Abstract
Heterogeneous data are a common problem in meta-analysis. John Ioannidis, Nikolaos Patsopoulos, and Hannah Rothstein show that final synthesis is possible and desirable in most cases
Some systematic reviews simply assemble the eligible studies without performing meta-analysis. This may be a legitimate choice. However, an interesting situation arises when reviews present forest plots (quantitative effects and uncertainty per study) but do not calculate a summary estimate (the diamond at the bottom). These reviews imply that it is important to visualise the quantitative data but final synthesis is inappropriate. For example, a review of sexual abstinence programmes for HIV prevention claimed that owing to “data unavailability, lack of intention-to-treat analyses, and heterogeneity in programme and trial designs… a statistical meta-analysis would be inappropriate.”1 As we discuss, options almost always exist for quantitative synthesis and sometimes they may offer useful insights. Reviewers and clinicians should be aware of these options, reflect carefully on their use, and understand their limitations.
Why meta-analysis is avoided
Of the 1739 systematic reviews that included at least one forest plot with at least two studies in issue 4 of the Cochrane Database of Systematic Reviews (2005), 135 reviews (8%) had 559 forest plots with no summary estimate.
The reasons provided for avoiding quantitative synthesis typically revolved around heterogeneity (table 1). The included studies were thought to be too different, either statistically or in clinical (including methodological) terms. Differences in interventions, metrics, outcomes, designs, participants, and settings were implied.
Table 1.
Reasons for not showing summary estimates in forest plots from systematic reviews in Cochrane database 2005 issue 4
| Reason | No (%) of systematic reviews (n=135)* |
|---|---|
| Statistical heterogeneity too high | 32 (24) |
| Different interventions compared | 41 (30) |
| Different metrics or outcomes evaluated | 26 (19) |
| Different metric of same outcome | 7 |
| Different outcome | 20 |
| Different study designs | 21 (16) |
| Non-randomised studies | 3 |
| Other design issues | 18 |
| Different study participants, settings | 21 (16) |
| Data with many counts per participant | 5 (4) |
| Data too limited | 11 (8) |
| Clinical heterogeneity (not otherwise specified) | 5 (4) |
| Synthesis considered inappropriate (not specified why) | 3 (2) |
| Non-normality of data | 1 (1) |
| No reason given | 10 (7) |
| Artefact† | 3 (2) |
| Quantitative synthesis given in text | 7 (5) |
*For another 7 reviews forest plots listed the names of two or more studies but data were available for only one or no study; 4 reviews listed as withdrawn are not included. Several reviews gave more than one reason without clarifying which was the most important. In these cases, all reasons are counted. However, when only one reason specifically was used to decide whether data synthesis would be appropriate, only this reason is counted, regardless of whether several other reasons were then secondarily probed.
†Meta-analyses of individual level data that used the RevMan forest plot function to show subgroup summary estimates.
How large is too large heterogeneity?
This question of lumping versus splitting is difficult to answer objectively for clinical heterogeneity. Logic models based on the PICO (population-intervention–comparator-outcomes) framework may help to deal with the challenges of deciding what to include and what not. Still, different reviewers, readers, and clinicians may disagree on the (dis)similarity of interventions, outcomes, designs, participant characteristics, and settings.
No widely accepted quantitative measure exists to grade clinical heterogeneity. Nevertheless, it may be better to examine clinical differences in a meta-analysis rather than use them as a reason for not conducting one. For example, a review identified 40 trials of diverse interventions to prevent falls in elderly people.2 Despite large diversity in the trials, the authors did a meta-analysis and also examined the effectiveness of different interventions. The analysis suggested that evidence was stronger for multifactorial risk assessment and management programmes and exercise and more inconclusive for environmental modifications and education.
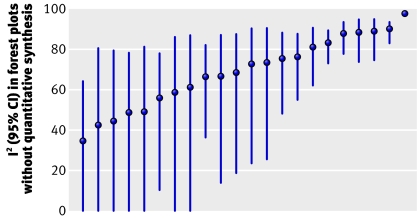
Statistical heterogeneity can be measured—for example, by calculating I2 and its uncertainty.3 4 5 I2, the proportion of variation between studies not due to chance, takes values from 0 to 100%. In the 22 forest plots including four or more studies that avoided synthesis because of heterogeneity, I2 ranged between 35% and 98% with a median of 71% (figure 1). Yet, 86 of the 1011 forest plots where reviewers had no hesitation in performing meta-analysis had I2 exceeding 71%.5 The lower 95% confidence limit of I2 was <25% in 11 of the 22 non-summarised forest plots—that is, for half of them we cannot exclude that statistical heterogeneity is limited. Therefore, even for statistical heterogeneity, there is substantial variability in what different reviewers consider too much. Statistical heterogeneity alone is a weak and inconsistently used argument for avoiding quantitative synthesis.
Fig 1 I2 point estimates and 95% CI for forest plots with at least 4 studies and no quantitative synthesis because of perceived high statistical heterogeneity
Potential methods for use in heterogeneity
Table 2 provides methodological approaches to quantitative synthesis of data that some researchers may deem unsuitable for meta-analysis. It is unknown whether researchers preparing systematic reviews were aware of these methods but thought that they were inapplicable; were aware of their existence but lacked the necessary experience and software; or were unaware of their existence. Detailed discussion of methods is beyond our scope here, but we present the principal options and caveats and provide references for interested readers. Some methods are experimental and extra caution is needed.
Table 2.
Methodological approaches to consider in the synthesis of heterogeneous data
| Problem | Possible methodological solution | Selected key caveats |
|---|---|---|
| High statistical heterogeneity* | Random effects | Does not explain heterogeneity, small study effects, limited data |
| Meta-regression | Choice of variables, ecological fallacy, limited data | |
| Bayesian meta-analysis | Prior specification | |
| Bayesian meta-regression | Similar to meta-regression and Bayesian meta-analysis | |
| Meta-analysis of individual level data | Unavailable individual level information | |
| Different interventions compared | Merge interventions in same class | Unrecognised heterogeneity |
| Network meta-analysis | Inconsistency in direct versus indirect comparisons | |
| Different metrics of same outcome | Conversion formulas | Difficulties in clinical interpretation |
| Different outcomes, same construct | Standardised effects | Difficulties in clinical interpretation |
| Different outcomes | Meta-analysis of multiple outcomes | Specification of correlations |
| Observational data | Generalised evidence synthesis | Spurious precision, confounding, selective reporting |
| Cluster randomised trials | Account for clustering correlation | Unavailable sufficient information |
| Crossover trials | Account for period or carry-over effect | Unavailable sufficient information |
| Other study design issues | Same as for high statistical heterogeneity | As for high statistical heterogeneity above |
| Different participants or settings | Same as for high statistical heterogeneity | As for high statistical heterogeneity above |
| Many counts per participant | Meta-analysis of multiple period or follow-up | Unavailable sufficient information |
| Limited data | Standard meta-analysis methods | Caution needed as for any meta-analysis |
Popular software such as RevMan can accommodate only random effects calculations, while Comprehensive Meta-Analysis also accommodates simple meta-regressions. Bayesian models and models of multiple treatments or outcomes can be run in WinBugs. Most models can also be run in STATA or R.
*The approach used for high statistical heterogeneity may also be applicable to situations where clinical heterogeneity is considered high because of differences in interventions, metrics, outcomes, designs, participants, or settings.
Models that can accommodate statistical heterogeneity between studies include traditional random effects (models that assume that different studies have different true treatment effects),6 meta-regressions (regressions that examine whether the treatment effect is related to one or more characteristics of the studies or patients),7 and bayesian methods (methods that combine various prior assumptions with the observed data).8 Random effects do not explain the heterogeneity: they distort estimates when large versus smaller studies differ in results and smaller studies are more biased, and they can be unstable with limited evidence;9 meta-regressions may suffer from post hoc selection of variables, the ecological fallacy, and poor performance with few studies;10 and bayesian results may depend on prior specifications.8 Meta-analysis of data at the individual level may permit fuller exploration of heterogeneity, but these data are usually unavailable.11
The availability of multiple interventions for the same condition and indication is increasingly common. Different regimens may be merged in common groups, but differences in treatment effects of merged regimens may remain unrecognised. Multiple treatments meta-analysis could be used to examine all the different treatments used for a given condition. For example, 242 chemotherapy trials are available covering 137 different regimens for advanced colorectal cancer.12 The number of possible comparisons is prohibitive. A meta-analysis grouped these regimens into 12 treatment types and then performed a network analysis that evaluated their relative effectiveness. Instead of taking one comparison at a time, the network considered concomitantly all the data from all relevant comparisons. Networks integrate information from both direct and indirect comparisons of different treatments.13 14 15 Main caveats include possible inconsistency in results between direct and indirect comparisons and the still limited experience on networks.13 14 15 16
Clinical trials on the same topic also commonly use many different outcomes. Meta-analysis of one outcome at a time offers a fragmented picture. Some outcomes simply differ in their measures—for example, global clinical improvement measured on a continuous scale or as a binary end point (yes/no). Continuous scales can be converted into binary ones and standardised metrics (popular in the social sciences)17 can accommodate different outcomes that measure the same construct (such as various psychometric scales). However, for medical applications, many clinicians think that anything other than plain absolute risk is insufficiently intelligible to inform practice and policy.18 19 Finally, some outcomes may represent truly different end points with partial correlation among themselves (for example, serum creatinine, creatinine clearance, progression to end stage renal disease, initiation of renal replacement therapy) and multivariate meta-analysis models can cater for two or more correlated outcomes.20 21 22 Such models borrow strength from all the available outcomes across trials. The main caveats are specification of correlations and sparse data.
The combination of data from randomised and non-randomised studies is possible using traditional meta-analysis models. The main caveats are the spurious precision,23 confounding, and potentially stronger selective reporting biases in observational studies.24 However, the generalised synthesis of both randomised and non-randomised studies on the same topic may offer complementary information.25 26 27 Other designs that require special care in meta-analysis include cluster28 and crossover trials.29
Appropriate methods also exist for synthesising data when each participant may count many times in the calculations (multiple periods at risk or multiple follow-up data).17 30
The authors of several systematic reviews state only that “data synthesis is inappropriate” or allude vaguely to “clinical heterogeneity.” Specifying the reasons would improve transparency of the implicit judgments. Finally, some reviews argue that data are too limited. However, meta-analysis is feasible even with two studies. For most medical questions, only few studies exist. Limited data typically yield uncertain estimates, but the quantitative accuracy of meta-analysis may actually be a reason to avoid narrative interpretation without synthesis. Limited data may also result from asking questions that are too narrow, trying to make data too similar before inclusion in the same forest plot. Forced similarity may fragment information; it is almost unavoidable that trials will differ in at least minor ways.
To synthesise or not?
If the limitations of these methods are properly acknowledged, the use of quantitative synthesis may be preferable to qualitative interpretation of the results, or hidden quasi-quantitative analysis—for example, judging studies based on P values of single studies being above or below 0.05. Such an approach can actually lead to the wrong conclusion, especially when statistical power is low.31 For example, if an intervention is effective but two studies are done with 40% power each, the chance of both of them getting a significant result is only 16%.
More complex “home made” qualitative rules may further compound the methodological problems. This applies not only to reviews that avoid the final synthesis but also to entirely narrative reviews without any forest plots. For example, the reviewers of interventions to promote physical activity in children and adolescents “used scores to indicate effectiveness—that is, whether there was no difference in effect between control and intervention group (0 score), a positive or negative trend (+ or −), or a significant difference (P<0.05) in favour of the intervention or control group (++ or −−, respectively) . . . If at least two thirds (66.6%) of the relevant studies were reported to have significant results in the same direction then we considered the overall results to be consistent.”32 Such rules have poor performance validity.
Meta-analysis is often understood solely as a means of combining information to produce a single overall estimate of effect. However, one of its advantages is to assess, examine, and model the consistency of effects and improve understanding of moderator variables, boundary conditions, and generalisability.8 33 Different patients and different studies are unavoidably heterogeneous. This diversity and the uncertainty associated with it should be explored whenever possible. Obtaining estimates of treatment effect (rather than simple narrative evaluations) may allow more rational decisions about the use of interventions in specific patients or settings. More sophisticated methods may also capture and model uncertainties more fully and thus may actually reach more conservative conclusions than more naive approaches. However, it is then essential that their assumptions and limitations are clearly stated and inferences drawn cautiously. Any meta-analysis method, simple or advanced, may be misleading, if we don’t understand how it works.
Summary points
Some reviews extract numerical data and generate forest plots but avoid meta-analysis
The typical reason for not doing meta-analysis is high heterogeneity across studies
Appropriate quantitative methods exist to handle heterogeneity and may be considered if their assumptions and limitations are acknowledged
Narrative summaries may sometimes be misleading
Contributors and sources: The authors have a longstanding interest in meta-analysis and sources of heterogeneity in clinical research. JPAI had the original idea for the survey. NAP and JPAI extracted the data for the survey and NAP also did the statistical heterogeneity analyses. HRR rekindled the interest in pursuing the project further and the discussion evolved with interactions between JPAI, NPA, and HRR. We thank Iain Chalmers and Alex Sutton for comments on the manuscript. JPAI wrote the manuscript and the coauthors commented on it and approved the final draft.
Competing interests: None declared.
References
- 1.Underhill K, Montgomery P, Operario D. Sexual abstinence only programmes to prevent HIV infection in high income countries: systematic review. BMJ 2007;335:248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chang JT, Morton SC, Rubenstein LZ, Mojica WA, Maglione M, Suttorp MJ, et al. Interventions for the prevention of falls in older adults: systematic review and meta-analysis of randomised clinical trials. BMJ 2004;328:680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ 2003;327:557-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med 2002;21:1539-58. [DOI] [PubMed] [Google Scholar]
- 5.Ioannidis JP, Patsopoulos NA, Evangelou E. Uncertainty in heterogeneity estimates in meta-analyses. BMJ 2007;335:914-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ades AE, Lu G, Higgins JPT. The interpretation of random-effects meta-analysis in decision models. Med Dec Making 2005;25:646-54. [DOI] [PubMed] [Google Scholar]
- 7.Thompson SG, Sharp SJ. Explaining heterogeneity in meta-analysis: a comparison of methods. Stat Med 1999;18:2693-708. [DOI] [PubMed] [Google Scholar]
- 8.Smith TC, Spiegelhalter DJ, Thomas A. Bayesian approaches to random-effects meta-analysis: a comparative study. Stat Med 1995;14:2685-99. [DOI] [PubMed] [Google Scholar]
- 9.Lau J, Ioannidis JP, Schmid CH. Summing up evidence: one answer is not always enough. Lancet 1998;351:123-7. [DOI] [PubMed] [Google Scholar]
- 10.Thompson SG, Higgins JP. How should meta-regression analyses be undertaken and interpreted? Stat Med 2002;21:1559-73. [DOI] [PubMed] [Google Scholar]
- 11.Simmonds MC, Higgins JP, Stewart LA, Tierney JF, Clarke MJ, Thompson SG. Meta-analysis of individual patient data from randomized trials: a review of methods used in practice. Clin Trials 2005;2:209-17. [DOI] [PubMed] [Google Scholar]
- 12.Golfinopoulos V, Salanti G, Pavlidis N, Ioannidis JP. Survival and disease-progression benefits with treatment regimens for advanced colorectal cancer: a meta-analysis. Lancet Oncol 2007;8:898-911. [DOI] [PubMed] [Google Scholar]
- 13.Glenny AM, Altman DG, Song F, Sakarovitch C, Deeks JJ, D’Amico R, et al. Indirect comparisons of competing interventions. Health Technol Assess 2005;9. [DOI] [PubMed]
- 14.Lu G, Ades AE. Combination of direct and indirect evidence in mixed treatment comparisons. Stat Med 2004;23:3105-24. [DOI] [PubMed] [Google Scholar]
- 15.Salanti G, Higgins J, Ades AE, Ioannidis JP. Evaluation of networks of randomized trials. Stat Methods Med Res 2008;17:279-301. [DOI] [PubMed] [Google Scholar]
- 16.Ioannidis JPA. Indirect comparisons: the mesh and mess of clinical trials. Lancet 2006;368:1470-2. [DOI] [PubMed] [Google Scholar]
- 17.Cooper HM, Hedges LV, eds. The handbook of research synthesis New York: Russell Sage Foundation, 1994
- 18.Schünemann HJ, Akl EA, Guyatt GH. Interpreting the results of patient reported outcome measures in clinical trials: the clinician’s perspective. Health Qual Life Outcomes 2006;4:62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jaeschke R, Guyatt G, Shannon H, Walter S, Cook D, Heddle N. Basic statistics for clinicians: 3. Assessing the effects of treatment: measures of association. CMAJ 1995;152:351-7. [PMC free article] [PubMed] [Google Scholar]
- 20.Berkey CS, Anderson JJ, Hoaglin DC. Multiple-outcome meta-analysis of clinical trials. Stat Med 1996;15:537-57. [DOI] [PubMed] [Google Scholar]
- 21.Berkey CS, Hoaglin DC, Antczak-Bouckoms A, Mosteller F, Colditz GA. Meta-analysis of multiple outcomes by regression with random effects. Stat Med 1998;17:2537-50. [DOI] [PubMed] [Google Scholar]
- 22.Riley RD, Abrams KR, Lambert PC, Sutton AJ, Thompson JR. An evaluation of bivariate random-effects meta-analysis for the joint synthesis of two correlated outcomes. Stat Med 2007;26:78-97. [DOI] [PubMed] [Google Scholar]
- 23.Egger M, Schneider M, Davey Smith G. Spurious precision? Meta-analysis of observational studies. BMJ 1998;316:140-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kavvoura FK, Liberopoulos G, Ioannidis JP. Selection in reported epidemiological risks: an empirical assessment. PLoS Med 2007;4:e79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ioannidis JP, Haidich AB, Pappa M, Pantazis N, Kokori SI, Tektonidou MG, et al. Comparison of evidence of treatment effects in randomized and nonrandomized studies. JAMA 2001;286:821-30. [DOI] [PubMed] [Google Scholar]
- 26.Sutton AJ, Abrams KR, Jones DR. Generalized synthesis of evidence and the threat of dissemination bias. The example of electronic fetal heart rate monitoring (EFM). J Clin Epidemiol 2002;55:1013-24. [DOI] [PubMed] [Google Scholar]
- 27.Shrier I, Boivin JF, Steele RJ, Platt RW, Furlan A, Kakuma R, et al. Should meta-analyses of interventions include observational studies in addition to randomized controlled trials? A critical examination of underlying principles. Am J Epidemiol 2007;166:1203-9. [DOI] [PubMed] [Google Scholar]
- 28.Campbell MK, Elbourne DR, Altman DG, CONSORT group. CONSORT statement: extension to cluster randomised trials. BMJ 2004;328:702-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Curtin F, Elbourne D, Altman DG. Meta-analysis combining parallel and cross-over clinical trials. III. The issue of carry-over. Stat Med 2002;21:2161-73. [DOI] [PubMed] [Google Scholar]
- 30.Lu G, Ades AE, Sutton AJ, Cooper NJ, Briggs AH, Caldwell DM. Meta-analysis of mixed treatment comparisons at multiple follow-up times. Stat Med 2007;26:3681-99. [DOI] [PubMed] [Google Scholar]
- 31.Pettiti D. Meta-analysis, decision analysis and cost effectiveness analysis 2nd ed. New York: Oxford University Press, 1999
- 32.Van Sluijs EM, McMinn AM, Griffin SJ. Effectiveness of interventions to promote physical activity in children and adolescents: systematic review of controlled trials. BMJ 2007;335:703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Berlin JA. Benefits of heterogeneity in meta-analysis of data from epidemiologic studies. Am J Epidemiol 1995;142:383-7. [DOI] [PubMed] [Google Scholar]