C, Conn P. Developemental Differences in Group III mGluR Regulation of Synaptic Transmission in Area CA1 of the Rat Hippocampus. Society for Neuroscience Abstracts. 2006 Program Number 626.2. [Google Scholar]
- Fitzjohn SM, Kingston AE, Lodge D, Collingridge GL. DHPG-induced LTD in area CA1 of juvenile rat hippocampus; characterisation and sensitivity to novel mGlu receptor antagonists. Neuropharmacology. 1999;38:1577–1583. doi: 10.1016/s0028-3908(99)00123-9. [DOI] [PubMed] [Google Scholar]
- Fritschy JM, Paysan J, Enna A, Mohler H. Switch in the expression of rat GABAA-receptor subtypes during postnatal development: an immunohistochemical study. J Neurosci. 1994;14:5302–5324. doi: 10.1523/JNEUROSCI.14-09-05302.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gereau RWt, Conn PJ. Multiple presynaptic metabotropic glutamate receptors modulate excitatory and inhibitory synaptic transmission in hippocampal area CA1. J Neurosci. 1995a;15:6879–6889. doi: 10.1523/JNEUROSCI.15-10-06879.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gereau RWt, Conn PJ. Roles of specific metabotropic glutamate receptor subtypes in regulation of hippocampal CA1 pyramidal cell excitability. J Neurophysiol. 1995b;74:122–129. doi: 10.1152/jn.1995.74.1.122. [DOI] [PubMed] [Google Scholar]
- Kamiya H, Ozawa S. Dual mechanism for presynaptic modulation by axonal metabotropic glutamate receptor at the mouse mossy fibre-CA3 synapse. J Physiol. 1999;518(Pt 2):497–506. doi: 10.1111/j.1469-7793.1999.0497p.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamiya H, Shinozaki H, Yamamoto C. Activation of metabotropic glutamate receptor type 2/3 suppresses transmission at rat hippocampal mossy fibre synapses. J Physiol. 1996;493(Pt 2):447–455. doi: 10.1113/jphysiol.1996.sp021395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kingston AE, Ornstein PL, Wright RA, Johnson BG, Mayne NG, Burnett JP, Belagaje R, Wu S, Schoepp DD. LY341495 is a nanomolar potent and selective antagonist of group II metabotropic glutamate receptors. Neuropharmacology. 1998;37:1–12. doi: 10.1016/s0028-3908(97)00191-3. [DOI] [PubMed] [Google Scholar]
- Koerner JF, Cotman CW. Micromolar L-2-amino-4-phosphonobutyric acid selectively inhibits perforant path synapses from lateral entorhinal cortex. Brain Res. 1981;216:192–198. doi: 10.1016/0006-8993(81)91288-9. [DOI] [PubMed] [Google Scholar]
- Koerner JF, Cotman CW. Response of Schaffer collateral-CA 1 pyramidal cell synapses of the hippocampus to analogues of acidic amino acids. Brain Res. 1982;251:105–115. doi: 10.1016/0006-8993(82)91278-1. [DOI] [PubMed] [Google Scholar]
- Kosinski CM, Risso Bradley S, Conn PJ, Levey AI, Landwehrmeyer GB, Penney JB, Jr, Young AB, Standaert DG. Localization of metabotropic glutamate receptor 7 mRNA and mGluR7a protein in the rat basal ganglia. J Comp Neurol. 1999;415:266–284. [PubMed] [Google Scholar]
- Lanthorn TH, Ganong AH, Cotman CW. 2-Amino-4-phosphonobutyrate selectively blocks mossy fiber-CA3 responses in guinea pig but not rat hippocampus. Brain Res. 1984;290:174–178. doi: 10.1016/0006-8993(84)90750-9. [DOI] [PubMed] [Google Scholar]
- Macek TA, Winder DG, Gereau RWt, Ladd CO, Conn PJ. Differential involvement of group II and group III mGluRs as autoreceptors at lateral and medial perforant path synapses. J Neurophysiol. 1996;76:3798–3806. doi: 10.1152/jn.1996.76.6.3798. [DOI] [PubMed] [Google Scholar]
- Maj M, Bruno V, Dragic Z, Yamamoto R, Battaglia G, Inderbitzin W, Stoehr N, Stein T, Gasparini F, Vranesic I, Kuhn R, Nicoletti F, Flor PJ. (−)-PHCCC, a positive allosteric modulator of mGluR4: characterization, mechanism of action, and neuroprotection. Neuropharmacology. 2003;45:895–906. doi: 10.1016/s0028-3908(03)00271-5. [DOI] [PubMed] [Google Scholar]
- Marino MJ, Valenti O, O'Brien JA, Williams DL, Jr, Conn PJ. Modulation of inhibitory transmission in the rat globus pallidus by activation of mGluR4. Ann N Y Acad Sci. 2003;1003:435–437. doi: 10.1196/annals.1300.045. [DOI] [PubMed] [Google Scholar]
- Marino MJ, Williams DL, Jr, O'Brien JA, Valenti O, McDonald TP, Clements MK, Wang R, DiLella AG, Hess JF, Kinney GG, Conn PJ. Allosteric modulation of group III metabotropic glutamate receptor 4: a potential approach to Parkinson's disease treatment. Proc Natl Acad Sci U S A. 2003;100:13668–13673. doi: 10.1073/pnas.1835724100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin R, Torres M, Sanchez-Prieto J. mGluR7 inhibits glutamate release through a PKC-independent decrease in the activity of P/Q-type Ca(2+) channels and by diminishing cAMP in hippocampal nerve terminals. Eur J Neurosci. 2007;26:312–322. doi: 10.1111/j.1460-9568.2007.05660.x. [DOI] [PubMed] [Google Scholar]
- Mitsukawa K, Yamamoto R, Ofner S, Nozulak J, Pescott O, Lukic S, Stoehr N, Mombereau C, Kuhn R, McAllister KH, van der Putten H, Cryan JF, Flor PJ. A selective metabotropic glutamate receptor 7 agonist: activation of receptor signaling via an allosteric site modulates stress parameters in vivo. Proc Natl Acad Sci U S A. 2005;102:18712–18717. doi: 10.1073/pnas.0508063102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niswender C, Johnson K, Luo Q, Ayala J, Kim C, Conn P, Weaver C. A novel HTS-compatible assay to measure Gi/o-linked G protein coupled receptor function using thallium flux through GIRK channels. Mol Pharmacol. 2008 doi: 10.1124/mol.107.041053. submitted. [DOI] [PubMed] [Google Scholar]
- Niswender CM, Myers KA, Williams R, Ayala JE, Luo Q, Saleh S, Jones CK, Weaver CD, Orton D, Conn PJ. Permissive antagonism induced by novel allosteric antagonists of metabotropic glutamate receptor 7. Neuropsychopharmacology. 31 Supplement 1:S127. [Google Scholar]
- Ohishi H, Akazawa C, Shigemoto R, Nakanishi S, Mizuno N. Distributions of the mRNAs for L-2-amino-4-phosphonobutyrate-sensitive metabotropic glutamate receptors, mGluR4 and mGluR7, in the rat brain. J Comp Neurol. 1995;360:555–570. doi: 10.1002/cne.903600402. [DOI] [PubMed] [Google Scholar]
- Perroy J, Prezeau L, De Waard M, Shigemoto R, Bockaert J, Fagni L. Selective blockade of P/Q-type calcium channels by the metabotropic glutamate receptor type 7 involves a phospholipase C pathway in neurons. J Neurosci. 2000;20:7896–7904. doi: 10.1523/JNEUROSCI.20-21-07896.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saugstad JA, Segerson TP, Westbrook GL. Metabotropic glutamate receptors activate G-protein-coupled inwardly rectifying potassium channels in Xenopus oocytes. J Neurosci. 1996;16:5979–5985. doi: 10.1523/JNEUROSCI.16-19-05979.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scanziani M, Salin PA, Vogt KE, Malenka RC, Nicoll RA. Use-dependent increases in glutamate concentration activate presynaptic metabotropic glutamate receptors. Nature. 1997;385:630–634. doi: 10.1038/385630a0. [DOI] [PubMed] [Google Scholar]
- Schoepp DD, Jane DE, Monn JA. Pharmacological agents acting at subtypes of metabotropic glutamate receptors. Neuropharmacology. 1999;38:1431–1476. doi: 10.1016/s0028-3908(99)00092-1. [DOI] [PubMed] [Google Scholar]
- Shigemoto R, Kinoshita A, Wada E, Nomura S, Ohishi H, Takada M, Flor PJ, Neki A, Abe T, Nakanishi S, Mizuno N. Differential presynaptic localization of metabotropic glutamate receptor subtypes in the rat hippocampus. J Neurosci. 1997;17:7503–7522. doi: 10.1523/JNEUROSCI.17-19-07503.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shigemoto R, Kulik A, Roberts JD, Ohishi H, Nusser Z, Kaneko T, Somogyi P. Target-cell-specific concentration of a metabotropic glutamate receptor in the presynaptic active zone. Nature. 1996;381:523–525. doi: 10.1038/381523a0. [DOI] [PubMed] [Google Scholar]
- Suzuki G, Tsukamoto N, Fushiki H, Kawagishi A, Nakamura M, Kurihara H, Mitsuya M, Ohkubo M, Ohta H. In vitro pharmacological characterization of novel isoxazolopyridone derivatives as allosteric metabotropic glutamate receptor 7 (mGluR7) antagonists. J Pharmacol Exp Ther. 2007 doi: 10.1124/jpet.107.124701. [DOI] [PubMed] [Google Scholar]
- Swann JW, Brady RJ, Martin DL. Postnatal development of GABA-mediated synaptic inhibition in rat hippocampus. Neuroscience. 1989;28:551–561. doi: 10.1016/0306-4522(89)90004-3. [DOI] [PubMed] [Google Scholar]
- Thomas NK, Wright RA, Howson PA, Kingston AE, Schoepp DD, Jane DE. (S)-3,4-DCPG, a potent and selective mGlu8a receptor agonist, activates metabotropic glutamate receptors on primary afferent terminals in the neonatal rat spinal cord. Neuropharmacology. 2001;40:311–318. doi: 10.1016/s0028-3908(00)00169-6. [DOI] [PubMed] [Google Scholar]
- Valenti O, Mannaioni G, Seabrook GR, Conn PJ, Marino MJ. Group III metabotropic glutamate-receptor-mediated modulation of excitatory transmission in rodent substantia nigra pars compacta dopamine neurons. J Pharmacol Exp Ther. 2005;313:1296–1304. doi: 10.1124/jpet.104.080481. [DOI] [PubMed] [Google Scholar]
- Vignes M, Clarke VR, Davies CH, Chambers A, Jane DE, Watkins JC, Collingridge GL. Pharmacological evidence for an involvement of group II and group III mGluRs in the presynaptic regulation of excitatory synaptic responses in the CA1 region of rat hippocampal slices. Neuropharmacology. 1995;34:973–982. doi: 10.1016/0028-3908(95)00093-l. [DOI] [PubMed] [Google Scholar]
- Yoshino M, Sawada S, Yamamoto C, Kamiya H. A metabotropic glutamate receptor agonist DCG-IV suppresses synaptic transmission at mossy fiber pathway of the guinea pig hippocampus. Neurosci Lett. 1996;207:70–72. doi: 10.1016/0304-3940(96)12486-1. [DOI] [PubMed] [Google Scholar]
- Zhai J, Tian MT, Wang Y, Yu JL, Koster A, Baez M, Nisenbaum ES. Modulation of lateral perforant path excitatory responses by metabotropic glutamate 8 (mGlu8) receptors. Neuropharmacology. 2002;43:223–230. doi: 10.1016/s0028-3908(02)00087-4. [DOI] [PubMed] [Google Scholar]