Abstract
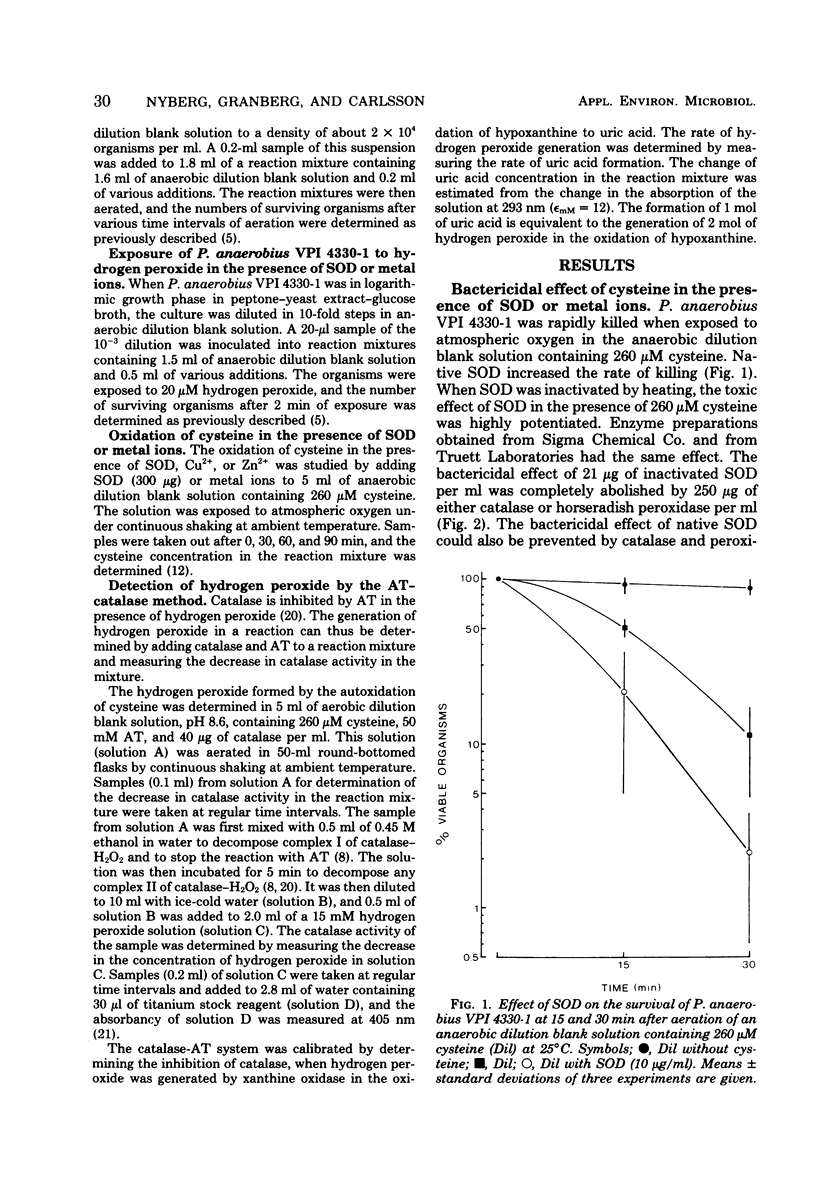
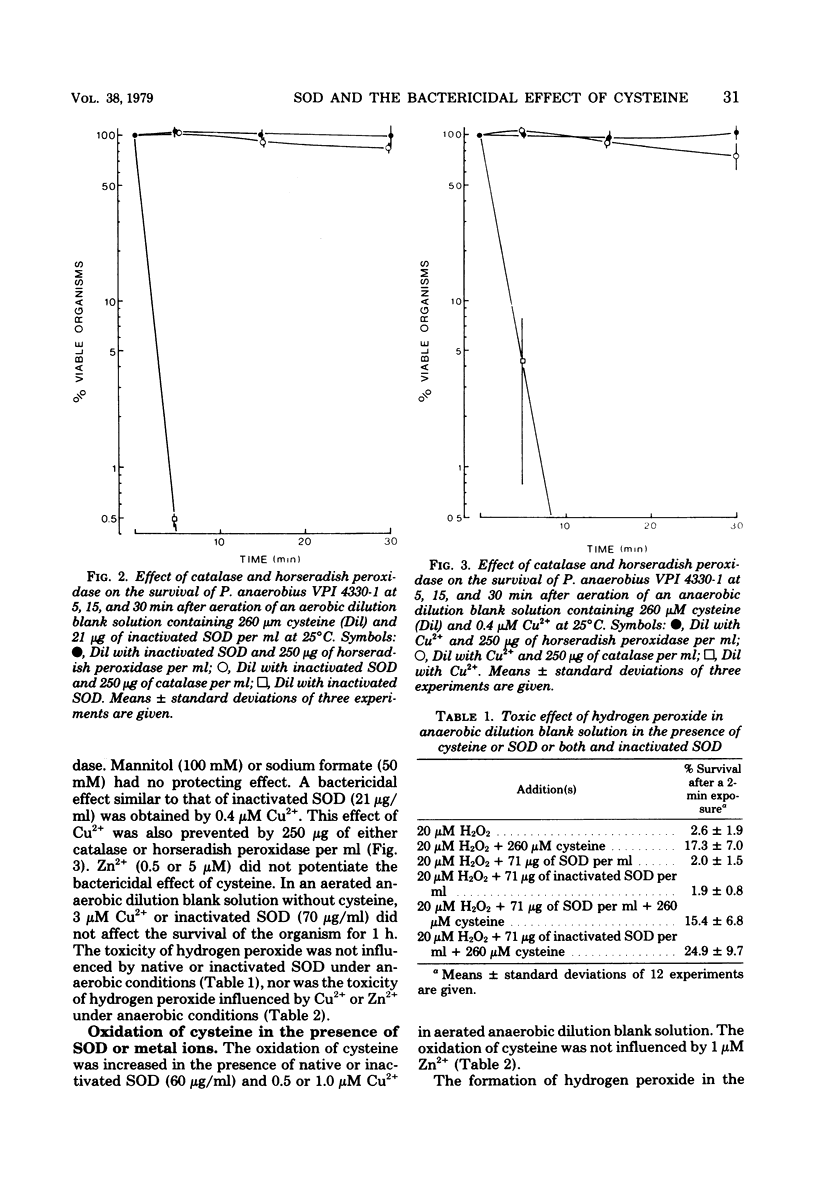
When cysteine is oxidized by oxygen, hydrogen peroxide is formed, and hydrogen peroxide is very toxic to Peptostreptococcus anaerobius VPI 4330-1. Native and inactivated superoxide dismutase increased the rate of oxidation of cysteine and thereby potentiated the toxic effect of cysteine. A similar increase in the rate of oxidation of cysteine and in the toxicity of cysteine was obtained with Cu2+.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Avigad G. An NADH coupled assay system for galactose oxidase. Anal Biochem. 1978 Jun 1;86(2):470–476. doi: 10.1016/0003-2697(78)90771-6. [DOI] [PubMed] [Google Scholar]
- BARRY V. C., CONALTY M. L., DENNENY J. M., WINDER F. Peroxide formation in bacteriological media. Nature. 1956 Sep 15;178(4533):596–597. doi: 10.1038/178596a0. [DOI] [PubMed] [Google Scholar]
- Bhuyan K. C., Bhuyan D. K. Superoxide dismutase of the eye: relative functions of superoxide dismutase and catalase in protecting the ocular lens from oxidative damage. Biochim Biophys Acta. 1978 Aug 3;542(1):28–38. doi: 10.1016/0304-4165(78)90229-5. [DOI] [PubMed] [Google Scholar]
- Boveris A., Martino E., Stoppani A. O. Evaluation of the horseradish peroxidase-scopoletin method for the measurement of hydrogen peroxide formation in biological systems. Anal Biochem. 1977 May 15;80(1):145–158. doi: 10.1016/0003-2697(77)90634-0. [DOI] [PubMed] [Google Scholar]
- Carlsson J., Granberg G. P., Nyberg G. K., Edlund M. B. Bactericidal effect of cysteine exposed to atmospheric oxygen. Appl Environ Microbiol. 1979 Mar;37(3):383–390. doi: 10.1128/aem.37.3.383-390.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carlsson J., Nyberg G., Wrethén J. Hydrogen peroxide and superoxide radical formation in anaerobic broth media exposed to atmospheric oxygen. Appl Environ Microbiol. 1978 Aug;36(2):223–229. doi: 10.1128/aem.36.2.223-229.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavallini D., De Marco C., Duprè S., Rotilio G. The copper catalyzed oxidation of cysteine to cystine. Arch Biochem Biophys. 1969 Mar;130(1):354–361. doi: 10.1016/0003-9861(69)90044-7. [DOI] [PubMed] [Google Scholar]
- Cohen G., Somerson N. L. Catalase-aminotriazole method for measuring secretion of hydrogen peroxide by microorganisms. J Bacteriol. 1969 May;98(2):543–546. doi: 10.1128/jb.98.2.543-546.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dempsey P. M., O'Leary J., Condon S. Polarographic assay of hydrogen peroxide accumulation in microbial cultures. Appl Microbiol. 1975 Feb;29(2):170–174. doi: 10.1128/am.29.2.170-174.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fridovich I. The biology of oxygen radicals. Science. 1978 Sep 8;201(4359):875–880. doi: 10.1126/science.210504. [DOI] [PubMed] [Google Scholar]
- Frölander F., Carlsson J. Bactericidal effect of anaerobic broth exposed to atmospheric oxygen tested on Peptostreptococcus anaerobius. J Clin Microbiol. 1977 Aug;6(2):117–123. doi: 10.1128/jcm.6.2.117-123.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaitonde M. K. A spectrophotometric method for the direct determination of cysteine in the presence of other naturally occurring amino acids. Biochem J. 1967 Aug;104(2):627–633. doi: 10.1042/bj1040627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gregory E. M., Fridovich I. Oxygen metabolism in Lactobacillus plantarum. J Bacteriol. 1974 Jan;117(1):166–169. doi: 10.1128/jb.117.1.166-169.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halliwell B. Biochemical mechanisms accounting for the toxic action of oxygen on living organisms: the key role of superoxide dismutase. Cell Biol Int Rep. 1978 Mar;2(2):113–128. doi: 10.1016/0309-1651(78)90032-2. [DOI] [PubMed] [Google Scholar]
- Harmon S. M., Kautter D. A. Recovery of clostridia on catalase-treated plating media. Appl Environ Microbiol. 1977 Apr;33(4):762–770. doi: 10.1128/aem.33.4.762-770.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodgson E. K., Fridovich I. The interaction of bovine erythrocyte superoxide dismutase with hydrogen peroxide: chemiluminescence and peroxidation. Biochemistry. 1975 Dec 2;14(24):5299–5303. doi: 10.1021/bi00695a011. [DOI] [PubMed] [Google Scholar]
- Hodgson E. K., Fridovich I. The interaction of bovine erythrocyte superoxide dismutase with hydrogen peroxide: inactivation of the enzyme. Biochemistry. 1975 Dec 2;14(24):5294–5299. doi: 10.1021/bi00695a010. [DOI] [PubMed] [Google Scholar]
- MARGOLIASH E., NOVOGRODSKY A., SCHEJTER A. Irreversible reaction of 3-amino-1:2:4-triazole and related inhibitors with the protein of catalase. Biochem J. 1960 Feb;74:339–348. doi: 10.1042/bj0740339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin S. E., Flowers R. S., Ordal Z. J. Catalase: its effect on microbial enumeration. Appl Environ Microbiol. 1976 Nov;32(5):731–734. doi: 10.1128/aem.32.5.731-734.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCord J. M., Keele B. B., Jr, Fridovich I. An enzyme-based theory of obligate anaerobiosis: the physiological function of superoxide dismutase. Proc Natl Acad Sci U S A. 1971 May;68(5):1024–1027. doi: 10.1073/pnas.68.5.1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen J., Davis L. The oxidation of dithiothreitol by peroxidases and oxygen. Biochim Biophys Acta. 1976 Sep 14;445(2):324–329. doi: 10.1016/0005-2744(76)90086-3. [DOI] [PubMed] [Google Scholar]
- PROOM H., WOIWOD A. J., BARNES J. M., ORBELL W. G. A growth-inhibitory effect on Shigella dysenteriae which occurs with some batches of nutrient agar and is associated with the production of peroxide. J Gen Microbiol. 1950 May;4(2):270–276. doi: 10.1099/00221287-4-2-270. [DOI] [PubMed] [Google Scholar]
- TAKAGI T., ISEMURA T. ACCELERATING EFFECT OF COPPER ION ON THE REACTIVATION OF REDUCED TAKA-AMYLASE A THROUGH CATALYSIS OF THE OXIDATION OF SULFHYDRYL GROUPS. J Biochem. 1964 Oct;56:344–350. doi: 10.1093/oxfordjournals.jbchem.a127999. [DOI] [PubMed] [Google Scholar]
- Wyss O., Clark J. B., Haas F., Stone W. S. The Role of Peroxide in the Biological Effects of Irradiated Broth. J Bacteriol. 1948 Jul;56(1):51–57. [PMC free article] [PubMed] [Google Scholar]
- Yamada T., Carlsson J. Regulation of lactate dehydrogenase and change of fermentation products in streptococci. J Bacteriol. 1975 Oct;124(1):55–61. doi: 10.1128/jb.124.1.55-61.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]


